INTRODUCTION
Hand, foot, and mouth disease (HFMD) is a common infectious disease that occurs in children aged <5 years and has been reported to be prevalent worldwide after its first description [Reference Alsop, Flewett and Foster1, Reference Ho2]. Large-scale outbreaks have occurred and caused death and neurological sequelae in Malaysia, Taiwan, Singapore, and recently in mainland China [Reference Chan3–Reference Yang7], which has become a growing public health concern throughout South East Asia. HFMD is caused by enterovirus infection. Coxsackie virus A16 (CV-A16) and enterovirus A71 (EV-A71) are the major causative agents of HFMD [Reference Ho2, Reference Robinson, Frances and Rhodes8, Reference Schmidt, Lennette and Ho9]. Enterovirus is a genus of the Picornavirus family, characterized by a single positive-strand genomic RNA. Based on molecular and biological properties of the viruses, human enterovirus is currently designated as four species (A–D), including more than 80 serotypes [10, Reference Nasri11]. Both EV-A71 and CV-A16 belong to human enterovirus A. EV-A71 has often been linked with outbreaks of severe neurological diseases and HFMD complicated by fatal outcome such as lethal central nervous system (CNS) disorders and cardiopulmonary collapse [Reference Ho2, Reference Weng12]. Currently, no vaccine or effective drug against EV-A71 are available for human use. Thus, epidemiological surveillance of HFMD and EV-A71 infection is important in order to understand the emerging trend of this disease and to take the correct and timely public health interventions to prevent the outbreak.
In May 2008, HFMD was included into the notifiable class C infectious diseases category in China because of its large-scale epidemic, beginning first in Fuyang and resulting in the deaths of many children. Since then, it has been observed that the periodic outbreak of HFMD occurs annually with increasing morbidity and mortality in recent years. During 2008–2010, a total of 3 419 149 cases and 1384 fatal cases attributable to HFMD were reported officially in China. HFMD has now become one of the leading causes of child death and a public health priority in China [13]. In fact, there were several regional outbreaks of HFMD in the 1980s in Tianjin caused by CV-A16, and in 2000 and 2007 in Shandong caused by EV-A71 [Reference Yu14–Reference Wang16]. Since 2007, it has been mandatory to report cases of HFMD to Shanghai Municipal Center for Disease Control and Prevention (CDC). Accumulating reported cases across Shanghai exceeded 40 000 during 2007–2010, with about 30% of reported HFMD cases from our hospital. This study retrospectively analyses the epidemiology of HFMD cases brought to our hospital for treatment during the past 4 years.
METHODS
Patients and definition of clinical diseases
All HFMD cases in this study were reported to Shanghai CDC on the ground of clinical diagnosis at the Children's Hospital of Fudan University between 2007 and 2010. Our hospital hosts the largest-scale dedicated HFMD clinics and wards in Shanghai. At least 90% of hospitalized HFMD cases were treated in our hospital.
HFMD is characterized by oral vesicular exanthema/ulcers plus vesicular lesions on the hands, and/or feet, and/or buttocks. A case was defined as severe if HFMD was accompanied with the occurrence of at least one of the following complications: aseptic meningitis, encephalitis, acute flaccid paralysis, pulmonary oedema or haemorrhage, cardiopulmonary collapse or myocarditis. All diagnostic criteria of clinical complications refer to the disease definition as described previously in Taiwan [Reference Chen17, Reference Chang18]. Cerebrospinal fluid (CSF) pleocytosis was defined as a white blood cell count >10×106 cells/l in a CSF sample if a patient was aged >1 month, which is a little different from the Taiwanese criteria. Encephalitis was diagnosed by a disturbance in the level of consciousness such as drowsiness or coma plus CSF pleocytosis.
The criteria for hospitalization included one of the following conditions: 3 days of persistent fever, recurrent vomiting, altered consciousness (lethargy or drowsiness or coma), neck stiffness, startle, myoclonic jerk, seizure, tachypenia, Occasionally, some patients were admitted to wards after strong parental request.
