Demand for ruminant products such as meat and milk is predicted to double by 2050( 1 ) and as such ensuring future meat and milk security in terms of availability, affordability and nutritional safety is at the forefront of agricultural research( 2 ). The WHO recommends that SFA intake should not exceed 0·35 of total fatty acid intake, the PUFA:SFA ratio should be about 0·4 and the n-6:n-3 PUFA ratio should be less than 4 in order to reduce the incidence of CVD( 3 ).
Ruminant products are often criticised for their SFA levels and PUFA:SFA ratio ( < 0·4), although they are noted for a beneficial n-6:n-3 PUFA ratio ( < 4)( Reference Scollan, Hocquette and Nuernberg 4 ). The low PUFA:SFA ratio is due to the fact that rumen microbiota biohydrogenate dietary PUFA to SFA, producing conjugated diene and triene intermediates( Reference Węsowska, Maia and Niedźwiedzka 5 – Reference Lourenço, Ramos-Morales and Wallace 7 ). Some intermediates of biohydrogenation such as conjugated linoleic acid (CLA; cis-9, trans-11-CLA) and trans-11-18 : 1 have been implicated to be important in human health( Reference Lock and Bauman 8 ). Current evidence suggests that many as yet uncultured rumen bacteria belonging to the families: Prevotella, Lachnospiraceae incertae sedis, and unclassified Bacteroidales, Clostridiales and Ruminococcaceae have biohydrogenating capacity( Reference Boeckaert, Vlaeminck and Fievez 9 – Reference Huws, Kim and Lee 12 ). Thus, manipulation of rumen bacteria in order to improve the fatty acid quality of meat and milk is exceptionally difficult given the complexity and inability to currently culture the bacterial taxa involved in biohydrogenation.
Irrespective of their potential biohydrogenation capabilities, microbial cells that flow to the duodenum from the rumen are an important source of fatty acids for absorption by the animal. In terms of the rumen bacteria, previous reports show that they are proportionally high in odd-chain and branched-chain SFA( Reference Williams and Dinusson 13 – Reference Or-Rashid, Odongo and McBride 15 ). There is some evidence that the rumen bacteria incorporate linoleic acid (18 : 2n-6)( Reference Hawke 16 ), but it is uncommon to find PUFA within the bacteria( Reference Goldfine 17 ). In contrast, rumen protozoa are rich in beneficial PUFA, MUFA and CLA compared with rumen bacteria( Reference Emmanuel 18 , Reference Devillard, McIntosh and Newbold 19 ). This is probably due to their ability to engulf chloroplasts, which contain most of total plant 18 : 3n-3 within their thylakoid membranes( Reference Hall, West and Coleman 20 – Reference Huws, Kim and Kingston-Smith 22 ). Indeed, approximately 60 % of the total chloroplast fatty acids are attributable to 18 : 3n-3( Reference Siebertz, Heinz and Linscheid 23 , Reference Sandelius and Aronsson 24 ). Thus, enriching the chloroplast content of protozoa offers a novel strategy of ensuring increased PUFA flow to the duodenum. It is also possible that co-localisation of chloroplasts with engulfed bacteria within food vacuoles can result in intra-protozoal lipolysis and biohydrogenation of the intra-protozoal chloroplasts, providing that co-localised bacteria have lipolytic and biohydrogenating capacities. This may contribute to the previously reported high proportional representation of CLA in rumen protozoa( Reference Or-Rashid, Odongo and McBride 15 , Reference Devillard, McIntosh and Newbold 19 ). Intra-protozoal chloroplast lipid metabolism may also aid direct uptake of the main chloroplast fatty acids (16 : 0, 18 : 3n-3 and 18 : 2n-6) into the rumen protozoal membranes. As such, our aims in the present experiment were to evaluate the effects of increasing the chloroplast content of rumen protozoa on the duodenal flow of fatty acids shown to be beneficial for human health.
