33 results
Order and Disorder Relations in the Distribution of the Substitutions in Smectites, Illites and Vermiculites
-
- Journal:
- Clays and Clay Minerals / Volume 22 / Issue 5-6 / December 1974
- Published online by Cambridge University Press:
- 01 July 2024, pp. 379-384
-
- Article
- Export citation
A Kinetic Survey of the Cation Exchange and of the Oxidation of a Vermiculite
-
- Journal:
- Clays and Clay Minerals / Volume 23 / Issue 4 / August 1975
- Published online by Cambridge University Press:
- 01 July 2024, pp. 318-322
-
- Article
- Export citation
Refined Relationships between Chemical Composition of Dioctahedral Fine-Grained Micaceous Minerals and Their Infrared Spectra Within the OH Stretching Region. Part II: The Main Factors Affecting OH Vibrations and Quantitative Analysis
-
- Journal:
- Clays and Clay Minerals / Volume 45 / Issue 2 / April 1997
- Published online by Cambridge University Press:
- 28 February 2024, pp. 170-183
-
- Article
- Export citation
An Improved Model for Structural Transformations of Heat-Treated Aluminous Dioctahedral 2:1 Layer Silicates
-
- Journal:
- Clays and Clay Minerals / Volume 43 / Issue 6 / December 1995
- Published online by Cambridge University Press:
- 28 February 2024, pp. 718-731
-
- Article
- Export citation
Refined Relationships between Chemical Composition of Dioctahedral Fine-Grained Mica Minerals and Their Infrared Spectra within the OH Stretching Region. Part I: Identification of the OH Stretching Bands
-
- Journal:
- Clays and Clay Minerals / Volume 45 / Issue 2 / April 1997
- Published online by Cambridge University Press:
- 28 February 2024, pp. 158-169
-
- Article
- Export citation
Radiofrequency ice dielectric measurements at Summit Station, Greenland
-
- Journal:
- Journal of Glaciology , First View
- Published online by Cambridge University Press:
- 09 October 2023, pp. 1-12
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
In situ, broadband measurement of the radio frequency attenuation length at Summit Station, Greenland
-
- Journal:
- Journal of Glaciology / Volume 68 / Issue 272 / December 2022
- Published online by Cambridge University Press:
- 30 May 2022, pp. 1234-1242
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Population survey of drug abusers attending a Swiss detoxification unit
-
- Journal:
- European Psychiatry / Volume 17 / Issue S1 / May 2002
- Published online by Cambridge University Press:
- 16 April 2020, p. 213s
-
- Article
-
- You have access
- Export citation
Topiramate in multidrug withdrawal – comparison with clonidine
-
- Journal:
- European Psychiatry / Volume 17 / Issue S1 / May 2002
- Published online by Cambridge University Press:
- 16 April 2020, pp. 213s-214s
-
- Article
-
- You have access
- Export citation
Gastroenteric parasite of wild Galliformes in the Italian Alps: implication for conservation management
-
- Journal:
- Parasitology / Volume 147 / Issue 4 / April 2020
- Published online by Cambridge University Press:
- 19 December 2019, pp. 471-477
-
- Article
- Export citation
Diffraction effects calculated for structural models of K-saturated montmorillonite containing different types of defects
-
- Journal:
- Clay Minerals / Volume 19 / Issue 4 / September 1984
- Published online by Cambridge University Press:
- 09 July 2018, pp. 541-561
-
- Article
- Export citation
Caracteristiques structurales d'une smectite dioctaedrique en fonction de l'ordre-desordre dans la distribution des charges electriques: I. Etudes des reflexions 00l
-
- Journal:
- Clay Minerals / Volume 22 / Issue 3 / September 1987
- Published online by Cambridge University Press:
- 09 July 2018, pp. 305-318
-
- Article
- Export citation
Methode diffractometrique de caracterisation des états d'hydratation des smectites stabilité relative des couches d'eau inserées
-
- Journal:
- Clay Minerals / Volume 21 / Issue 2 / June 1986
- Published online by Cambridge University Press:
- 09 July 2018, pp. 111-124
-
- Article
- Export citation
Ferripyrophyllite and related Fe3+-rich 2:1 clays in Recent deposits of Atlantis II Deep, Red Sea
-
- Journal:
- Clay Minerals / Volume 27 / Issue 2 / June 1992
- Published online by Cambridge University Press:
- 09 July 2018, pp. 227-244
-
- Article
- Export citation
Interpretation of the IR spectra of celadonites and glauconites in the region of OH-stretching frequencies
-
- Journal:
- Clay Minerals / Volume 21 / Issue 3 / September 1986
- Published online by Cambridge University Press:
- 09 July 2018, pp. 377-388
-
- Article
- Export citation
Le césium, revelateur de structure des smectites
-
- Journal:
- Clay Minerals / Volume 18 / Issue 1 / March 1983
- Published online by Cambridge University Press:
- 09 July 2018, pp. 11-19
-
- Article
- Export citation
Complementarite des rayons X et de la microscopie electronique pour la determination des diverses phases d'une argile zincifere
-
- Journal:
- Clay Minerals / Volume 20 / Issue 1 / March 1985
- Published online by Cambridge University Press:
- 09 July 2018, pp. 53-67
-
- Article
- Export citation
Dehydroxylation of Fe3+ , Mg-rich dioctahedral micas: (I) structural transformation
-
- Journal:
- Clay Minerals / Volume 35 / Issue 3 / June 2000
- Published online by Cambridge University Press:
- 09 July 2018, pp. 491-504
-
- Article
- Export citation
X-ray study of the nature of stacking faults in the structure of glauconites
-
- Journal:
- Clay Minerals / Volume 25 / Issue 4 / December 1990
- Published online by Cambridge University Press:
- 09 July 2018, pp. 419-435
-
- Article
- Export citation
Occurrence of a ferrous, trioctahedral smectite in Recent sediments of Atlantis II Deep, Red Sea
-
- Journal:
- Clay Minerals / Volume 20 / Issue 3 / September 1985
- Published online by Cambridge University Press:
- 09 July 2018, pp. 389-404
-
- Article
- Export citation


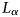
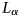
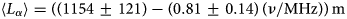
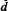 . Ces fluctuations sont interprétées comme provenant de l'hétérogénéité de la distribution des charges dans le feuillet. Enfin, un processus théorique d'hydratation et de déshydratation est proposé.
. Ces fluctuations sont interprétées comme provenant de l'hétérogénéité de la distribution des charges dans le feuillet. Enfin, un processus théorique d'hydratation et de déshydratation est proposé.