Identification of EV-A71 and CV-A16
The virological surveillance for severe (clinically confirmed or highly suspected) and lethal cases has been in operation since 2009 by Shanghai CDC. Once a patient was confirmed as being severe enough or highly suspected of being very ill, a stool sample and throat swab were collected and sent to the CDC reference laboratory for enterovirus detection. In 2009, 120 (69·36%) out of 173 severe cases were tested for enterovirus; 84 inpatients with no complications had virus detected in clinical samples at the CDC laboratory, since these 84 cases were excluded from having neurological disorders but were highly suspected of being severe on admission. From May 2010 onwards, EV-A71 and CV-A16 were routinely detected in our clinical virology laboratory. Moreover, it was mandatory for clinicians to send clinical samples from severe cases (in terms of clinical situation) to the CDC laboratory for virus identification. Altogether in 2010, 1506 (74·37%) of 2025 inpatients were tested for EV-A71 and CV-A16, and 431 (97·07%) out of 444 severe cases were tested for enterovirus. In addition, 413 CSF specimens from inpatients with CNS involvement were tested for EV-A71 and CV-A16 in our hospital virology laboratory in 2010.
A commercial licensed kit (Da An Gene Co. Ltd, lot no.: Cox A16 YZB-0354-2009, EV-A71 YZB-0356-2009) was recommended by China CDC for detection of EV-A71 and CV-A16 for clinical samples in the CDC laboratory and hospital virology laboratory. The detection method is based on one-step real-time RT–PCR assay. The detection sensitivity of the kit is 1×103 p.f.u/ml. A specimen is considered positive for virus if reaction growth curves cross the threshold line within 35·1 cycles.
Statistics
Statistical analysis was performed using SPSS version 11.5 software (SPSS Inc., USA). The χ2 test was used for proportional data. Age comparison was analysed using the Mann–Whitney U test. A difference with a P value <0·05 was considered as significant.
RESULTS
Trend and seasonality of HFMD epidemic in Shanghai
There were a total 28 058 cases with HFMD reported to CDC in our hospital, of whom 3948 (14·07%) were admitted to the isolation wards for treatment, 730 (2·60%) were severe and 11 (0·04%) died from HFMD. The fatality of severe cases was 1·51% (11/730). The numbers of HFMD cases increased markedly year on year (Table 1).
Table 1. Case distribution and demographic characteristics of reported HFMD cases in the Children's Hospital of Fudan University, Shanghai
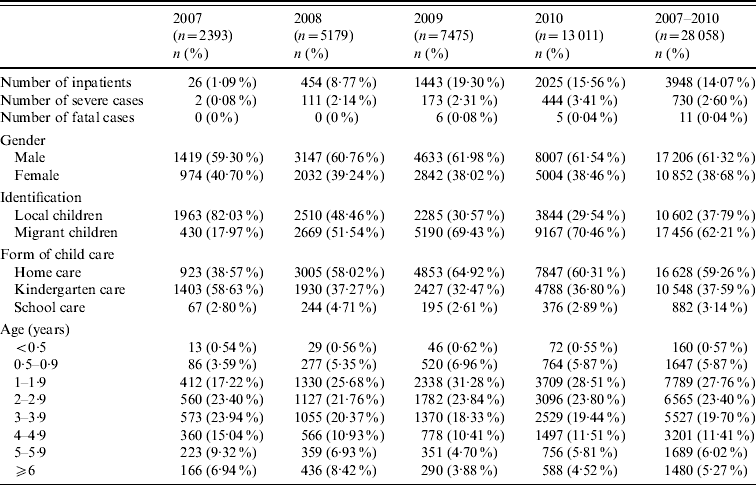
Percentages in parentheses indicate a proportion in the total number of a corresponding year.
HFMD was prevalent throughout a year in Shanghai and peaked between May and July during summertime (Fig. 1). In 2010 when the largest-scale outbreak of HFMD occurred in Shanghai, the peak season was in April, 1 month earlier than that seen in previous years. In August, the activity of HFMD fell sharply. However, a second smaller and unusual epidemic wave of HFMD was observed in middle autumn and winter of 2009, just preceding the largest-scale outbreak of HFMD in 2010. The numbers of hospitalized cases and severe cases were parallel with the peak of HFMD (shown in Figs 1 and 2).