Experimental methods
Animals, diets and experimental design
The experiment was conducted under the authorities of the UK Animals (Scientific Procedures) Act (1986). A total of six Hereford × Friesian steers (Bos p. taurus) prepared with rumen and duodenal cannulae were employed and the experiment consisted of a two-period changeover design with two diets: fresh perennial ryegrass (PRG, Lolium perenne) cultivar AberElan, 3 weeks into a secondary re-growth (high chloroplast; PRG) and barley (Hordeum vulgare) straw:concentrate (S:C; 40:60 on a DM basis; Table 1; low chloroplast diet; S:C). Each diet was offered for a 12 d adaptation period before sample collection. Steers were allowed to eat ad libitum at 09.00 and 16.00 hours with refusals collected at 08.45 and 13.45 hours and DM intake determined. The building was well ventilated, with animals having free access to fresh water and minerals (Baby Red Rockies, Tithebarn Limited; composed of: 380 g/kg Na, 5000 mg/kg Mg, 1500 mg/kg Fe, 300 mg/kg Cu, 300 mg/kg Zn, 200 mg/kg Mn, 150 mg/kg I, 50 mg/kg Co and 10 mg/kg Se).
Table 1 Feed formulation for straw:concentrate mixture (40:60 on a DM basis)*
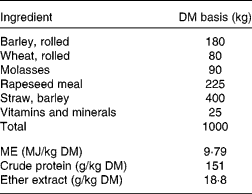
ME, metabolisable energy.
* Concentrate was formulated and purchased from Wynnstay Group Plc.
Digesta flow at the duodenum was estimated using a dual-phase marker technique with ytterbium acetate and Cr EDTA as the particulate and liquid phase markers, respectively( Reference Faichney 25 ). Ytterbium acetate (375 mg Yb/d) and Cr EDTA (2401 mg Cr/d) were infused via separate lines intra-ruminally at a rate of 28 ml/h commencing on day 8. On days 13 and 16, 400 ml of duodenal digesta were collected manually every 3 h over a 24 h period, bulked, stored at 4°C and processed as described by Kim et al. ( Reference Kim, Huws and Lee 10 ).
Sample preparation and chemical analysis
Feed was sampled once daily for straw and twice daily at feed times for PRG. Straw samples for the first 8 d and the latter 8 d were bulked, as chemical composition is unlikely to change across days, whereas due to the greater risk of chemical composition fluctuations between days( Reference Turner, Cairns and Armstead 26 ), daily PRG samples were kept separate. Samples were frozen, freeze-dried, ground and stored at − 20°C for later chemical analysis. Water-soluble carbohydrate (WSC) concentration of forages was determined spectrophotometrically using anthrone in sulphuric acid on a Technicon Autoanalyser (Technicon Corporation)( Reference Thomas 27 ). Neutral-detergent fibre was determined as described by Van Soest et al. ( Reference Van Soest, Robertson and Lewis 28 ), without the use of amylase or sodium sulphite for forages and using sodium sulphite and termamyl in place of amylase for concentrates, while using the Tecator Fibretec System (Tecator Limited). Acid-detergent fibre was analysed according to the method of Van Soest & Wine( Reference Van Soest and Wine 29 ) using the Tecator Fibretec System (Tecator Limited). The fatty acid concentration of the diets was measured using a one-step extraction–transesterification procedure( Reference Sukhija and Palmquist 30 ). N concentration of diets and microbial fractions was analysed by combustion at 550°C using a LECO FP-428 analyser (LECO Corporation). Oil content was measured by diethyl ether extract( Reference Lee, Harris and Moorby 31 ).
Collection of rumen samples took place 2 h after feeding on day 14 on both consecutive diets as described( Reference Huws, Kim and Kingston-Smith 22 ). Bacterial contamination of fractionated protozoa was assessed using total eubacterial quantitative PCR as described in the following sections. Chlorophyll concentration was measured as an indicator of plant contamination extracellular to the protozoa alongside fluorescent microscopy( Reference Arnon 32 ). Fatty acid composition of the microbial fractions and digesta was determined using a one-step extraction bimethylation approach as described by Kim et al. ( Reference Kim, Huws and Lee 10 ).
DNA extraction from rumen protozoal and whole duodenal samples
Genomic DNA was extracted from rumen protozoal and whole duodenal samples (10 mg DM) using the BIO101 FastDNA® SPIN Kit for Soil (Qbiogene, Inc.) in conjunction with a FastPrep® cell disrupter instrument (Bio101, ThermoSavant, Qbiogene, Inc.) according to the manufacturer's instructions, with the exception that the samples were processed for 3 × 30 s at speed 6·0 in the FastPrep instrument. DNA was quantified and the quality assured using the ND1000 nanodrop system (Labtech International Limited).
