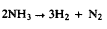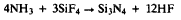Symposium R – Chemical Vapor Deposition of Refractory Metals and Ceramics II
Research Article
Therirmochemistry in C.V.D. - on the Choice of Haliide Gas Species
-
- Published online by Cambridge University Press:
- 15 February 2011, 3
-
- Article
- Export citation
Chemical Vapor Deposition Modeling for High Temperature Materials
-
- Published online by Cambridge University Press:
- 15 February 2011, 17
-
- Article
- Export citation
Theoretical Study of Gas-Phase Thermodynamics Relevant to Silicon Carbide Chemical Vapor Deiposition
-
- Published online by Cambridge University Press:
- 15 February 2011, 29
-
- Article
- Export citation
Kinetic Modelling of the Deposition of SiC from Methyltrichlorosilane
-
- Published online by Cambridge University Press:
- 15 February 2011, 35
-
- Article
- Export citation
Kinetics of Chemical Vapor Deposition of Sic Between 750 and 850°C at 1 Bar Total Pressure
-
- Published online by Cambridge University Press:
- 15 February 2011, 41
-
- Article
- Export citation
Kinetic Characteristics of Si3N4 CVD
-
- Published online by Cambridge University Press:
- 15 February 2011, 47
-
- Article
- Export citation
Onset Conditions for Gas Phase Reaction and Nucleation in the CVD of Transition Metal Oxides
-
- Published online by Cambridge University Press:
- 15 February 2011, 53
-
- Article
- Export citation
Rapid Growth of Ceramic Films by Particle-Vapor Codeposition
-
- Published online by Cambridge University Press:
- 15 February 2011, 59
-
- Article
- Export citation
Step Coverage Modeling of Thin Films Deposited by CVD Using Finite Element Method
-
- Published online by Cambridge University Press:
- 15 February 2011, 65
-
- Article
- Export citation
Pyrolytic Carbon Deposition on Graphitic Surfaces
-
- Published online by Cambridge University Press:
- 15 February 2011, 71
-
- Article
- Export citation
Surface Chemistry of Fluorine-Containing Molecules Related to CVD Process on Silicon Nitride: SiF4, XeF2, and HF
-
- Published online by Cambridge University Press:
- 15 February 2011, 79
-
- Article
- Export citation
Determination of the Roughness of CVD Surfaces by Laser Scattering
-
- Published online by Cambridge University Press:
- 15 February 2011, 85
-
- Article
- Export citation
In-Situ Light-Scattering Measurements During the CVD of Polycrystalline Silicon Carbide
-
- Published online by Cambridge University Press:
- 15 February 2011, 93
-
- Article
- Export citation
Interactive use of Electron Microscopy and Light Scattering as Diagnostics for Pyrogenic Aggregates
-
- Published online by Cambridge University Press:
- 15 February 2011, 101
-
- Article
- Export citation
Tunable Diode Laser Absorption Spectroscopy of the Pyrolysis of Methylsilazane
-
- Published online by Cambridge University Press:
- 15 February 2011, 107
-
- Article
- Export citation
Axial Concentration Profile of H2 Produced in the CVD of Si3N4.
-
- Published online by Cambridge University Press:
- 15 February 2011, 113
-
- Article
- Export citation
Laser Induced Fluorescence for Temperature Measurement in Reacting Flows
-
- Published online by Cambridge University Press:
- 15 February 2011, 119
-
- Article
- Export citation
In-Situ Optical Emission Spectra of Ti, TiN and TiSi2 Plasma During Thin Film Growth by Pulsed Laser Evaporation
-
- Published online by Cambridge University Press:
- 15 February 2011, 125
-
- Article
- Export citation
Molecular Precursors to Boron Nitride then Films: the Reactions of Diborane with Ammonia and with Hydrazine on Ru(0001)
-
- Published online by Cambridge University Press:
- 15 February 2011, 131
-
- Article
- Export citation
Near-Room Temperature Deposition of W and WO3 Thin Films by Hydrogen Atom Assisted Chemical Vapor Deposition
-
- Published online by Cambridge University Press:
- 15 February 2011, 137
-
- Article
- Export citation

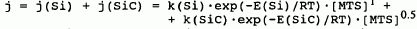 with E(Si) = 160 and E(SiC) = 300 kJ/mol. Reaction mechanisms are presented to account for the observed reaction orders with respect to MTS. Between 900 and 970 °C, the reaction rate decreased with temperature indicating a change in the deposition mechanism.
with E(Si) = 160 and E(SiC) = 300 kJ/mol. Reaction mechanisms are presented to account for the observed reaction orders with respect to MTS. Between 900 and 970 °C, the reaction rate decreased with temperature indicating a change in the deposition mechanism.