64 results
Antimicrobial use before and during COVID-19: data from 108 Veterans Affairs medical centers
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue 1 / 2024
- Published online by Cambridge University Press:
- 07 August 2024, e109
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Research agenda for transmission prevention within the Veterans Health Administration, 2024–2028
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 11 April 2024, pp. 1-10
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
They’re Still There, He’s All Gone: American Fatalities in Foreign Wars and Right-Wing Radicalization at Home
-
- Journal:
- American Political Science Review / Volume 118 / Issue 3 / August 2024
- Published online by Cambridge University Press:
- 19 October 2023, pp. 1577-1583
- Print publication:
- August 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
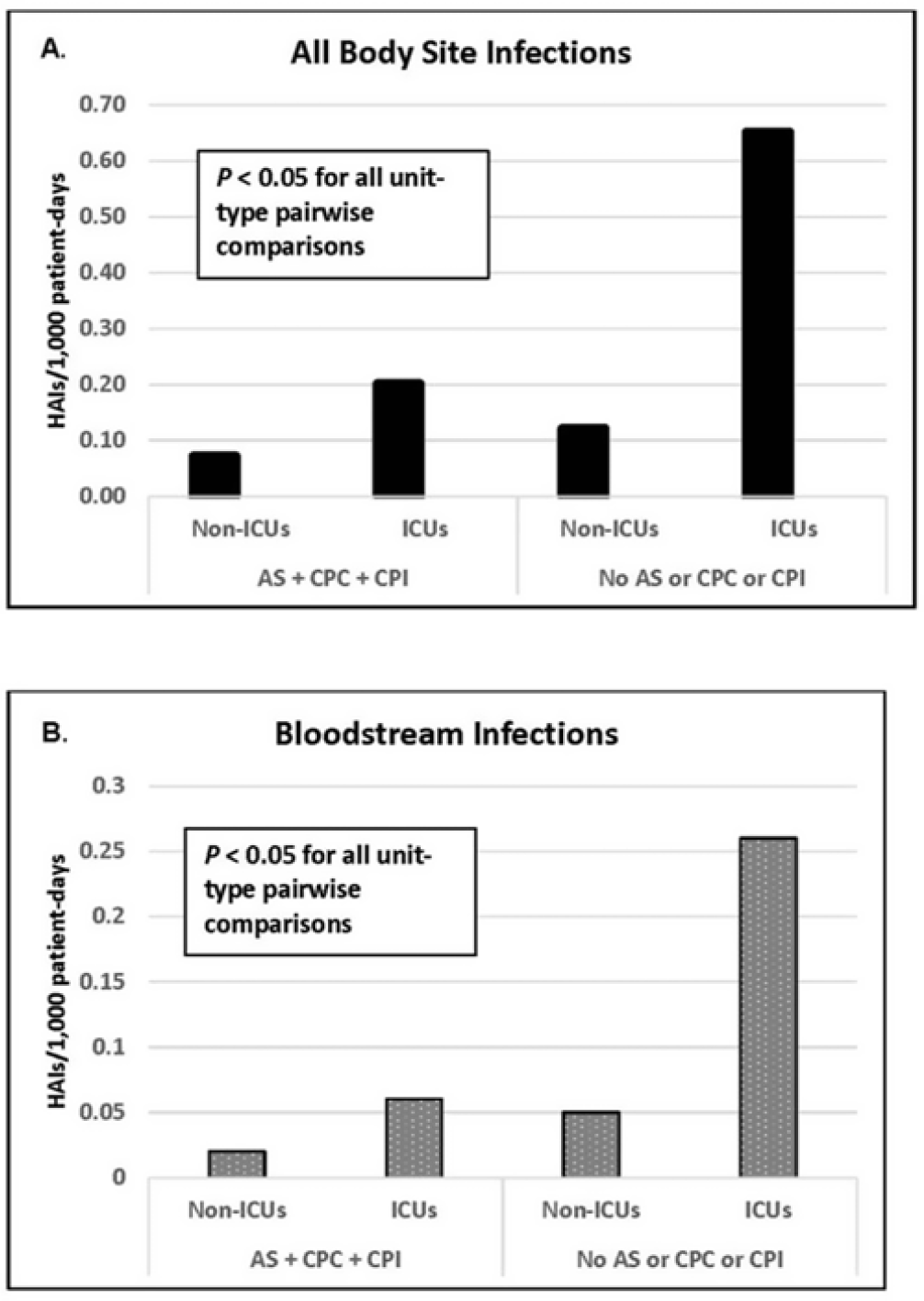
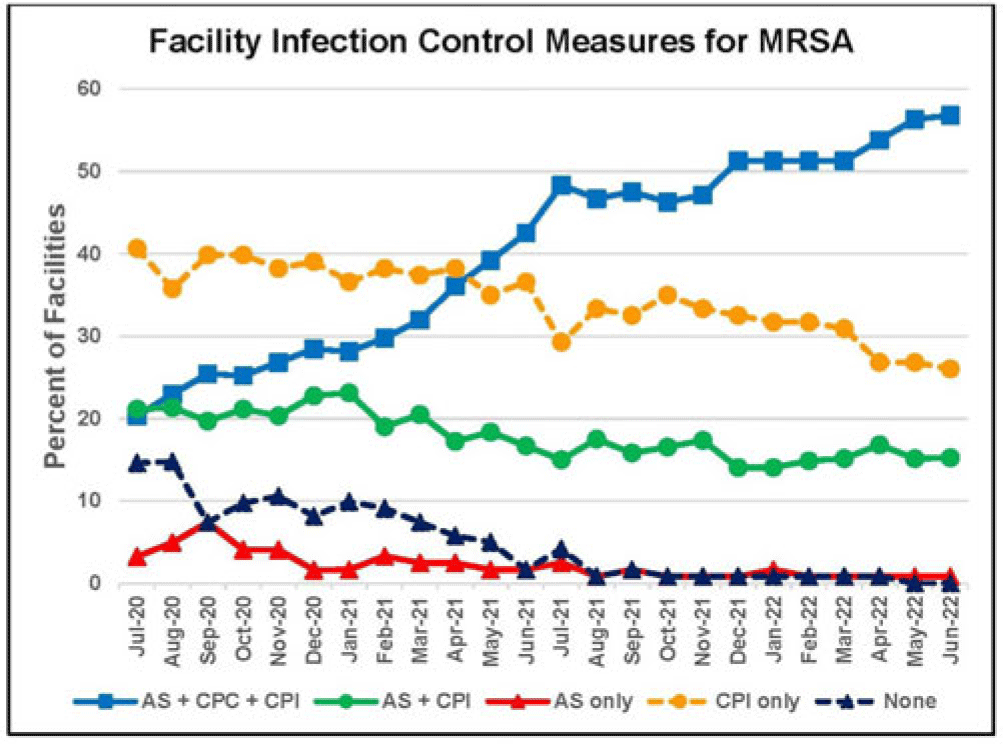
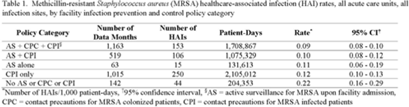
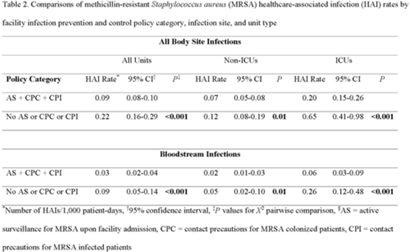
Active surveillance and contact precautions for preventing MRSA healthcare-associated infections during the COVID-19 pandemic
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s117-s118
-
- Article
-
- You have access
- Open access
- Export citation
Antimicrobial physician and pharmacist experience and perception of an antimicrobial Self-Stewardship Time-Out Program (SSTOP) intervention at eight Veterans’ Affairs medical centers
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 9 / September 2023
- Published online by Cambridge University Press:
- 24 January 2023, pp. 1511-1514
- Print publication:
- September 2023
-
- Article
- Export citation
Increased carbapenemase testing following implementation of national VA guidelines for carbapenem-resistant Enterobacterales (CRE)
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 02 June 2022, e88
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Real World Performance of SARS-CoV-2 Antigen Rapid Diagnostic Tests in Various Clinical Settings
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Accepted manuscript
- Published online by Cambridge University Press:
- 02 March 2022, pp. 1-20
-
- Article
- Export citation
The Hidden Hand: Middle East Fears of Conspiracy, Daniel Pipes, New York: St. Martin's Press, 1996, xii + 404 pp.