Total bacterial quantitative PCR
In order to assess bacterial contamination in the protozoal fractions, quantitative PCR of bacterial 16S rDNA was performed as described( Reference Edwards, Huws and Kim 33 , Reference Edwards, Huws and Kim 34 ).
Protozoal 18S rRNA-based PCR-denaturing gradient gel electrophoresis
In order to validate that rumen and duodenal protozoal diversity was similar under both diets, PCR-denaturing gradient gel electrophoresis (DGGE) was used as described( Reference Huws, Kim and Kingston-Smith 22 ).
Total protozoal quantitative PCR
Total protozoal 18S rDNA amplification was carried out on ruminal digesta samples in a final volume of 25 μl containing 12·5 μl SYBR® Green JumpStart™ Taq ReadyMix™ (Sigma-Aldrich), 250 nm each of 316f 5′-GCTTTCGWTGGTAGTGTATT-3′ and 539r 5′-ACTTGCCCTCAAATCGT-3′( Reference Huws, Kim and Kingston-Smith 22 ), and 2 μl of a 1:50 dilution of extracted genomic DNA. The thermal cycling programme was thirty cycles of 94°C for 30 s and 54°C for 30 s, with an initial cycle of 94°C for 5 min. After PCR, a dissociation curve (melting curve) was constructed in the range of 55–95°C. All samples were run in triplicate and quantitative PCR were conducted on two separate occasions for the validation of results. A rumen protozoal DNA sample obtained from this study and determined to be low in plant and bacterial contamination was used as a protozoal standard.
Microscopy
Total protozoal densities as well as proportions of holotrich and entodinomorphid protozoa were assessed using a Sedgewick rafter counting chamber and an Olympus BH-2 microscope (Olympus UK Limited). Proportional representation of the main entodinomorphid protozoa was also recorded. Total protozoa, as well as proportions of holotrichs and entodinomorphid protozoa containing intracellular auto-fluorescing chloroplasts, were assessed using an Olympus BH-2 fluorescent microscope by scanning 100 protozoa in each of the samples. Percentage of total protozoa as well as proportions of holotrichs and entodinomorphid protozoa saturated (>10/cell) with intracellular chloroplast was also recorded.
Calculations and statistical analysis
Digesta flows were estimated after mathematical reconstitution of true digesta as described by Faichney( Reference Faichney 25 ). The N:total DNA ratios and the N:individual fatty acid ratios were recorded for rumen protozoal samples, and the duodenal flows of protozoal DNA, N and individual fatty acids were calculated as described( Reference Skillman, Toovey and Williams 35 – Reference Yáñez-Ruiz, Scollan and Merry 38 ). Data for protozoal density, chloroplast, N, DNA, fatty acid content, intakes and flows of DM, fatty acids following feeding with either S:C or PRG were subjected to ANOVA using GenStat 13th Edition (VSNi)( Reference Payne, Murray and Harding 39 ).
Results
Chemical composition of the experimental diets
The S:C diet was lower in WSC and oil, but was similar in N, neutral- detergent fibre and acid-detergent fibre concentration compared with the fresh PRG diet (Table 2). Fatty acid composition of the S:C diet was: 7·9 % 18 : 3n-3, 14·4 % 16 : 0 and 35·5 % 18 : 2n-6. The DM, total N, WSC, acid-detergent fibre and neutral-detergent fibre of the PRG were comparable to that stated by Lee et al. ( Reference Lee, Harris and Moorby 31 ) and Huws et al. ( Reference Huws, Kim and Kingston-Smith 22 ) for the same PRG cultivar. Fatty acid composition of the PRG was 63·3 % 18 : 3n-3, 15·1 % 16 : 0 and 14·5 % 18 : 2n-6.
Table 2 Chemical composition (g/kg DM, unless stated) and fatty acid profile of the experimental diets (Mean values, n 4 for both diets)
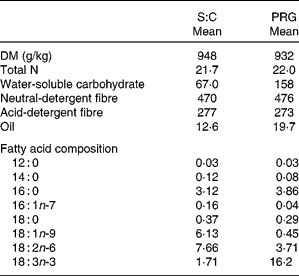
S:C, straw:concentrate; PRG, perennial ryegrass.