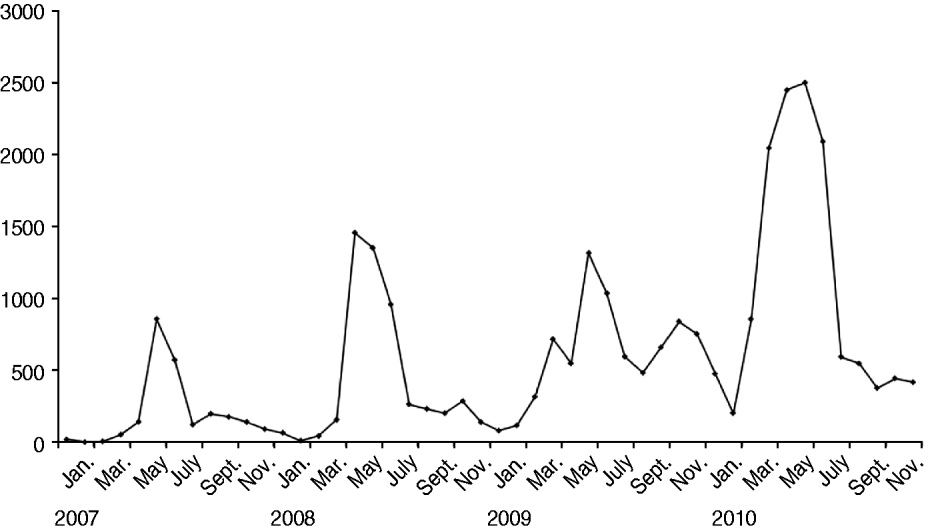
Fig. 1. Monthly numbers of outpatients with HFMD reported to Shanghai Municipal CDC at the Children's Hospital of Fudan University, 2007–2010.
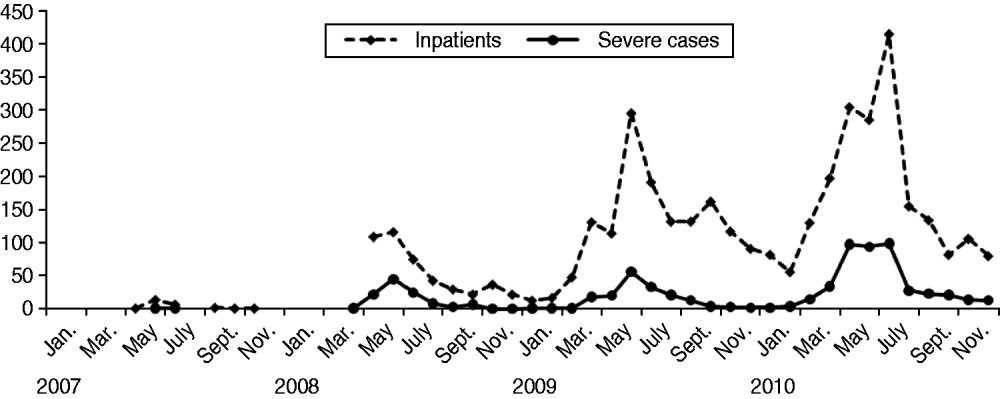
Fig. 2. Monthly numbers of inpatients and severe cases with HFMD from the Children's Hospital of Fudan University, 2007–2010.
Demographic characteristics of HFMD
Of 28 058 children with HFMD, 61·32% (17 206) were boys and 38·68% (10 852) were girls (male-to-female ratio 1·6:1), and the male predominance was found in each year. In total, local Shanghainese children accounted for 37·79% (10 602) and migrant children accounted for 62·21% (17 456). The proportion of susceptible population varied with year. In 2007, local children constituted 82·03% of total cases; moreover, the majority (58·63%) of outpatients were children attending kindergarten. However in 2008, most children affected shifted to migrant children and children cared for at home (see Table 1) and this trend was more marked in 2009 and 2010. In addition, of 10 602 local children with HFMD, children cared for at home and institutionalized children were 4074 (38·43%) and 6528 (61·57%), respectively. Of 17 456 migrant children with HFMD there were 12 554 (71·92%) cared for at home and 4902 (28·08%) cared for in an institution. The incidences of HFMD in institutional care and home-care settings between local and migrant children were statistically significant (χ2=3064·544, P=0·000).
