22 results
Design of an ICF plant using a nuclear-driven solid-state laser
-
- Journal:
- Laser and Particle Beams / Volume 13 / Issue 1 / March 1995
- Published online by Cambridge University Press:
- 09 March 2009, pp. 95-109
-
- Article
- Export citation
Microwave excitation and applications of an elliptical excimer lamp
-
- Journal:
- Laser and Particle Beams / Volume 16 / Issue 3 / September 1998
- Published online by Cambridge University Press:
- 09 March 2009, pp. 509-524
-
- Article
- Export citation
Synergism in inertial confinement fusion: a total direct energy conversion package
-
- Journal:
- Laser and Particle Beams / Volume 7 / Issue 3 / August 1989
- Published online by Cambridge University Press:
- 09 March 2009, pp. 449-466
-
- Article
- Export citation
Charged particle spectra from U-235 and B-10 micropellets and slab coatings
-
- Journal:
- Laser and Particle Beams / Volume 2 / Issue 2 / May 1984
- Published online by Cambridge University Press:
- 09 March 2009, pp. 201-211
-
- Article
- Export citation
Sensitivity analysis of
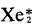 excimer fluorescence generated from charged particle excitation
excimer fluorescence generated from charged particle excitation
-
- Journal:
- Laser and Particle Beams / Volume 5 / Issue 1 / February 1987
- Published online by Cambridge University Press:
- 09 March 2009, pp. 125-132
-
- Article
- Export citation
Nuclear driven flashlamps
-
- Journal:
- Laser and Particle Beams / Volume 6 / Issue 1 / February 1988
- Published online by Cambridge University Press:
- 09 March 2009, pp. 25-62
-
- Article
- Export citation
Effects of helium buffer gas on the atomic carbon nuclear-pumped laser at low power densities
-
- Journal:
- Laser and Particle Beams / Volume 9 / Issue 4 / December 1991
- Published online by Cambridge University Press:
- 09 March 2009, pp. 829-839
-
- Article
- Export citation
Diamond photovoltaic cells as a first-wall material and energy conversion system for inertial confinement fusion
-
- Journal:
- Laser and Particle Beams / Volume 11 / Issue 1 / March 1993
- Published online by Cambridge University Press:
- 09 March 2009, pp. 65-79
-
- Article
- Export citation
Micro Characterization of the Diffusion of Cr in Natural Diamond Particles
-
- Journal:
- Microscopy and Microanalysis / Volume 12 / Issue S02 / August 2006
- Published online by Cambridge University Press:
- 31 July 2006, pp. 1026-1027
- Print publication:
- August 2006
-
- Article
-
- You have access
- Export citation
Surface Phenomena of the Thin Diamond-Like Carbon Films
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 555 / 1998
- Published online by Cambridge University Press:
- 10 February 2011, 345
- Print publication:
- 1998
-
- Article
- Export citation
Boron diffusion into diamond under electric bias
-
- Journal:
- Journal of Materials Research / Volume 12 / Issue 5 / May 1997
- Published online by Cambridge University Press:
- 31 January 2011, pp. 1169-1171
- Print publication:
- May 1997
-
- Article
- Export citation
Influence of Forced Diffusion of Boron on Electrical Conductivity of Diamond Films
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 423 / 1996
- Published online by Cambridge University Press:
- 15 February 2011, 649
- Print publication:
- 1996
-
- Article
- Export citation
Determination of Hydrogen in CVD Diamond by Notched Neutron Spectrum Technique and Ftir
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 416 / 1995
- Published online by Cambridge University Press:
- 10 February 2011, 361
- Print publication:
- 1995
-
- Article
- Export citation
Boron Diffusion Coefficient in Diamond
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 416 / 1995
- Published online by Cambridge University Press:
- 10 February 2011, 467
- Print publication:
- 1995
-
- Article
- Export citation
Raman and FTIR Study of Neutron Irradiated CVD Diamond
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 416 / 1995
- Published online by Cambridge University Press:
- 10 February 2011, 223
- Print publication:
- 1995
-
- Article
- Export citation
Conductivity and Ftir Measurements of the Hydrogen Content of Heat Treated Diamond Films
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 339 / 1994
- Published online by Cambridge University Press:
- 21 February 2011, 643
- Print publication:
- 1994
-
- Article
- Export citation
Diffusion of Impurities Under Bias in CVD Diamond Films
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 339 / 1994
- Published online by Cambridge University Press:
- 21 February 2011, 601
- Print publication:
- 1994
-
- Article
- Export citation
Phase Transformation of Smooth Diamond Films Grown by hot Filament Chemical Vapor Deposition on Positively Biased Silicon Substrates.
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 339 / 1994
- Published online by Cambridge University Press:
- 21 February 2011, 325
- Print publication:
- 1994
-
- Article
- Export citation
Electrical Characterization of Aluminum Nitride Films on Silicon Grown by Chemical Vapor Deposition
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 339 / 1994
- Published online by Cambridge University Press:
- 21 February 2011, 637
- Print publication:
- 1994
-
- Article
- Export citation
Photoresponse Study of Polycrystalline Diamond thin Film Schottky Diodes
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 339 / 1994
- Published online by Cambridge University Press:
- 21 February 2011, 191
- Print publication:
- 1994
-
- Article
- Export citation



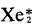 excimer fluorescence generated from charged particle excitation
excimer fluorescence generated from charged particle excitation