Rumen and duodenal protozoal diversity
DGGE comparisons of rumen v. duodenal protozoal diversity on each diet separately showed that rumen and duodenal protozoal diversity was similar following S:C and PRG feeding (Fig. 1). Fig. 1 illustrates the data for steer no. 1 post-PRG and S:C feeding only but these data are representative of similarities found within the other steers.

Fig. 1 Denaturing gradient gel electrophoresis profiles comparing rumen and duodenal protozoal 18S rDNA diversity for steer 1 following (a) straw:concentrate (60:40; DM basis) and (b) fresh perennial ryegrass feeding of steers.
Bacterial and plant contamination levels of rumen protozoal samples
Bacterial 16S rDNA:protozoal 18S rDNA density in the rumen protozoal samples was on average 0·18:1. Microscopy and extra-protozoal chlorophyll quantification revealed that plant contamination was comparatively minimal within these samples (average protozoal extracellular chlorophyll quantity was 6·2 mg/g protozoal DM following S:C feeding compared with a 30·8 mg/g following PRG feeding, while chlorophyll intake was 48·2 g/d following S:C feeding and 290·1 g/d following PRG feeding, on a DM basis).
Daily nutrient intake and duodenal flows
DM intake averaged 8·80 and 9·42 kg/d following feeding of S:C and PRG, respectively (Table 3). DM, N, neutral-detergent fibre and acid-detergent fibre intake did not differ between diets, whereas WSC intake was higher when steers were offered PRG compared with when offered S:C (Table 3). Intake of 16 : 0 and 18 : 3n-3 was greater following PRG feeding compared with S:C feeding (Table 3). Conversely, 16 : 1n-7, 18 : 1n-9 and 18 : 2n-6 intake were greater following feeding of S:C compared with PRG (Table 3). Duodenal flow of DM and organic matter were comparable post-feeding on both experimental diets (Table 3). Total N flowing to the duodenum was nonetheless higher post-feeding on PRG compared with the S:C diet, although the difference in total N flow did not reach significance at P < 0·05 (Table 3). Protozoal N flowing to the duodenum was nonetheless significantly higher post-feeding on S:C compared with the PRG diet (Table 3).
Table 3 Daily intakes and duodenal flows of DM, organic matter (OM) and nitrogen in steers, straw:concentrate (S:C) or fresh perennial ryegrass (PRG; g/d, unless stated)(Mean values and standard errors of the difference)

* Values were not significantly different (P>0·10).
Ruminal protozoal chloroplast, fatty acid, DNA and nitrogen content
Protozoal density post-feeding of PRG was lower compared with post-feeding with S:C (Table 4). All microscopically visualised protozoa were entodinomorpids, with undetectable densities of holotrichs present on both experimental diets (Table 4). The entodinomorphid protozoal genera present post-feeding of both diets was similar (Table 4). Although post-PRG feeding the percentage of protozoa containing intracellular chloroplasts was not different to the percentage of protozoa containing intracellular chloroplasts post-S:C feeding, 5 % of the population (mainly Polyplastron spp. and Diplodinium spp.) were saturated (>10) with intracellular chloroplasts while none of the visualised protozoa had >10 intracellular chloroplasts post-S:C feeding (Table 4). This increase in the number of intracellular chloroplasts post-PRG feeding coincided with an increased protozoal concentration of 18 : 3n-3, expressed on a N basis (Table 4). Concentrations of the biohydrogenation intermediate trans-11-18 : 1 was also higher post-PRG feeding compared with levels post-S:C feeding (Table 4). Protozoal concentrations of 18 : 2n-6, cis-9, trans-11-CLA and trans-10, cis-12-CLA were conversely higher post-S:C feeding as opposed to PRG feeding (Table 4).
Table 4 Rumen protozoal density and protozoal chloroplast, nitrogen, DNA and fatty acid content following feeding with either straw:concentrate (S:C) or fresh perennial ryegrass (PRG) (Mean values and standard errors of the difference, n 4)

ND, not detected; NA, not applicable; CLA, conjugated linoleic acid.
* Mean values were not significantly different (P>0·10).
† sed values are in log scale.
