20 results
Impact of an electronic medical-record–embedded clinical-decision support tool on duration of antibiotics for outpatient pediatric skin and soft-tissue infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 3 / March 2024
- Published online by Cambridge University Press:
- 03 November 2023, pp. 395-397
- Print publication:
- March 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
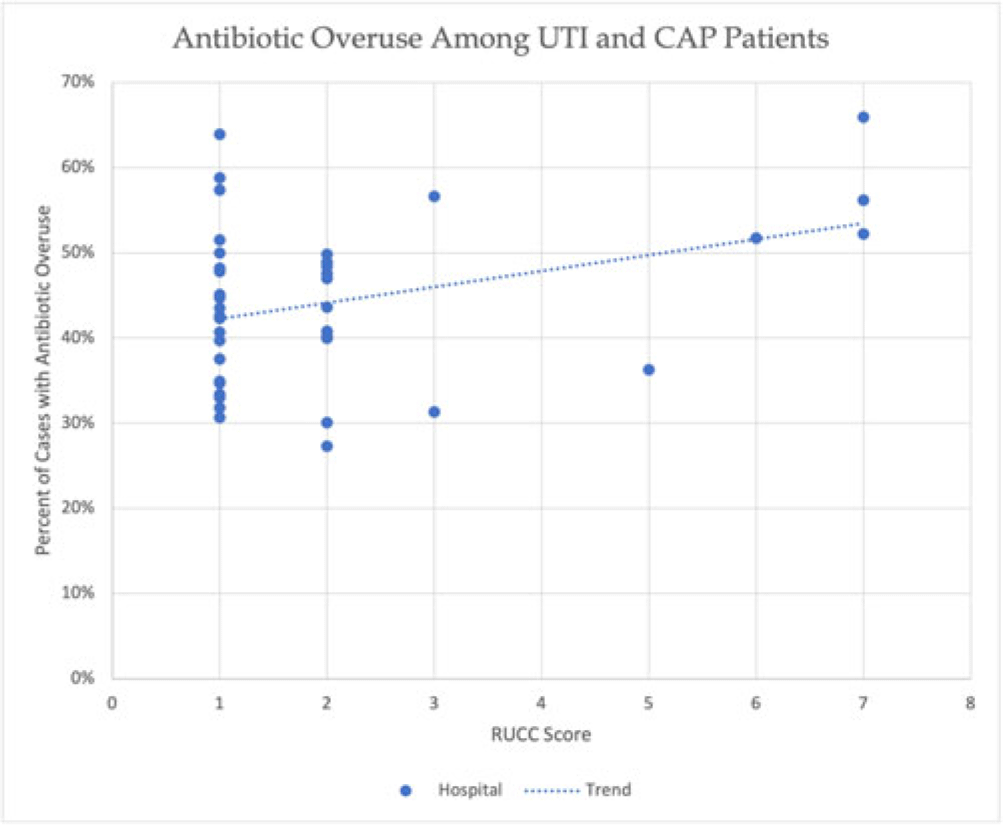
Identifying the relationship between hospital rurality and antibiotic overuse
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s34-s35
-
- Article
-
- You have access
- Open access
- Export citation
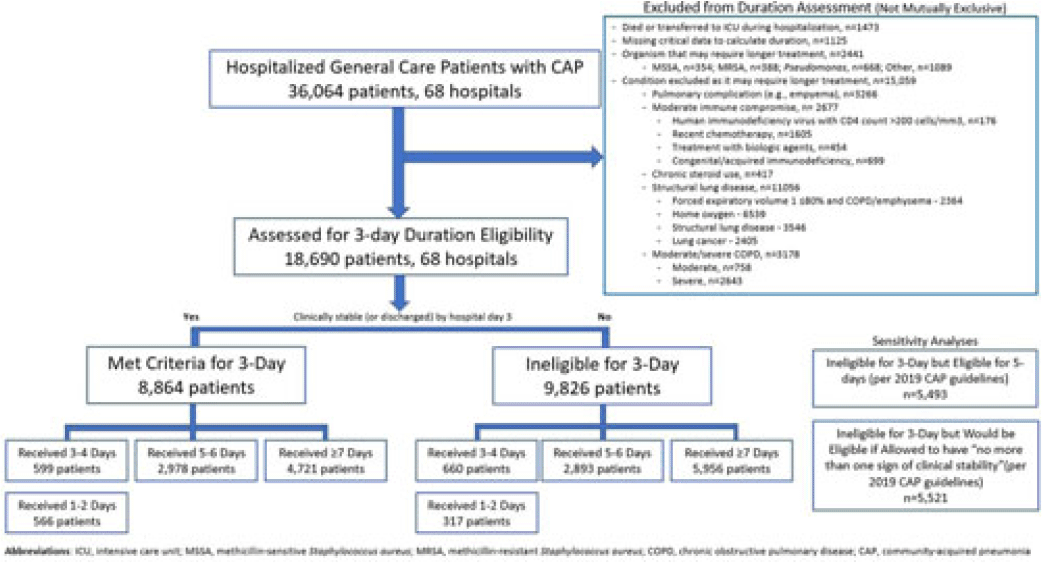
Three-day antibiotic duration in patients with pneumonia: A sixty-eight–hospital cohort
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s22
-
- Article
-
- You have access
- Open access
- Export citation
