Relapse after treatment for acute malnutrition is a serious problem facing the humanitarian and development communities. A few studies that have followed up children after initial recovery from moderate acute malnutrition (MAM) have shown that such a relapse is common( Reference Chang, Trehan and Wang 1 – Reference Stobaugh, Bollinger and Adams 3 ). For example, previous research in Malawi found that only 51–63 % of children sustain recovery for 12 months after initial treatment for MAM( Reference Chang, Trehan and Wang 1 , Reference Stobaugh, Bollinger and Adams 3 ). Common childhood illnesses, such as fever, cough, malaria and diarrhoea, are prevalent among those who relapse or die after initial MAM recovery( Reference Chang, Trehan and Wang 1 , Reference Ashraf, Alam and Chisti 4 – Reference Somasse, Dramaix and Bahwere 6 ). Mid-upper-arm circumference (MUAC) and weight-for-height z score (WHZ) at enrolment and discharge from a supplementary feeding programme (SFP) have shown to be predictive of relapse during the following year, although duration of treatment was not( Reference Trehan, Banerjee and Murray 7 ). A recent study from Burkina Faso that included 90 % of children with MAM and 10 % with SAM reported factors with relapse to be low MUAC at discharge, low oil/fat consumption during the follow-up period and incomplete vaccination( Reference Somasse, Dramaix and Bahwere 6 ). Among children with SAM, a study in Malawi identified children with HIV to have a higher risk of relapse after treatment( Reference Kerac, Bunn and Chagaluka 8 ). Currently, there is no standardised definition of relapse, and the varying rates that have been identified may be because of different follow-up time periods and methodologies( Reference Chang, Trehan and Wang 1 – Reference Begashaw 5 , Reference Somasse, Dramaix and Bahwere 9 ). These studies have begun to shed light on factors associated with relapse, yet large gaps in knowledge remain.
It is plausible that returning to the same household (HH) environment that may have contributed to the initial development of MAM may predispose children to relapse. Infant and young child feeding (IYCF) practices( Reference Khatoon, Mollah and Choudhury 10 – Reference Sawadogo, Martin-Prével and Savy 13 ), unsanitary living conditions( Reference Humphrey 14 , Reference Ngure, Reid and Humphrey 15 ), poor food security and dietary diversity( Reference Kennedy, Pedro and Seghieri 16 , Reference Moursi, Arimond and Dewey 17 ) and impoverished socio-economic status (SES)( Reference Nandy, Irving and Gordon 18 , Reference O’Donnell, van Doorslear and Wagstaff 19 ) have all been linked to developing acute malnutrition in various contexts. However, prior studies have not specifically correlated these factors with relapse following initial recovery from MAM. Identifying HH risk factors associated with relapse could have significant implications for how best to prevent relapse and improve the sustainability of MAM treatment. To identify such factors, we conducted an in-depth HH survey among children who were followed up for 1 year after recovery from MAM.
Methods
Study design and objective
This observational study consisted of an in-depth HH survey administered prospectively at the time of SFP discharge to a sub-sample of participants within a larger cluster randomised controlled trial (cRCT). Complete methods of the parent cRCT have previously been described in detail( Reference Stobaugh, Bollinger and Adams 3 ). In brief, children aged 6–62 months who had recovered from MAM, defined as MUAC≥12·5 cm without bipedal oedema( 20 ), were recruited from rural health clinics in southern Malawi. In all, eleven clinics were randomly assigned to receive nutrition counselling in addition to a package of health and nutrition interventions at the time of SFP discharge, whereas ten clinics were randomly selected to receive nutrition counselling only. Children were followed up for 12 months after discharge to assess the impact of the intervention package on sustained recovery.
The aim of this study was to identify HH factors that may be associated with sustained recovery, including SES, dietary diversity, food insecurity, child care practices and water, sanitation and hygiene (WASH). The HH survey was conducted only among children in the control group in the parent cRCT( Reference Stobaugh, Bollinger and Adams 3 ).