Contribution of protozoa to duodenal fatty acid flow
Total duodenal flow of 14 : 0, 15 : 0, cis-9, trans-11-CLA and 18 : 2n-6 was lower post-PRG compared with S:C feeding (Table 5). Conversely, duodenal flow of 16 : 0, 17 : 0, 18 : 0, trans-11-18 : 1, 18 : 3n-3 and trans-10, cis-12-CLA was greater post-PRG compared with S:C feeding (Table 5). Following S:C feeding of steers, protozoal flow to the duodenum accounted for 80 % of total cis-9, trans-11, 26·2 % trans-11-18 : 1, 21·5 % 16 : 0, 12·9 % 18 : 2n-6 and 8·4 % 18 : 3n-3 (Table 5). Nonetheless, contribution of protozoa to the flow of all reported fatty acids post-PRG feeding was low due to the low duodenal ciliate 18S rDNA concentration within duodenal samples (protozoal 18S rDNA concentration in duodenum following S:C feeding was 11·7 μg/mg, compared with 0·3 μg/mg following PRG feeding; although rumen protozoal 18S rDNA concentration following S:C feeding was 117·2 μg/mg, compared with 87·8 μg/mg following PRG feeding).
Table 5 Total fatty acid and protozoal-related fatty acid duodenal flow in steers fed straw:concentrate (S:C) or fresh perennial ryegrass (PRG) (Mean values and standard errors of the difference)

CLA, conjugated linoleic acid.
* Contribution of protozoa to fatty acid duodenal flow (protozoal fatty acid flow/total fatty acid flow × 100 %).
Discussion
In the present study, we used developed methods to estimate the contribution that protozoa make to duodenal fatty acid flow as a consequence of their intracellular chloroplast content( Reference Skillman, Toovey and Williams 35 – Reference Yáñez-Ruiz, Scollan and Merry 38 ). These methods are based on having sufficient similarity in rumen and duodenal protozoal 18S rDNA diversity, such that rumen samples can then be used as standards to calculate protozoal 18S rDNA:N, N:protozoal individual fatty acid, allowing duodenal protozoal N and individual fatty acid duodenal flow to be calculated. This subsequently allows us to calculate the protozoal contribution to total fatty acid duodenal flow.
Rumen protozoal samples were relatively low in bacterial contamination (18 % of total DNA was bacterial in origin), and values were comparable to those reported previously( Reference Huws, Kim and Kingston-Smith 22 , Reference Yáñez-Ruiz, Scollan and Merry 38 , Reference Kingston-Smith and Theodorou 40 ). Microscopical visualisation of samples coupled with extra-protozoal chlorophyll quantification also confirmed that samples were low in plant contamination. Average N concentration of the protozoa was also comparable with previous data( Reference Huws, Kim and Kingston-Smith 22 , Reference Volden, Mydland and Harstad 41 , Reference Merry and McAllan 42 ). Fatty acid content of the rumen protozoa, on a N basis, was similar to those reported by Yáñez-Ruiz et al. ( Reference Yáñez-Ruiz, Scollan and Merry 38 ); however, both 18 : 2n-6 and 18 : 3n-3 protozoal content were lower, on a N basis, following feeding on both diets compared with the Yáñez-Ruiz et al. ( Reference Yáñez-Ruiz, Scollan and Merry 38 ) data. The reasoning for this discrepancy is unclear. When expressed as mg/g, our data are similar to the data we obtained in the previous study with respect to all intra-protozoal fatty acid concentrations reported. Specifically, protozoal 18 : 3n-3 content was similar between this study and our previous work (S:C average – 0·57 mg/g DM, PRG – 0·78 mg/g DM in this study compared with average values reported in Huws et al. ( Reference Huws, Kim and Kingston-Smith 22 ): hay/concentrate – 0·46 mg/g DM, fresh PRG – 1·36 mg/g DM). The slightly lower protozoal 18 : 3n-3 content post-PRG feeding may be due to the fact that the steers used in this study contained type A protozoa whereas the steers contained type B protozoa in our previous study( Reference Huws, Kim and Kingston-Smith 22 ). We previously noted that Epidinium spp. were commonly saturated with intracellular chloroplast post-PRG feeding, and were consequentially the most concentrated in 18 : 3n-3. In this study, type A