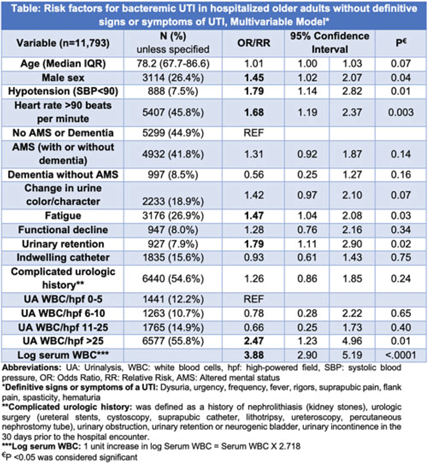
Prevalence of and risk factors for bacteremic UTIs in hospitalized adults without definitive signs or symptoms of UTI
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s27-s28
-
- Article
-
- You have access
- Open access
- Export citation
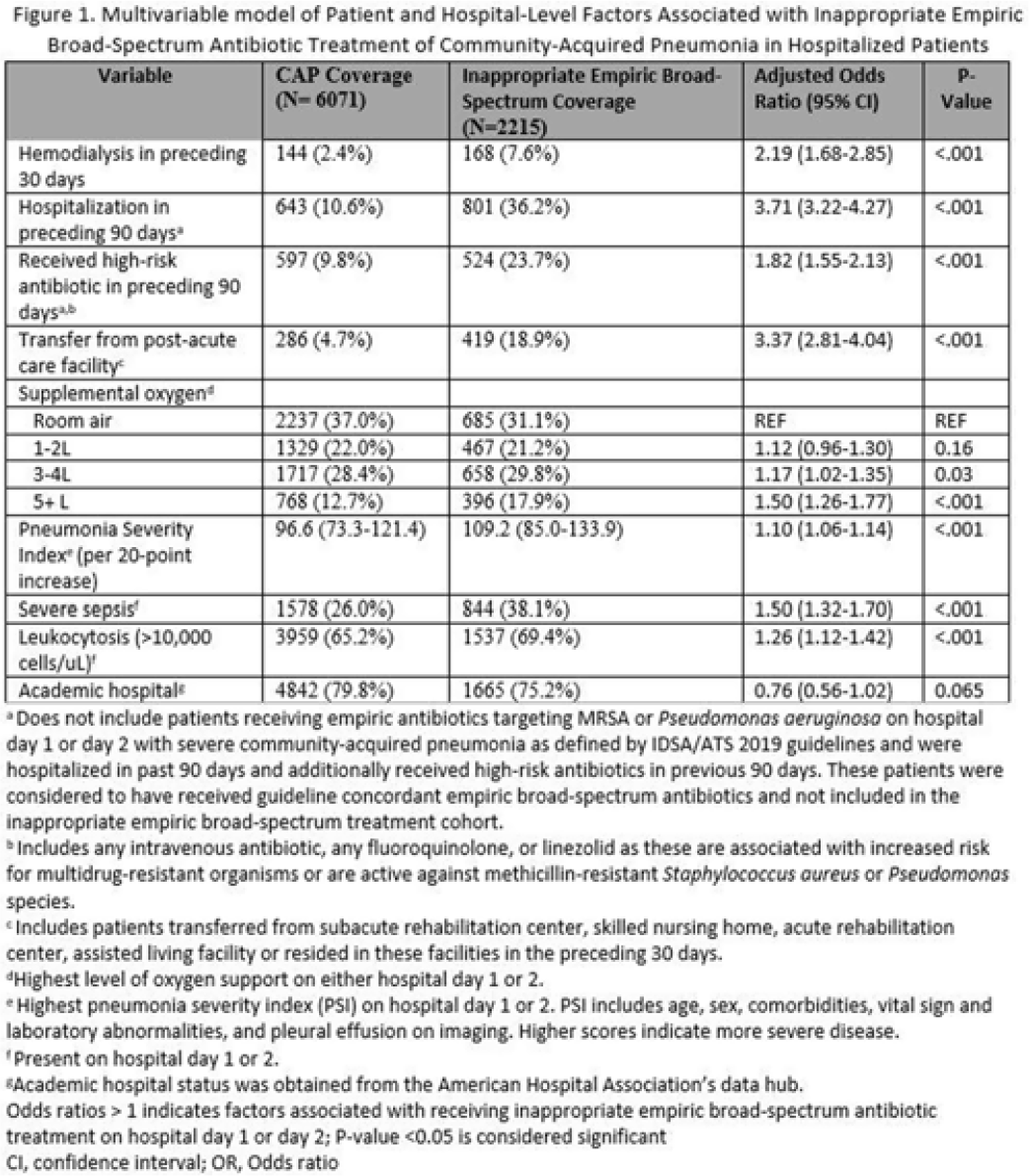
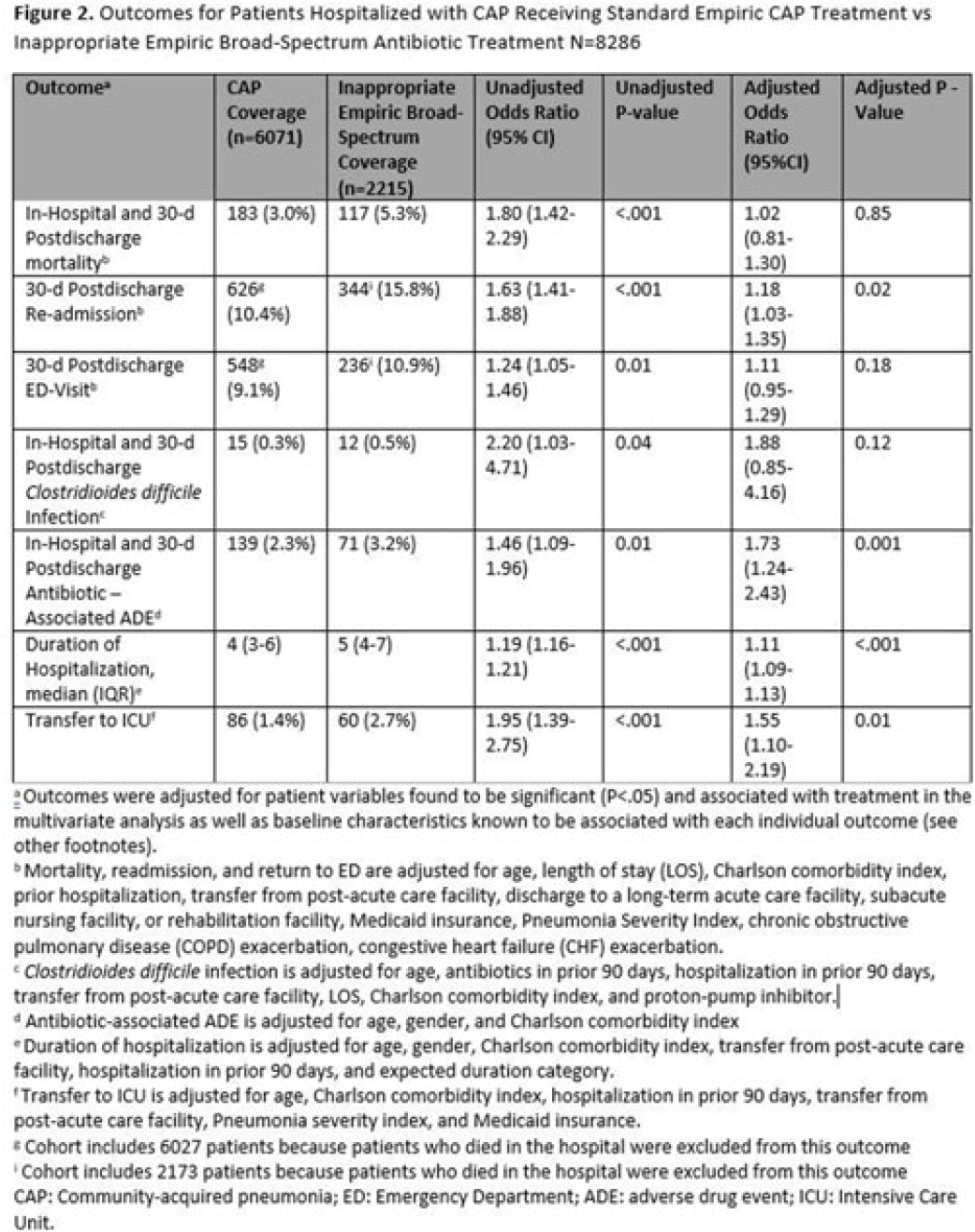
Risk Factors and outcomes associated with inappropriate empiric broad-spectrum antibiotic use in hospitalized patients with community-acquired pneumonia
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s31-s32
-
- Article
-
- You have access
- Open access
- Export citation
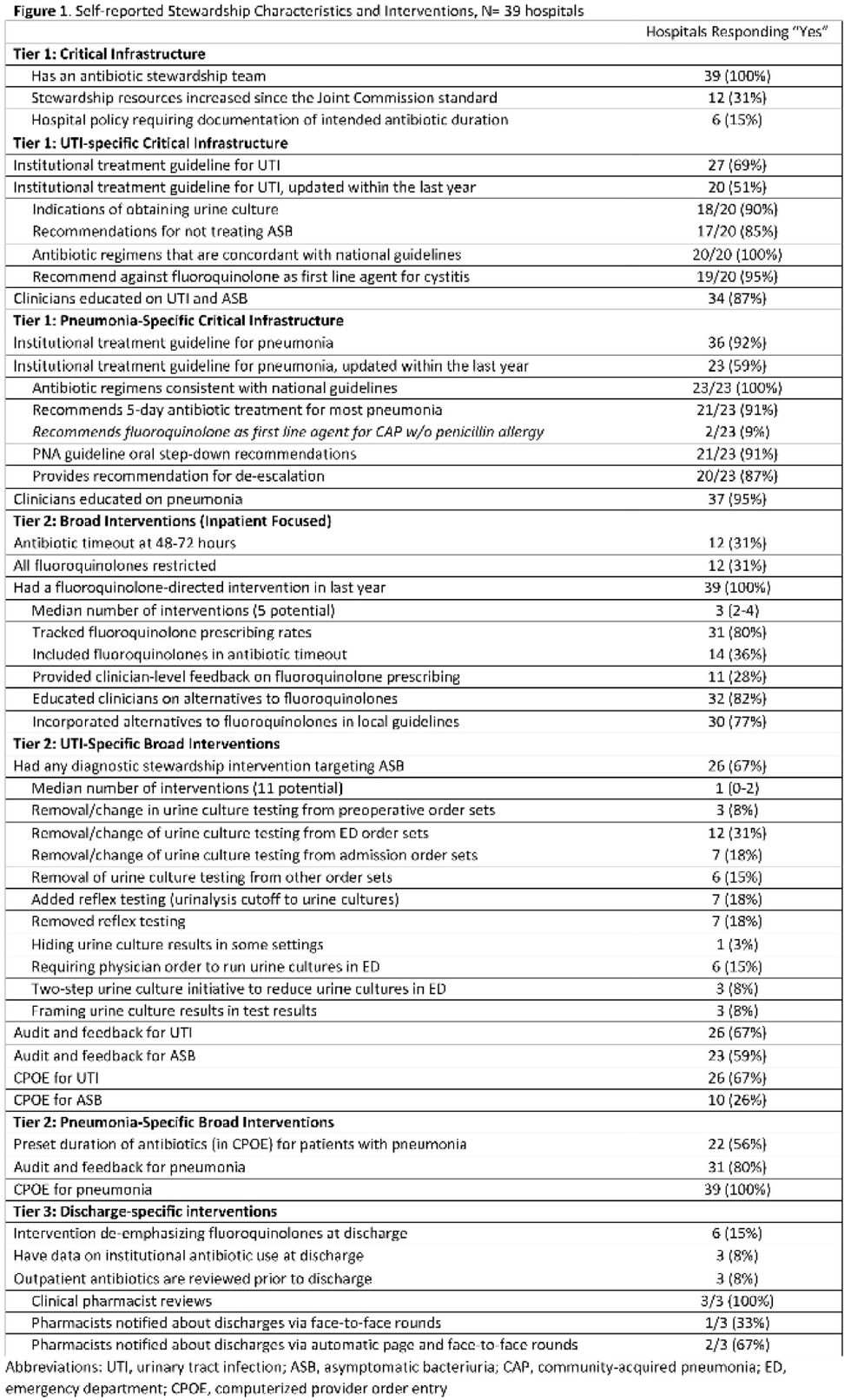
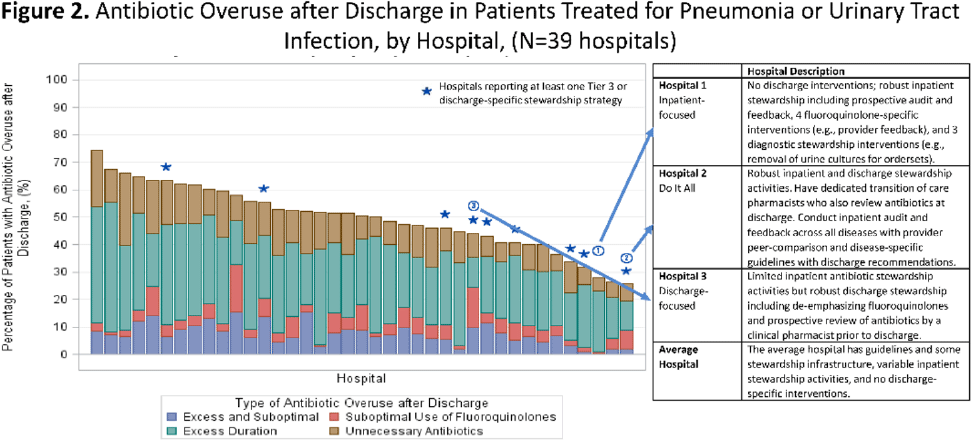
Antibiotic stewardship strategies and antibiotic overuse after hospital discharge: Analysis of the ROAD Home Framework
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s16-s17