Of 28 058 patients aged between 22 days and 16 years, 23 082 (82·27%) were young children aged 1–4 years. The proportion of 1-year-old children was largest, accounting for 29·67% of total cases, except in 2007 when the 1-year-old group constituted 17·22% of all cases (Table 1). There was an obvious upward trend in the numbers of children aged 1 year by year.
Characteristics of severe and fatal HFMD cases
Of the total 730 severe cases, 711 were confirmed as having complications of aseptic meningitis or encephalitis, six had encephalitis combined with acute flaccid paralysis, three cases had encephalitis combined with pulmonary oedema/haemorrhage, one CV-A16-infected child died without a definite death cause, and nine cases were complicated by pulmonary oedema/haemorrhage. The nine cases with pulmonary oedema/haemorrhage were too critical to have lumbar puncture performed for CSF test; however, they met a clinical diagnostic criterion of encephalitis since they all manifested with fever, drowsiness or coma, vomiting, convulsion, tremor, myoclonic jerk, and limb weakness in various combinations. The temporal distribution of severe HFMD cases was parallel to the peak of HFMD epidemics (Figs 1, 2). With one exception, all very critical pulmonary oedema/haemorrhage cases occurred between April and July during the HFMD epidemic.
The mean age of the 730 severe cases was 28 months (range 3 months to 12 years). The proportions of age groups in children aged <0·5 year, 0·5–0·9 year, 1 year, 2 years, 3 years, 4 years, 5 years and ⩾6 years were 0·14% (1), 5·77% (42), 31·73% (233), 25·41% (185), 19·51% (142), 10·23% (73), 3·57% (26) and 3·85% (28), respectively. Of 730 severe cases, 473 (64·79%) were boys and 257 were (35·21%) girls. The incidences of severe HFMD in males and females were 2·75% (473/17 206) and 2·37% (257/10 852), respectively. There was no statistical difference between the two groups (χ2=3·808, P=0·051). The 730 severe cases comprised 522 (71·51%) migrant children and 208 (28·49%) local children. The incidences of severe HFMD in migrant children and local children were 3·00% (522/17 396) and 1·95% (208/10 662), respectively. There was statistical significance in the two groups (χ2=28·751, P=0·000).
Among 12 critically severe cases with pulmonary oedema/haemorrhage and one fatal case of unknown cause, the ages ranged from 7 months to 63 months with the median age of 22·5 months (mean age 25·75±14·92 months). The age distribution was as follows: <1-year group (n=1, 7·69%), 1-year group (n=5, 38·46%), 2-year group (n=5, 38·46%), 3-year group (n=1, 7·69%) and 5-year group (n=1, 7·69%). There were seven (53·85%) boys and six (46·15%) girls. Eleven (84·62%) were migrant children and two (15·38%) were local children. Thirteen cases resided in the suburban or rural areas and six (46·15%) had definite contact history.
Laboratory detection of EV-A71 and CV-A16
During the period April–November 2009, it was found that 99·2% of severe inpatients, 83·33% of deceased cases and 51·19% of uncomplicated inpatients were confirmed with EV-A71 infection at the CDC laboratory; only one severe case with meningitis and one fatal case of unknown complication were caused by CV-A16 infection; CV-A16 and other untyped enteroviruses accounted for 36·90% and 14·29%, respectively, of uncomplicated HFMD inpatients (Table 2). Of 1506 inpatients with virus detected in stool samples in 2010, 815 (54·12%) were EV-A71 positive, 211 (14·01%) were CV-A16 positive, five (0·33%) were positive for both EV-A71 and CV-A16, and the remaining 475 (31·54%) were negative for both EV-A71 and CV-A16 (Table 2). It was observed that EV-A71 was a principal causative agent responsible for 86·08% of severe HFMD cases and 100% of death; minor severe cases were infected with CV-A16 and untyped enterovirus which accounted for 2·32% and 11·83%, respectively (Table 2). Based on pathogen finding from 1506 cases, severe complications occurred in 45·37% (372/820) of EV-A71-positive cases, 4·17% (9/216) of CV-A16-positive cases and 10·74% (51/475) of cases with non-EV-A71 and non-CV-A16 infection (χ2=277·861, P=0·000). In the meantime, 413 severe cases with meningitis or encephalitis also had virus detected in CSF, and only six (1·44%) specimens were positive for EV-A71. Overall, in 12 children complicated with pulmonary oedema or haemorrhage, EV-A71 was positive in both fecal specimens and throat swabs, but EV-A71 was negative in CSF collected from three critically ill cases.