protozoal populations do not harbour Epidinium spp., therefore potentially explaining the slightly lower 18 : 3n-3 content post-PRG, although it should be noted that some Polyplastron and Diplodinium spp. within this study did contain >10 intracellular chloroplasts. The fatty acid contents of protozoa post-S:C feeding of steers were also similar to those previously reported when a conserved forage was fed( Reference Or-Rashid, Odongo and McBride 15 , Reference Emmanuel 18 , Reference Devillard, McIntosh and Newbold 19 ). Irrespective of this, post-fresh PRG feeding rumen protozoa were enriched in: 17 : 0, trans-11-18 : 1, 18 : 3n-3 and lower in 18 : 2n-6, cis-9, trans-11-and trans-10, cis-12-CLA compared with concentrations post-S:C feeding. These differences in protozoal fatty acid content are similar to those reported in our previous study comparing hay/concentrate or fresh PRG( Reference Huws, Kim and Kingston-Smith 22 ). In this study, 18 : 3n-3 content of protozoa post-PRG feeding was 5·5 × higher than the levels post-S:C feeding. The difference in 18 : 3n-3 content of the protozoa was not a product of having differential numbers of genera known to engulf chloroplasts more effectively. Polyplastron spp. made up 2·8 and 4·8 % of the total protozoal population while Diplodinium spp. made up 10·8 and 14·2 % of the total protozoal population post-feeding of S:C and PRG, respectively. Taking into account the total protozoal numbers, Polyplastron spp. were present at 97·5 × 103 and 17·1 × 103 cells/ml following S:C and PRG-feeding, respectively. Likewise, taking into account the total protozoal numbers, Diplodinium spp. were present at 25·3 × 103 and 5·08 × 103 cells/ml following S:C and PRG feeding, respectively. Post-PRG feeding, 5 % of protozoa had >10 chloroplasts/cell but no protozoa had reached these saturation levels post-S:C feeding. Thus, this 18 : 3n-3 differential is likely to be due to differences in chloroplast concentration between the diets, intake and subsequent protozoal content. If food vacuoles containing chloroplasts also contained lipolytic bacteria, then this 18 : 3n-3 differential could also be due to direct uptake into membranes. Intake of 18 : 3n-3 by the steers was 10 × fold higher following PRG feeding compared with S:C feeding, associated with the higher chloroplast content of PRG. Studies have shown that a high proportion (>50 %) of plant cells are intact post-mastication( Reference Martin, Williams and Michalet-Doreau 43 ). Thus, mastication may have caused some lysis and release of 18 : 3n-3 from the chloroplast thylakoid membranes to the extracellular mileu, thereby reducing the available chloroplasts for protozoal ingestion and thus potentially explaining the slight difference in 18 : 3n-3 intake. Bioavailability of some chloroplasts may also be problematic, which may also mean that not all ingested chloroplasts can be engulfed by the rumen protozoa. It may also be the case that protozoal chloroplast intake was maximum irrespective of the availability of chloroplast for protozoal engulfment.
CLA concentration of ruminal protozoa fractionated post-PRG feeding was negligible compared with concentrations following S:C feeding. Conversely, concentrations of trans-11-18 : 1 were higher in ruminal protozoa fractionated from steers fed PRG compared with those from S:C-fed steers. Recent data suggest that the protozoa do not play a role in the saturation of PUFA or the desaturation of trans-11-18 : 1 to CLA( Reference Devillard, McIntosh and Newbold 19 ). As such, the reason for the intracellular CLA and trans-11-18 : 1 concentrations within these eukaryotes has been hypothesised to be due to preferential incorporation of these fatty acids into their cellular membranes, with their formation related to differences between diets( Reference Devillard, McIntosh and Newbold 19 ). It could also be the case that lipolytic and biohydrogenating bacteria may be co-localised with the intra-protozoal chloroplasts causing intra-protozoal formation of trans-11-18 : 1.
