66 results
Cover crop management strategies affect weeds and profitability of organic no-till soybean
-
- Journal:
- Renewable Agriculture and Food Systems / Volume 39 / 2024
- Published online by Cambridge University Press:
- 19 January 2024, e3
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
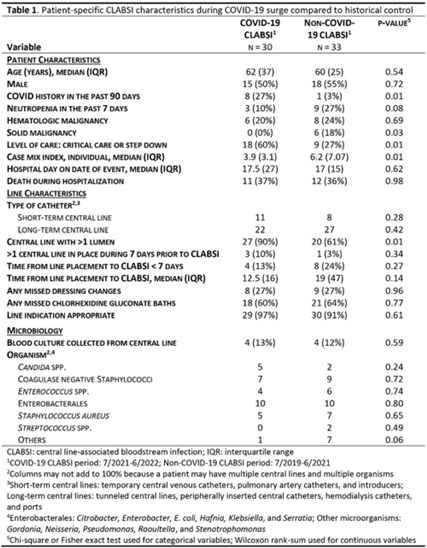
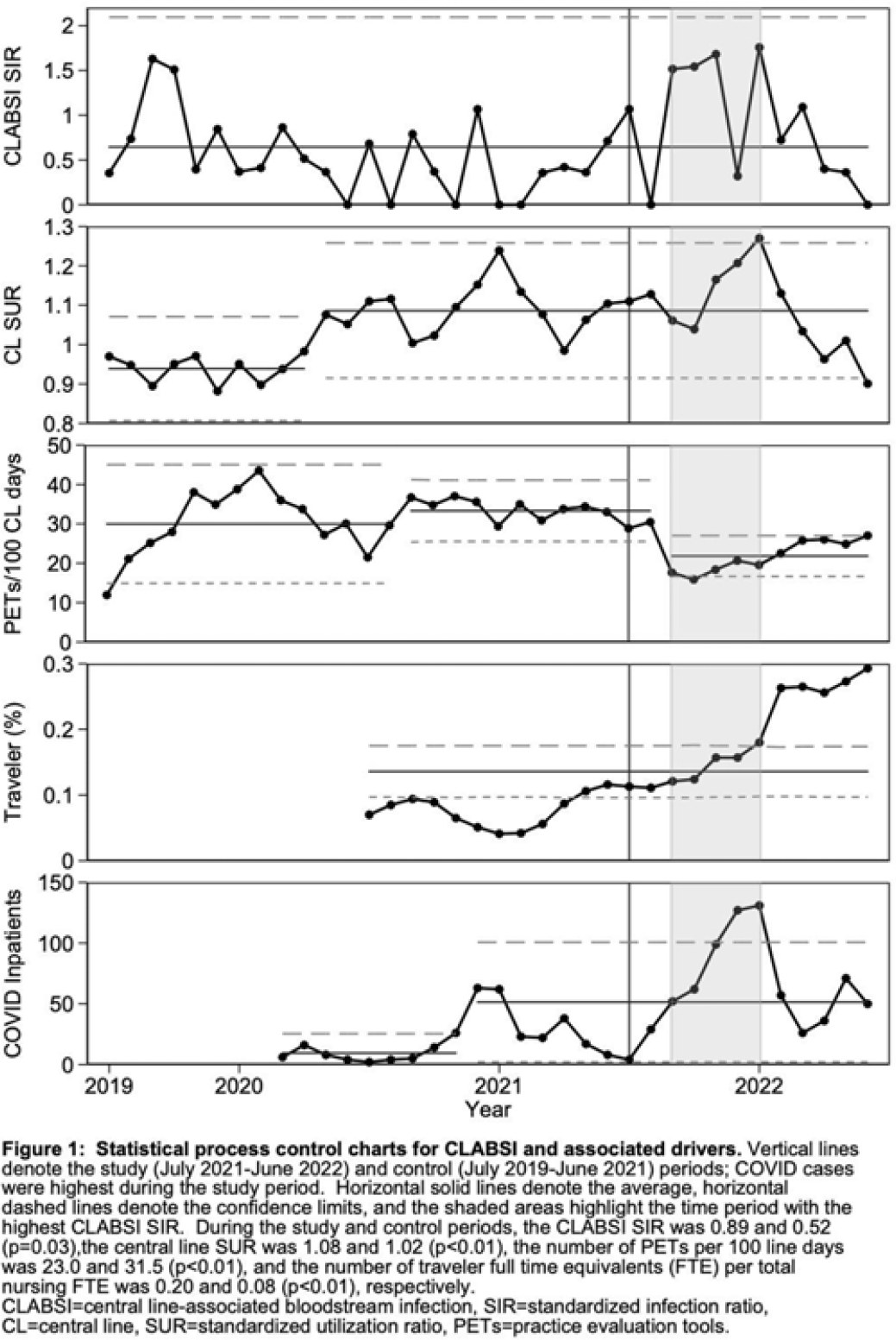
Investigating potential drivers of increased central-line–associated bloodstream infections during the SARS-CoV-2 omicron-variant surge
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s46-s47
-
- Article
-
- You have access
- Open access
- Export citation
Analysis of seasonal variation of antibiotic prescribing for respiratory tract diagnoses in primary care practices
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 05 September 2023, e147
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
A comparison of National Healthcare Safety Network surveillance definitions to physician review for central-line–associated bloodstream infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 12 / December 2023
- Published online by Cambridge University Press:
- 23 June 2023, pp. 2062-2064
- Print publication:
- December 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Association between in situ steroids and spine surgical site infections among instrumented procedures
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 10 / October 2023
- Published online by Cambridge University Press:
- 08 March 2023, pp. 1596-1600
- Print publication:
- October 2023
-
- Article
- Export citation
Patient-centered care to the detriment of the standardized infection ratio
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 3 / March 2023
- Published online by Cambridge University Press:
- 08 November 2022, pp. 524-525
- Print publication:
- March 2023
-
- Article
-
- You have access
- HTML
- Export citation
A survey study of the association between active utilization of National Healthcare Safety Network resources and central-line–associated bloodstream infection reporting
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 15 July 2022, pp. 962-964
- Print publication:
- June 2023
-
- Article
- Export citation
Angiography and Pre-operative Embolization of an Extra-axial, Supratentorial Atypical Teratoid Rhabdoid Tumor
-
- Journal:
- Canadian Journal of Neurological Sciences / Volume 50 / Issue 2 / March 2023
- Published online by Cambridge University Press:
- 03 January 2022, pp. 294-296
-
- Article
-
- You have access
- HTML
- Export citation
Teaching for implementation: A framework for building implementation research and practice capacity within the translational science workforce
-
- Journal:
- Journal of Clinical and Translational Science / Volume 5 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 05 July 2021, e147