Table 2. Detection of EV-A71 and CV-A16 in inpatients with HFMD, 2009–2010

Percentages in parentheses indicate the proportion in the same group. Severe group included fatal group.
* Two cases were infected with EV-A71 and CV-A16.
† One case was infected with EV-A71 and CV-A16.
‡ Four cases were infected with EV-A71 and CV-A16.
The distribution of enterovirus types by month in 2010 is shown in Figure 3. A gradual decline in the proportion of EV-A71 in inpatients was observed from the peak month onwards. However, the positivity of EV-A71 tended to rise from September, coinciding with the opening of the kindergarten.
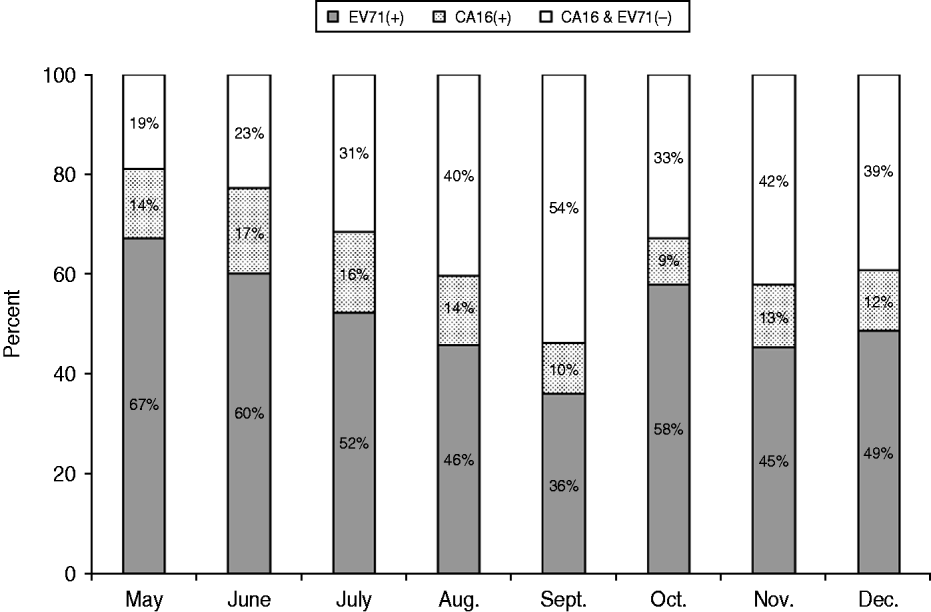
Fig. 3. Monthly distribution of EV-A71, CV-A16 and untyped enterovirus in hospitalized children with HFMD from the Children's Hospital of Fudan University, between May and December 2010.
DISCUSSION
Our data reveals that HFMD was epidemic annually in Shanghai and the prevalence of HFMD has tended to increase over the past 4 years; HFMD usually affects younger children and most severe neurological diseases and child death are caused by EV-A71 infection. The dominant circulation of EV-A71 gave rise to the continuous outbreaks of HFMD in Shanghai. So far, Shanghai has experienced an unprecedentedly large outbreak of HFMD in 2010, and this pattern is consistent with the nationwide trend released by Chinese Ministry of Health. The largest-scale outbreak of HFMD in Taiwan appeared in 1998, and afterwards the activity of HFMD was not higher than that seen in 1998 [Reference Wang16, Reference Chen17]. We postulate that the distinct epidemic model of HFMD in mainland China is due to China's highly dense and dynamic population and vast geographical area.