Using established methods( Reference Skillman, Toovey and Williams 35 – Reference Yáñez-Ruiz, Scollan and Merry 38 ), we have shown that post-S:C feeding of steers, protozoal N flowing to the duodenum is approximately 34·4 g/d. This value is higher than the estimates of 13–18 g/d obtained by some researchers( Reference Yáñez-Ruiz, Scollan and Merry 38 , Reference Punia, Leibholz and Faichney 44 – Reference Coleman 46 ), but close to the 38·1 g/d estimated by Sylvester et al. ( Reference Sylvester, Karnati and Yu 36 ) in the post-feeding of Holstein cows on a high-fibre diet. This difference is probably due to the differing diets between the studies. A high-concentrate diet is known to increase the protozoal N content( Reference Harrison, Beever and Osbourn 47 ), and thus is likely to lead to higher protozoal N duodenal flow. Protozoal N flowing to the duodenum post-PRG feeding of steers was substantially less at approximately 0·70 g/d. This value is closer to values of approximately 2·0 g/d obtained by Harrison et al. ( Reference Harrison, Beever and Osbourn 47 ) using other pre-molecular technologies and more recently those obtained by Belanche et al. ( Reference Belanche, Abecia and Holtrop 48 ) using molecular techniques as described within this study (0·15 and 0·5 g/d protozoal N flow following feeding of sheep alfalfa (Medicago sativa) hay and a mixed sward hay, respectively).
Post-S:C feeding, the contribution of protozoa to fatty acids flowing to the duodenum was broadly similar to the results of Yáñez-Ruiz et al. ( Reference Yáñez-Ruiz, Scollan and Merry 38 ). Trans-11-18 : 1 is important due to its conversion to cis-9, trans-11-18 : 2 in bovine mammary glands( Reference Griinari, Corl and Lacy 49 ). Thus, this study along with the previous study( Reference Yáñez-Ruiz, Scollan and Merry 38 ) highlights the potentially important contribution that protozoa make to the duodenal flow of key beneficial fatty acids, which are beneficial to human health, in animals on concentrate-based diets. Nonetheless, we were interested in testing the hypothesis that an improvement in 18 : 3n-3 flow to the duodenum could be made by increasing the chloroplast content of the diet, and thus the chloroplast content of the protozoa. Contribution of ruminal protozoa to the flow of all fatty acids was substantially lower post-PRG feeding compared with S:C feeding. Focusing on the contribution of protozoa post-PRG feeding to 18 : 3n-3 duodenal flow, this was 0·33 % compared with 8·43 % post-S:C feeding. This was due to the much lower protozoal 18S rDNA concentrations present within the duodenal samples post-PRG feeding of steers. This suggests that fresh grass feeding results in substantial ruminal retention of protozoa. DGGE profiles suggest that the retention was not selective, as the rumen and duodenal profiles were similar. There is evidence to show that some protozoa preferentially attach to plant material containing a high soluble sugar content( Reference Orpin and Letcher 50 ). Thus, the reason for this retention following fresh grass feeding is possibly due to the high WSC content of the PRG which provides the rumen protozoa with a favourable ecosystem potentially causing them to evade flow through to the abomasum; for example, associated with large food particles or inhabiting the bottom of the raft.
In conclusion, PRG feeding increases the intracellular chloroplast and coincidently the 18 : 3n-3 content of protozoa. This may be due directly to engulfment of the 18 : 3n-3-rich chloroplasts and/or due to intra-protozoal lipid metabolism and subsequent uptake of PUFA into the protozoa cell membranes. The fresh grass diet did, however, cause ruminal protozoal retention and therefore this enhanced protozoal n-3 PUFA content did not result in an increased flow to the duodenum. The challenge ahead is to capitalise on the benefits offered by chloroplast uptake by rumen protozoa in terms of capture of PUFA, while ensuring adequate protozoa flow and maintenance of rumen protozoal density.
Acknowledgements
The authors acknowledge funding from the Biotechnology and Biological Sciences Research Council (UK), Department of Environment Food and Rural Affairs, English Beef and Lamb Executive, Hybu Cig Cymru, Quality Meat Scotland and European Union Prosafebeef (FOOD-CT-2006-36241). The authors state that there are no conflicts of interest. The present experiment was instigated and designed by S. A. H. and N. D. S. Sample retrieval was accomplished by S. A. H., M. R. F. L., E. J. K. and M. B. S., while sample analysis was conducted by S. A. H., A. H. K.-S., M. B. S. and J. K. S. T. The authors are grateful to Animal and Microbial Science technical staff for their help with animal care.








