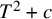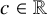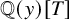326 results
X-Ray Study of Interactions between Montmorillonite Platelets
-
- Journal:
- Clays and Clay Minerals / Volume 15 / February 1967
- Published online by Cambridge University Press:
- 01 July 2024, pp. 321-330
-
- Article
- Export citation
D.4 Safety and efficacy of delandistrogene moxeparvovec versus placebo in Duchenne muscular dystrophy (EMBARK): Pivotal Phase 3 primary results
-
- Journal:
- Canadian Journal of Neurological Sciences / Volume 51 / Issue s1 / June 2024
- Published online by Cambridge University Press:
- 24 May 2024, pp. S9-S10
-
- Article
-
- You have access
- Export citation
Lower Bounds for the Canonical Height of a Unicritical Polynomial and Capacity
- Part of
-
- Journal:
- Forum of Mathematics, Sigma / Volume 12 / 2024
- Published online by Cambridge University Press:
- 02 April 2024, e45
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
The effect of a low-calorie diet on depressive symptoms in individuals with overweight or obesity: a systematic review and meta-analysis of interventional studies – ADDENDUM
-
- Journal:
- Psychological Medicine / Volume 54 / Issue 8 / June 2024
- Published online by Cambridge University Press:
- 22 January 2024, p. 1886
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
The effect of a low-calorie diet on depressive symptoms in individuals with overweight or obesity: a systematic review and meta-analysis of interventional studies
-
- Journal:
- Psychological Medicine / Volume 54 / Issue 8 / June 2024
- Published online by Cambridge University Press:
- 12 December 2023, pp. 1671-1683
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Differing impact of the COVID-19 pandemic on youth mental health: combined population and clinical study
-
- Journal:
- BJPsych Open / Volume 9 / Issue 6 / November 2023
- Published online by Cambridge University Press:
- 20 November 2023, e217
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Validation of histological and visual scoring systems for foot-pad dermatitis in broiler chickens
-
- Journal:
- Animal Welfare / Volume 29 / Issue 2 / May 2020
- Published online by Cambridge University Press:
- 01 January 2023, pp. 185-196
-
- Article
-
- You have access
- Open access
- Export citation
The animal welfare implications of civet coffee tourism in Bali
- Part of
-
- Journal:
- Animal Welfare / Volume 25 / Issue 2 / May 2016
- Published online by Cambridge University Press:
- 01 January 2023, pp. 199-205
-
- Article
- Export citation
Comparison of inpatient and outpatient palliative sedation practice – A prospective observational study
-
- Journal:
- Palliative & Supportive Care , First View
- Published online by Cambridge University Press:
- 18 November 2022, pp. 1-7
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Implications of the loess record for Holocene climate and human settlement in Heye Catchment, Jiuzhaigou, eastern Tibetan Plateau, Sichuan, China
-
- Journal:
- Quaternary Research / Volume 112 / March 2023
- Published online by Cambridge University Press:
- 07 October 2022, pp. 36-50
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Developmental plasticity in deep time: a window to population ecological inference
-
- Journal:
- Paleobiology / Volume 49 / Issue 2 / May 2023
- Published online by Cambridge University Press:
- 04 August 2022, pp. 259-270
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Depression, anxiety and PTSD symptoms before and during the COVID-19 pandemic in the UK
-
- Journal:
- Psychological Medicine / Volume 53 / Issue 12 / September 2023
- Published online by Cambridge University Press:
- 26 July 2022, pp. 5428-5441
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Integrative Structural Analysis of Human Nuclear Pore Complex
-
- Journal:
- Microscopy and Microanalysis / Volume 28 / Issue S1 / August 2022
- Published online by Cambridge University Press:
- 22 July 2022, p. 1114
- Print publication:
- August 2022
-
- Article
-
- You have access
- Export citation
Interplay between the Genetics of Personality Traits, severe Psychiatric Disorders, and COVID-19 Host Genetics in the Susceptibility to SARS-CoV-2 Infection - ADDENDUM
-
- Journal:
- BJPsych Open / Volume 7 / Issue 6 / November 2021
- Published online by Cambridge University Press:
- 18 November 2021, e206
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Socio-political context as determinant of childhood maltreatment: a population-based study among women and men in East and West Germany
-
- Journal:
- Epidemiology and Psychiatric Sciences / Volume 30 / 2021
- Published online by Cambridge University Press:
- 18 November 2021, e72
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Interplay between the genetics of personality traits, severe psychiatric disorders and COVID-19 host genetics in the susceptibility to SARS-CoV-2 infection
-
- Journal:
- BJPsych Open / Volume 7 / Issue 6 / November 2021
- Published online by Cambridge University Press:
- 07 October 2021, e188
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Characterisation of age and polarity at onset in bipolar disorder
-
- Journal:
- The British Journal of Psychiatry / Volume 219 / Issue 6 / December 2021
- Published online by Cambridge University Press:
- 25 August 2021, pp. 659-669
- Print publication:
- December 2021
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Tailored nanoscale plasmon-enhanced vibrational electron spectroscopy
-
- Journal:
- Microscopy and Microanalysis / Volume 27 / Issue S1 / August 2021
- Published online by Cambridge University Press:
- 30 July 2021, pp. 320-321
- Print publication:
- August 2021
-
- Article
-
- You have access
- Export citation
Development of the Response to Fearful Situations Scale
-
- Journal:
- Behavioural and Cognitive Psychotherapy / Volume 48 / Issue 6 / November 2020
- Published online by Cambridge University Press:
- 28 July 2020, pp. 688-704
- Print publication:
- November 2020
-
- Article
- Export citation