-
- Article
-
- You have access
- Open access
- Export citation
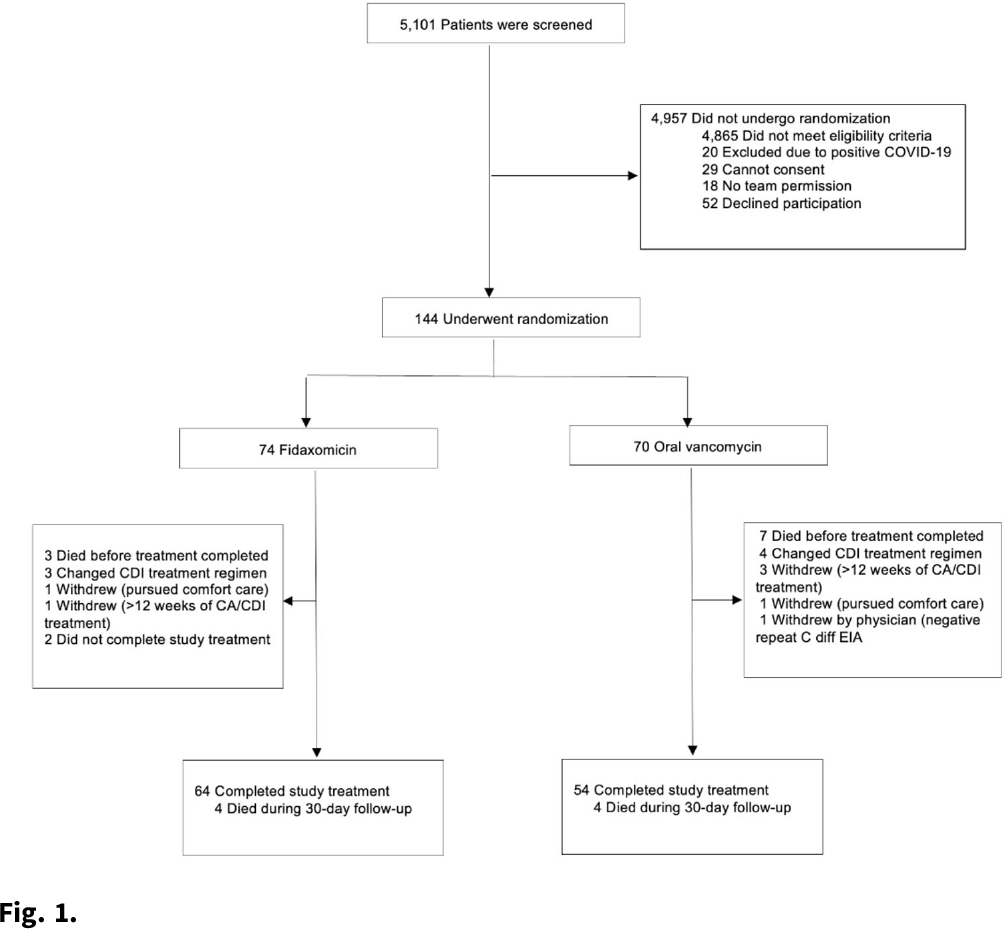
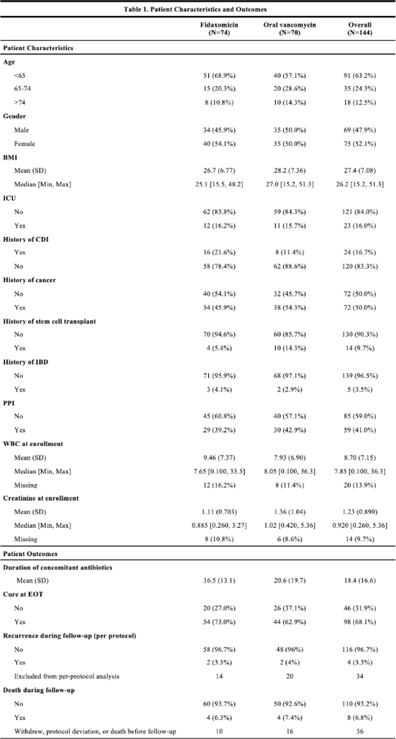
Comparison of fidaxomicin to oral vancomycin for the treatment of Clostridioides difficile infection in hospitalized patients
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s72-s73
-
- Article
-
- You have access
- Open access
- Export citation
Improving antibiotic use for sinusitis and upper respiratory tract infections: A virtual-visit antibiotic stewardship initiative
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 12 / December 2022
- Published online by Cambridge University Press:
- 31 January 2022, pp. 1890-1893
- Print publication:
- December 2022
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Antibiotic overuse after discharge from medical short-stay units
