12 results
Uranium and technetium interactions with wüstite [Fe1–xO] and portlandite [Ca(OH)2] surfaces under geological disposal facility conditions
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 5 / October 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1097-1113
-
- Article
- Export citation
Notes on Contributors
-
-
- Book:
- Ralph Waldo Emerson in Context
- Published online:
- 05 December 2013
- Print publication:
- 09 December 2013, pp xi-xviii
-
- Chapter
- Export citation
Unstable Richtmyer–Meshkov growth of solid and liquid metals in vacuum
-
- Journal:
- Journal of Fluid Mechanics / Volume 703 / 25 July 2012
- Published online by Cambridge University Press:
- 13 June 2012, pp. 60-84
-
- Article
- Export citation
Contributors
-
-
- Book:
- Core Topics in Cardiac Anesthesia
- Published online:
- 05 April 2012
- Print publication:
- 15 March 2012, pp x-xiii
-
- Chapter
- Export citation
Contributors
-
-
- Book:
- Behavioral and Psychopharmacologic Pain Management
- Published online:
- 10 January 2011
- Print publication:
- 25 November 2010, pp ix-xii
-
- Chapter
- Export citation
Gene expression profiles of pork samples divergent in intramuscular fat content
-
- Journal:
- Advances in Animal Biosciences / Volume 1 / Issue 1 / April 2010
- Published online by Cambridge University Press:
- 29 November 2010, p. 235
- Print publication:
- April 2010
-
- Article
-
- You have access
- Export citation
3 - Patterns
-
-
- Book:
- A Gallery of Fluid Motion
- Published online:
- 25 January 2010
- Print publication:
- 12 January 2004, pp 28-41
-
- Chapter
- Export citation
Looking Backward, Looking Forward: MLA Members Speak
-
- Journal:
- PMLA / Publications of the Modern Language Association of America / Volume 115 / Issue 7 / December 2000
- Published online by Cambridge University Press:
- 23 October 2020, pp. 1986-2078
- Print publication:
- December 2000
-
- Article
- Export citation
Acoustic neuroma (schwannoma) surgery 1978–1990
-
- Journal:
- The Journal of Laryngology & Otology / Volume 105 / Issue 3 / March 1991
- Published online by Cambridge University Press:
- 29 June 2007, pp. 165-173
- Print publication:
- March 1991
-
- Article
- Export citation
A battered baby with pharyngeal atresia
-
- Journal:
- The Journal of Laryngology & Otology / Volume 85 / Issue 7 / July 1971
- Published online by Cambridge University Press:
- 29 June 2007, pp. 729-732
- Print publication:
- July 1971
-
- Article
- Export citation
Neurilemmoma of the larynx
-
- Journal:
- The Journal of Laryngology & Otology / Volume 84 / Issue 7 / July 1970
- Published online by Cambridge University Press:
- 29 June 2007, pp. 747-750
- Print publication:
- July 1970
-
- Article
- Export citation
Deafness following acute carbon monoxide poisoning
-
- Journal:
- The Journal of Laryngology & Otology / Volume 83 / Issue 12 / December 1969
- Published online by Cambridge University Press:
- 29 June 2007, pp. 1219-1225
- Print publication:
- December 1969
-
- Article
- Export citation

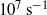 ) and high strains (700 %) as the fundamental link to the onset of ejecta formation. Our experimental observations allow us to approximate the strength of Cu at high strains and strain rates, revealing a unique diagnostic method for use at these extreme conditions.
) and high strains (700 %) as the fundamental link to the onset of ejecta formation. Our experimental observations allow us to approximate the strength of Cu at high strains and strain rates, revealing a unique diagnostic method for use at these extreme conditions.