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Development and validation of antibiotic stewardship metrics for outpatient respiratory tract diagnoses and association of provider characteristics with inappropriate prescribing
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 1 / January 2022
- Published online by Cambridge University Press:
- 02 June 2021, pp. 56-63
- Print publication:
- January 2022
-
- Article
- Export citation
The Effect of Total Household Decolonization on Clearance of Colonization With Methicillin-Resistant Staphylococcus aureus
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 37 / Issue 10 / October 2016
- Published online by Cambridge University Press:
- 28 July 2016, pp. 1226-1233
- Print publication:
- October 2016
-
- Article
- Export citation
Contributors
-
-
- Book:
- The Cambridge Dictionary of Philosophy
- Published online:
- 05 August 2015
- Print publication:
- 27 April 2015, pp ix-xxx
-
- Chapter
- Export citation
Risk Factors for Recurrent Colonization With Methicillin-Resistant Staphylococcus aureus in Community-Dwelling Adults and Children
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 36 / Issue 7 / July 2015
- Published online by Cambridge University Press:
- 14 April 2015, pp. 786-793
- Print publication:
- July 2015
-
- Article
- Export citation
Contributors
-
-
- Book:
- Health and Education in Early Childhood
- Published online:
- 05 February 2015
- Print publication:
- 19 February 2015, pp xiii-xiv
-
- Chapter
- Export citation
Multilevel assessment of the neurobiological threat system in depressed adolescents: Interplay between the limbic system and hypothalamic–pituitary–adrenal axis
-
- Journal:
- Development and Psychopathology / Volume 26 / Issue 4pt2 / November 2014
- Published online by Cambridge University Press:
- 25 November 2014, pp. 1321-1335
-
- Article
- Export citation
Measurement of Implementation Components and Contextual Factors in a Two-State Healthcare Quality Initiative to Reduce Ventilator-Associated Pneumonia
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 35 / Issue S3 / October 2014
- Published online by Cambridge University Press:
- 10 May 2016, pp. S116-S123
- Print publication:
- October 2014
-
- Article
-
- You have access
- Export citation
List of contributors
-
-
- Book:
- Contraception
- Published online:
- 05 August 2013
- Print publication:
- 11 July 2013, pp vii-viii
-
- Chapter
- Export citation
Contributors
-
-
- Book:
- The Cambridge Dictionary of Christianity
- Published online:
- 05 August 2012
- Print publication:
- 20 September 2010, pp xi-xliv
-
- Chapter
- Export citation
Contributors
-
-
- Book:
- Morbid Obesity
- Published online:
- 04 May 2010
- Print publication:
- 11 March 2010, pp -
-
- Chapter
- Export citation
The Road That I See: Implications of New Reproductive Technologies
-
- Journal:
- Cambridge Quarterly of Healthcare Ethics / Volume 4 / Issue 3 / Summer 1995
- Published online by Cambridge University Press:
- 29 July 2009, pp. 351-354
-
- Article
- Export citation