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 11 / November 2022
- Published online by Cambridge University Press:
- 24 August 2021, pp. 1689-1692
- Print publication:
- November 2022
-
- Article
- Export citation
Risk factors and outcomes associated with community-onset and hospital-acquired coinfection in patients hospitalized for coronavirus disease 2019 (COVID-19): A multihospital cohort study
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 9 / September 2022
- Published online by Cambridge University Press:
- 26 July 2021, pp. 1184-1193
- Print publication:
- September 2022
-
- Article
- Export citation
Asymptomatic bacterisputia: Rethinking diagnostic stewardship in pneumonia
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 6 / June 2021
- Published online by Cambridge University Press:
- 03 May 2021, pp. 737-739
- Print publication:
- June 2021
-
- Article
- Export citation
Antimicrobial never events: Objective application of a framework to assess vancomycin appropriateness
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 9 / September 2021
- Published online by Cambridge University Press:
- 29 December 2020, pp. 1121-1123
- Print publication:
- September 2021
-
- Article
- Export citation
Antibiotic Overuse at Discharge in Hospitalized Patients with Bacteriuria or Treated for Pneumonia: A Multihospital Study
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s459-s461
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Influence of Infectious Disease Physician Approval on Appropriateness of PICC Use and Outcomes
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s485-s487
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Misdiagnosis of Urinary Tract Infection Linked to Misdiagnosis of Pneumonia: A Multihospital Cohort Study