-
- Journal:
- Iranian Studies / Volume 30 / Issue 1-2 / Spring Winter 1997
- Published online by Cambridge University Press:
- 01 January 2022, pp. 177-181
- Print publication:
- Spring Winter 1997
-
- Article
- Export citation
Fihrist-i asnād-i qadīm-i vizārat-i umūr-i khārijah: dawrān-i Qājārīyah, 1124–1316, Tehran: Institute for Political and International Studies, 1371 Sh./1992, xiii + 598 pp., 4,500 rials.
-
- Journal:
- Iranian Studies / Volume 29 / Issue 3-4 / Summer Fall 1996
- Published online by Cambridge University Press:
- 01 January 2022, pp. 394-395
- Print publication:
- Summer Fall 1996
-
- Article
- Export citation
Stumbling through the “Open Door”: The U.S. in Persia and the Standard-Sinclair Oil Dispute, 1920–1925
-
- Journal:
- Iranian Studies / Volume 28 / Issue 3-4 / Summer Fall 1995
- Published online by Cambridge University Press:
- 01 January 2022, pp. 203-229
- Print publication:
- Summer Fall 1995
-
- Article
- Export citation
Hippocampal volume and volume asymmetry prospectively predict PTSD symptom emergence among Iraq-deployed soldiers
-
- Journal:
- Psychological Medicine / Volume 53 / Issue 5 / April 2023
- Published online by Cambridge University Press:
- 22 November 2021, pp. 1906-1913
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Implementation of an Antibiotic Timeout at Veterans’ Affairs Medical Centers (VAMC): COVID-19 Facilitators and Barriers
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s3
-
- Article
-
- You have access
- Open access
- Export citation
Chapter 2 - How Much Does the Family Want to Be Involved in Decision-Making?
-
-
- Book:
- Shared Decision Making in Adult Critical Care
- Published online:
- 27 May 2021
- Print publication:
- 17 June 2021, pp 7-12
-
- Chapter
- Export citation
Inpatient antibiotic utilization in the Veterans’ Health Administration during the coronavirus disease 2019 (COVID-19) pandemic
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 6 / June 2021
- Published online by Cambridge University Press:
- 20 October 2020, pp. 751-753
- Print publication:
- June 2021
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Risk Factors for Carbapenemase Producing-Carbapenem Resistant Enterobacteriaceae in Those With CRE Positive Cultures
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s376-s377
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Evaluating Healthcare Worker Movements and Patient Interactions Within ICU Rooms
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s222-s224
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Inpatient and Discharge Fluoroquinolone Prescribing in Veterans’ Affairs Hospitals Between 2014 and 2017
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s487-s488
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
A survey of infection control strategies for carbapenem-resistant Enterobacteriaceae in Department of Veterans’ Affairs facilities
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 7 / July 2022
- Published online by Cambridge University Press:
- 22 September 2020, pp. 939-942
- Print publication:
- July 2022
-
- Article
- Export citation
Evaluation of the National Healthcare Safety Network standardized infection ratio risk adjustment for healthcare-facility-onset Clostridioides difficile infection in intensive care, oncology, and hematopoietic cell transplant units in general acute-care hospitals
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue 4 / April 2020
- Published online by Cambridge University Press:
- 13 February 2020, pp. 404-410
- Print publication:
- April 2020
-
- Article
- Export citation
Validation of the SHEA/IDSA severity criteria to predict poor outcomes among inpatients and outpatients with Clostridioides difficile infection
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue 5 / May 2020
- Published online by Cambridge University Press:
- 30 January 2020, pp. 510-516
- Print publication:
- May 2020
-
- Article
- Export citation