578 results
The equitable impact of sugary drink taxation structures on sugary drink consumption among Canadians: a modelling study using the 2015 Canadian Community Health Survey-Nutrition
-
- Journal:
- Public Health Nutrition / Accepted manuscript
- Published online by Cambridge University Press:
- 15 April 2024, pp. 1-27
-
- Article
-
- You have access
- Open access
- Export citation
546 Using Contingency Management to Understand the Cardiovascular, Immune and Psychosocial Benefits of Reduced Cocaine Use: A Protocol for a Randomized Controlled Trial
- Part of
-
- Journal:
- Journal of Clinical and Translational Science / Volume 8 / Issue s1 / April 2024
- Published online by Cambridge University Press:
- 03 April 2024, p. 163
-
- Article
-
- You have access
- Open access
- Export citation
The prevalence of gram-negative bacteria with difficult-to-treat resistance and utilization of novel β-lactam antibiotics in the southeastern United States
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue 1 / 2024
- Published online by Cambridge University Press:
- 18 March 2024, e35
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Integral equivariant cohomology of affine Grassmannians
- Part of
-
- Journal:
- Canadian Mathematical Bulletin , First View
- Published online by Cambridge University Press:
- 08 February 2024, pp. 1-15
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Simultaneous submission of seven CTSA proposals: UM1, K12, R25, T32-predoctoral, T32-postdoctoral, and RC2: strategies, evaluation, and lessons learned
-
- Journal:
- Journal of Clinical and Translational Science / Volume 8 / Issue 1 / 2024
- Published online by Cambridge University Press:
- 25 January 2024, e33
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Investigating oral human papillomavirus co-infection with Neisseria gonorrhoeae and Chlamydia trachomatis
-
- Journal:
- Epidemiology & Infection / Volume 152 / 2024
- Published online by Cambridge University Press:
- 08 January 2024, e8
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
A severe acute respiratory coronavirus virus 2 (SARS-CoV-2) nosocomial cluster with inter-facility spread: Lessons learned
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 5 / May 2024
- Published online by Cambridge University Press:
- 04 January 2024, pp. 635-643
- Print publication:
- May 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
The impact of minimally invasive surgical approaches on surgical-site infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 5 / May 2024
- Published online by Cambridge University Press:
- 03 January 2024, pp. 557-561
- Print publication:
- May 2024
-
- Article
- Export citation
The NEONATE score predicts freedom from interstage mortality or transplant in a modern cohort
-
- Journal:
- Cardiology in the Young , First View
- Published online by Cambridge University Press:
- 28 November 2023, pp. 1-8
-
- Article
- Export citation
Strategies used for the COVID-OUT decentralized trial of outpatient treatment of SARS-CoV-2
-
- Journal:
- Journal of Clinical and Translational Science / Volume 7 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 07 November 2023, e242
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
COVID-19 and British Columbia’s volunteer search and rescue workers: risk recognition and risk mitigation
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 03 November 2023, e195
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
19 - Structure Constants
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp 350-369
-
- Chapter
- Export citation
15 - Homogeneous Varieties
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp 265-289
-
- Chapter
- Export citation
14 - Symplectic Schubert Polynomials
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp 240-264
-
- Chapter
- Export citation
Appendix A - Algebraic Topology
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp 370-387
-
- Chapter
- Export citation
2 - Defining Equivariant Cohomology
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp 9-25
-
- Chapter
- Export citation
Preface
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp xi-xvi
-
- Chapter
- Export citation
11 - Degeneracy Loci
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp 182-207
-
- Chapter
- Export citation
6 - Conics
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp 79-92
-
- Chapter
- Export citation
9 - Schubert Calculus on Grassmannians
-
- Book:
- Equivariant Cohomology in Algebraic Geometry
- Published online:
- 07 October 2023
- Print publication:
- 26 October 2023, pp 126-151
-
- Chapter
- Export citation



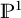 .
.