Articles
Characterization of GaN Grown on SiC on Si/SiO2/Si by Metalorganic Chemical Vapor Deposition
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1171-1174
-
- Article
- Export citation
Nanostructure of GaN and SiC Nanowires Based on Carbon Nanotubes
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1175-1177
-
- Article
- Export citation
Intercalation of ε-caprolactam Ions into Inorganic Hosts
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1178-1180
-
- Article
- Export citation
Enthalpy of Formation of Rare-earth Silicates Y2SiO5 and Yb2SiO5 and N-containing Silicate Y10(SiO4)6N2
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1181-1185
-
- Article
- Export citation
Two- and Three-dimensional Arrays of Magnetic Microspheres
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1186-1189
-
- Article
- Export citation
Microstructural and Ferroelectric Properties of a Chemical Solution Deposited Epitaxial PbZr0.5 Ti0.5O3 Thin Film on a SrRuO3/SrTiO3 Substrate
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1190-1193
-
- Article
- Export citation
Epitaxial Growth of Patterned SrBi2Ta2O9 Lines by Channel Stamping
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1194-1196
-
- Article
- Export citation
Thin Film Microstructure Control Using Glancing Angle Deposition by Sputtering
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1197-1199
-
- Article
- Export citation
Thermoelectric Properties and Power Factor of YBa2Cu3O6+xwith Rare-earth Dopants
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1200-1203
-
- Article
- Export citation
Speeding up Film Deposition Rate: Its Effects on Microstructures of YBa2Cu3Oy Superconducting Thick Films
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1204-1211
-
- Article
- Export citation
Effects of the Heating Rate on Conversion of the Precursor Powders Used for Melt Processes into YBa2Cu3O7−y
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1212-1220
-
- Article
- Export citation
Electrochemical Synthesis and Characterization of La2CuO4+δ Single Crystals
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1221-1226
-
- Article
- Export citation
Photoluminescence Spectral Lines Identification in Ho3+-, Er3+-, and Tm3+-doped MgxZn1–xSe Single Crystals
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1227-1234
-
- Article
- Export citation
Local and Electronic Structure of Siloxene
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1235-1237
-
- Article
- Export citation
Atmospheric Pressure Chemical Vapor Deposition Growth Window for Undoped Gallium Antimonide
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1238-1245
-
- Article
- Export citation
Stress Relaxation in Al–Cu and Al–Si–Cu Thin Films
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1246-1254
-
- Article
- Export citation
Changes in Preferred Orientation of Pt Thin Films Deposited by dc Magnetron Sputtering Using Ar/O2 Gas Mixtures
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1255-1260
-
- Article
- Export citation
The Role of Ni in the Formation of Low Resistance Ni–Ge–Au Ohmic Contacts to n+ GaAs Heterostructures
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1261-1271
-
- Article
- Export citation
Nanoscale Heterogeneities in Amorphous Semiconductorxmetal1–x Alloys: A Small-angle X-ray Scattering Study
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1272-1278
-
- Article
- Export citation
Effect of Ti Substitution on the Microstructure and Properties of Zr–Mn–V–Ni AB2 Type Hydride Electrode Alloys
-
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1279-1285
-
- Article
- Export citation


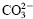 in its double hydroxide gallery and also that of ε-caprolactam cation (ε–CL
in its double hydroxide gallery and also that of ε-caprolactam cation (ε–CL