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s488-s489
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Incorporating preauthorization into antimicrobial stewardship pharmacist workflow reduces Clostridioides difficile and gastrointestinal panel testing
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue 10 / October 2020
- Published online by Cambridge University Press:
- 03 June 2020, pp. 1136-1141
- Print publication:
- October 2020
-
- Article
- Export citation
It is time to define antimicrobial never events
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 2 / February 2019
- Published online by Cambridge University Press:
- 05 December 2018, pp. 206-207
- Print publication:
- February 2019
-
- Article
- Export citation
Evaluating the Need for Antibiotic Stewardship Prospective Audit and Feedback on Weekends
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 38 / Issue 10 / October 2017
- Published online by Cambridge University Press:
- 22 August 2017, pp. 1262-1263
- Print publication:
- October 2017
-
- Article
-
- You have access
- HTML
- Export citation
Strategies to Prevent Catheter-Associated Urinary Tract Infections in Acute Care Hospitals: 2014 Update
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 35 / Issue S2 / September 2014
- Published online by Cambridge University Press:
- 10 May 2016, pp. S32-S47
- Print publication:
- September 2014
-
- Article
-
- You have access
- Export citation
Strategies to Prevent Catheter-Associated Urinary Tract Infections in Acute Care Hospitals: 2014 Update
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 35 / Issue 5 / May 2014
- Published online by Cambridge University Press:
- 10 May 2016, pp. 464-479
- Print publication:
- May 2014
-
- Article
- Export citation