Volume 81 - Issue 5 - October 2017
Research Article
Leószilárdite, the first Na,Mg-containing uranyl carbonate from the Markey Mine, San Juan County, Utah, USA
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1039-1050
-
- Article
- Export citation
Crystal chemistry of zinc incorporation in strunzite-group minerals containing zeolitic water
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1051-1062
-
- Article
- Export citation
First crystal-structure determination of natural lansfordite, MgCO3·5H2O
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1063-1071
-
- Article
- Export citation
Dzierżanowskite, CaCu2S2 – a new natural thiocuprate from Jabel Harmun, Judean Desert, Palestine Autonomy, Israel
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1073-1085
-
- Article
- Export citation
Complex exsolution microstructures in ilmenite–pyrophanite from the Garnet Codera dyke pegmatite (Central Italian Alps): an electron microscopy investigation
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1087-1104
-
- Article
- Export citation
Centennialite, CaCu3(OH)6Cl2.nH2O, n ≈ 0.7, a new kapellasite-like species, and a reassessment of calumetite
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1105-1124
-
- Article
- Export citation
The discreditation of girdite
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1125-1128
-
- Article
- Export citation
Synthesis and crystal structure of C2/c Ca(Co,Mg)Si2O6 pyroxenes: effect of the cation substitution on cell volume
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1129-1139
-
- Article
- Export citation
Currierite, Na4Ca3MgAl4(AsO3OH)12·9H2O, a new acid arsenate with ferrinatrite-like heteropolyhedral chains from the Torrecillas mine, Iquique Province, Chile
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1141-1149
-
- Article
- Export citation
Omariniite, Cu8Fe2ZnGe2S12, the germanium analogue of stannoidite, a new mineral species from Capillitas, Argentina
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1151-1159
-
- Article
- Export citation
Correction
Erratum to Gibson (2017) On the nature and origin of garnet in highly-refractory Archean lithospheric mantle: constraints from garnet exsolved in Kaapvaal craton orthopyroxenes (Mineralogical Magazine, 81, 781–809).
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1161-1163
-
- Article
-
- You have access
- Export citation
Research Article
Fluorophlogopite-bearing and carbonate metamorphosed xenoliths from theCampanian Ignimbrite (Fiano, southern Italy): crystal chemical, geochemical and volcanological insights
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1165-1189
-
- Article
- Export citation
Crystal structure determination of karibibite, an Fe3+ arsenite, using electron diffraction tomography
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1191-1202
-
- Article
- Export citation
Three-dimensional study by synchrotron radiation computed tomography of melt distribution in samples doped to enhance contrast
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1203-1222
-
- Article
- Export citation
A re-examination of water in agate and its bearing on the agate genesis enigma
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1223-1244
-
- Article
- Export citation
Dissolution kinetics of hydrated calcium aluminates (AFm-Cl) as a function of pH and at room temperature
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1245-1259
-
- Article
-
- You have access
- Open access
- Export citation
Cancrinite–vishnevite solid solution from Cinder Lake (Manitoba, Canada): crystal chemistry and implications for alkaline igneous rocks
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1261-1277
-
- Article
- Export citation
IMA Commission on New Minerals, Nomenclature and Classification (CNMNC) Newsletter 39
CNMNC Newsletter
New minerals and nomenclature modifications approved in 2017
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1279-1286
-
- Article
-
- You have access
- Export citation

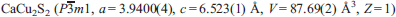 , a thiocuprate, was found in larnite pseudoconglomerate rocks of the Hatrurim Complex at Jabel Harmun, Palestinian Autonomy, Israel. Dzierżanowskite occurs in larnite pebbles, which are embedded in a low-temperature mineral matrix. Associated minerals are larnite, brownmillerite, fluorellestadite, ye'elimite, gehlenite, periclase, ternesite, nabimusaite, vorlanite, vapnikite, fluormayenite, fluorkyuygenite, oldhamite, jasmundite, covellite, chalcocite and pyrrhotite. Electron microprobe analyses yield an average composition of Cu 55.25, Fe 0.13, S 27.46 and Ca 16.99, total 99.83 wt.%. The empirical formula of dzierżanowskite, based on 5 atoms, is Ca
, a thiocuprate, was found in larnite pseudoconglomerate rocks of the Hatrurim Complex at Jabel Harmun, Palestinian Autonomy, Israel. Dzierżanowskite occurs in larnite pebbles, which are embedded in a low-temperature mineral matrix. Associated minerals are larnite, brownmillerite, fluorellestadite, ye'elimite, gehlenite, periclase, ternesite, nabimusaite, vorlanite, vapnikite, fluormayenite, fluorkyuygenite, oldhamite, jasmundite, covellite, chalcocite and pyrrhotite. Electron microprobe analyses yield an average composition of Cu 55.25, Fe 0.13, S 27.46 and Ca 16.99, total 99.83 wt.%. The empirical formula of dzierżanowskite, based on 5 atoms, is Ca , which can be explained in terms of preservation of the oxygen close packing between the ilmenite and columbite structures. The interfaces between any two of the three different phases are coherent but show lattice strain contrast and sometimes dislocations because of their different unit-cell dimensions. On the basis of textural observations, titanohematite is supposed to exsolve first, followed by columbite-tantalite at temperatures below 500°C. The addition of MnO to the Fe
, which can be explained in terms of preservation of the oxygen close packing between the ilmenite and columbite structures. The interfaces between any two of the three different phases are coherent but show lattice strain contrast and sometimes dislocations because of their different unit-cell dimensions. On the basis of textural observations, titanohematite is supposed to exsolve first, followed by columbite-tantalite at temperatures below 500°C. The addition of MnO to the Fe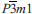 ,
,