HFMD is prevalent year round and usually peaks in the summer month. However, the peak season of HFMD in the pandemic year of 2010 appeared in advance, as early as April. High activity of HFMD ends in August soon after the summer vacation. The seasonality of HFMD seen in Shanghai is similar to the national seasonal pattern [13]. The summer peak was also observed in Taiwan and Hong Kong [Reference Chen17, Reference Ma19]. However, in subtropical areas such as Singapore and Malaysia, the HFMD peak occurs usually in March or May (Singapore) and between February and April (Malaysia) [Reference Ang20, Reference Podin21]. Seasonal variation in different regions could be related to climate, as suggested by some studies [Reference Ma22, Reference Urashima, Shindo and Okabe23]. Of particular note, a second HFMD peak was prominent in Shanghai between mid-autumn and winter in 2009, and subsequently the largest outbreak of HFMD occurred in 2010. Thus, we hypothesize that unusually high activity of HFMD in autumn and winter could predict a large outbreak in the following year. Ongoing monitoring of HFMD is useful to understand the factors correlated with the HFMD epidemic, which is important in allowing public health authorities to take the corresponding measures to control and mitigate the outbreak.
Our study showed that 81·93% of children with HFMD and 86·71% of severe cases were aged 1–4 years, comprising the majority of reported cases. In general, the age distribution of susceptible children is similar to that observed in other Asian regions [Reference Chen17, Reference Ma19–Reference Podin21]. Of note, children aged <2 years constituted 75% of severe cases in Taiwan, but only 37·64% of severe cases in Shanghai [Reference Chen17]. The reason for this difference was that severe HFMD cases occurred in infants much more commonly in Taiwan than in Shanghai. A reasonable explanation of this phenomenon could be relevant to the levels of maternal antibody in infants. The seroepidemiology from Taiwan showed maternal antibody against EV-A71 declined to almost undetectable levels by age 6 months [Reference Luo24]. In Shanghai, the majority of severe HFMD cases were caused by EV-A71. Dominant circulation of EV-A71 in recent years in Shanghai could boost maternal EV-A71-specific antibody, as a result, the titres of antibody in infants could be at a higher level and persist for longer. We have observed that the 1-year age group was most susceptible to HFMD, accounting for about 30% of total cases since 2008. This speculation needs further seroprevalence study.
We found that more boys were affected by HFMD than girls. The male predominance was also found in Taiwan, Singapore, Malaysia and Hong Kong and other enterovirus infection reported by USA and Spain [Reference Chen17, Reference Ma19–Reference Podin21 Reference Chen17, Reference Ma19–Reference Podin21, Reference Trallero25, Reference Khetsuriani26]. This may indicate that boys are more susceptible to enterovirus. However, our study does not show that boys are more likely to develop severe complications than girls. Whether host genetic background is linked with susceptibility of HFMD and severe complications requires further investigation.
Interestingly, we observe that children cared for locally in kindergarten constituted the majority of HFMD cases in 2007 when HFMD was first officially reported. However, since 2008, migrant children have substantially outnumbered local children. In the meantime, we observe that HFMD occurred more often in children in local institutional care and in migrant children cared for at home. The shift and characteristics of affected population suggests: (1) the outbreak of HFMD in Shanghai originated from the kindergarten setting and spread widely throughout the community. This pattern is similar to the transmission mode of 2009 pandemic H1N1 influenza outbreak in Shanghainese children (Shanghai Municipal CDC, personal communication). Although we do not know the epidemiological data of HFMD prior to 2007, we reason that children living in Shanghai were susceptible to HFMD in 2007; a large outbreak of HFMD ensues once a sudden increase of disease attack occurs in children in an institutionalized setting, and moreover, spread of HFMD in non-childcare centres is the main mode of transmission in Shanghai. Based on the studies in Taiwan, transmission rates of EV-A71-associated HFMD/herpangina in children following household contact and non-family contact were as high as 74% and 69%, respectively [Reference Chang27], and the transmission rate of CA16 in children attending preschool was 61·9% [Reference Wu28]. Only 29% of EV-A71-seropositive preschool children develop HFMD/herpangina [Reference Chang27]. Hence, the high infectivity of EV-A71 and high ratio of asymptomatic EV-A71 infection pose a big challenge for infection and control. (2) The constant outbreak of HFMD in Shanghai is relevant to susceptible migrant children cared for at home. Since the national outbreak of HFMD in May 2008, the China Ministry of Health and local relevant government agencies have initiated several measures to control the outbreak of HFMD in institutional setting. These actions include stringent disinfection of toys and eating utensils, morning check, hand hygiene, and isolation of symptomatic children for 2 weeks. From 2009 onwards, if clusters of two or more HFMD cases in one class or ten cases in one preschool/school within 1 week are reported, a field investigation will be carried out and school/class closure will be recommended. Evidently, the fact that institutional HFMD cases decreased markedly indicates that implementation of these strategies had some impact on reducing the transmission of HFMD in preschool children, and especially for local Shanghainese children. In Singapore, the stringent implementation of infection and control measures prevents the outbreak of HFMD in the institutional setting and to some extent decreases the attack rate of HFMD, which also leads to more cases occurring in children aged >5 years [Reference Podin21, Reference Ma22]. Nevertheless, public health policies have little effect on migrant children cared for at home, who are usually from families of low socioeconomic status and poor educational background, live in rural areas and crowded areas with poor sanitation facilities, have poor personal hygiene, and have more person-to-person contact between siblings, neighbours and relatives, and also more outdoor activity. Additionally, some migrant children have one or more siblings. These factors are correlated with the risk of contracting HFMD [Reference Chang27, Reference Park29].
EV-A71-associated HFMD was actually brought to the attention of the public health agency in 2007, when a HFMD outbreak occurred in Shandong province and EV-A71 was identified as a leading pathogen contributing to the HFMD epidemic [Reference Zhang6]. In 2008, a national outbreak of HFMD originating from Fuyang, Anhui province occurred and many fatal cases were linked to the prevalence of EV-A71 alerted by public health and social concerns [Reference Yang7]. We found that EV-A71 was associated with almost all fatal HFMD and >50% of child admission for HFMD during 2009 and 2010. Based on virus examination in clinical samples in 2010, EV-A71 was far more likely to cause severe neurological complications compared to CV-A16 and other types of enterovirus. Nevertheless, EV-A71 was rarely detected in CSF samples from cases involving CNS. Previous studies have also shown that EV-A71 is difficult to detect in CSF samples [Reference Chang30–Reference Pérez-Vélez32]. Possible explanation could be a lower amount of viral load in CSF, sensitivity of the PCR assay, or different neuropathogenesis associated with EV-A71. A study from our department reported that EV-A71 was identified in 22 (46·81%) of 47 faecal samples from HFMD inpatients in 2008 [Reference Liu33]. The above data reflect that the widespread and dominant circulation of EV-A71 in children in Shanghai results in a sustained outbreak of HFMD and severe diseases over recent years. Previous studies from China reported EV-A71-associated HFMD occurred in Shanghai in 2002 and in Shenzhen between 1999 and 2004; however, CA-A16 was a principal causative agent at that time, when the ratio of EV-A71 to CV-A16 was 1:6 and 1:2·2 in Shanghai and Shenzhen, respectively [Reference Yang34, Reference Li35]. On the basis of the epidemiological data, an EV-A71 epidemic occurs cyclically every 2 or 3 years in Malaysia and Japan [Reference Podin21, 36], and HFMD has occurred in the UK periodically [37]. Whether this cyclical pattern of EV-A71 and HFMD will be seen in Shanghai requires ongoing surveillance. Despite the fact that EV-A71 is a major public health concern in mainland China, attention should be paid to other types of enterovirus as well because 56% of all HFMD/herpangina-associated deaths in Taiwan between 1998 and 2005 were related to non-EV-A71 infection [Reference Chen38]. The limitation of our study is that we failed to detect other serotypes of enterovirus except EV-A71 and CV-A16 and monitor enterovirus in outpatients with HFMD.
This study shows that HFMD will not be brought under control even if public health measures are taken rigorously for 3 years. Therefore, the HFMD epidemic will remain in subsequent years and impose a threat to child health before vaccination can be introduced for children. Development of an effective vaccine should be a priority in controlling HFMD outbreaks and fatal outcomes. An ongoing HFMD surveillance programme is needed for early warning of outbreaks and taking correct decisions for containing the outbreak, and for future vaccine development.
DECLARATION OF INTEREST
None.