Study setting
The study was conducted in five districts (Chikwawa, Chiradzulu, Machinga, Mulanji and Zomba Districts) across the Southern Region in Malawi where most of the population consists of subsistence farmers. Conditions are often unsanitary, without access to clean drinking water, and HH are often at far distances from functioning health clinics. Child malnutrition is widespread in Malawi; over 42 % of children under 5 years old are stunted and 17 % are underweight( 21 ).
Subject participation
Children were enrolled in the study at the time of discharge from an SFP. These children were the last 315 children enrolled in all control sites from the parent cRCT study. Upon enrolment, caregivers scheduled an appointment for a home visit within a week of the child’s discharge from the SFP. The survey was administered at the child’s home by a trained data collector (either a senior paediatric research nurse or a community health worker) and lasted approximately 1 h. Caregivers were asked to return to the clinic for subsequent follow-up visits at 1, 3, 6 and 12 months after enrolment to reassess the child’s nutrition status and clinical signs of illness, including diarrhoea and fever. HH food security was also assessed at each follow-up visit using the validated, nine-item Household Food Insecurity Access Scale (HFIAS)( Reference Coates, Swindale and Bilinsky 22 ).
Household survey
Data collected from the HH survey consisted of information pertaining to (1) SES; (2) IYCF practices; (3) HH food security; (4) WASH; and (5) maternal perceptions of MAM, the SFP and relapse following MAM.
Socio-economic status
We collected indicators based on an SES index by Psaki et al.( Reference Psaki, Seidman and Miller 23 ), recently validated in an eight-country study. This index, called the WAMI index, comprised four main components – water and sanitation, assets, maternal education and HH income. WAMI was chosen because of its simplicity for field use and association with height-for-age z score( Reference Victora, Requejo and Barros 24 ) in childhood( Reference Psaki, Seidman and Miller 23 ). However, in this study, monthly HH income was not surveyed because many of the families in this context have only informal avenues of widely fluctuating monthly income( Reference O’Donnell, van Doorslear and Wagstaff 19 ). In addition, the water and sanitation component was not included to avoid redundancy with other WASH indicators elsewhere in the final model. To account for these deviations, we collected information on additional assets and livestock to help distinguish different levels of wealth within the local context. After conducting a principal component analysis, we determined that a single SES measure was not appropriate for this data set because principal component analysis showed lack of correlation between indicators. Therefore, rather than using one overall SES index, we included the following individual SES-related variables in the final regression model: maternal education, number of HH assets (out of eleven total), number of rooms in a house and ownership of any livestock. Table 1 contains information on the definition and scoring of each SES indicator.
Table 1 Socio-economic status indicators from the household survey

Infant and young child feeding practices
Indicators included in the HH survey regarding IYCF practices were based on current international recommendations( 25 , 26 ). Indicators included (1) breast-feeding practices, (2) the introduction of complementary foods, (3) minimum dietary diversity and (4) minimum meal frequency. Because recommended feeding practices vary by age( Reference Sawadogo, Martin-Prével and Savy 13 , 25 , 26 ), information used in the breast-feeding practices and minimum meal frequency indicators differed according to the age of the child. Table 2 contains information on the definition and scoring of each IYCF practice indicator.
Table 2 Infant and young child feeding practices indicators from the household survey

Food security
HH food security was assessed using HFIAS( Reference Coates, Swindale and Bilinsky 22 ), a series of nine questions regarding the food security situation at the HH level. The HFIAS has been validated in numerous countries around the world( 20 ). Scores range from 0 to 27, and higher scores represent increased food insecurity, whereas lower scores represent better food security. HFIAS was administered to the caregiver at the time of admission into the SFP, as well as 1, 3, 6 and 12 months after SFP discharge. Scores from all time points throughout the year were averaged. Food security fluctuates throughout the year between times when food availability is higher just after the single annual harvest and times when food is scarcer before harvest. An arithmetic average over the course of the year was chosen to help account for these fluctuations.
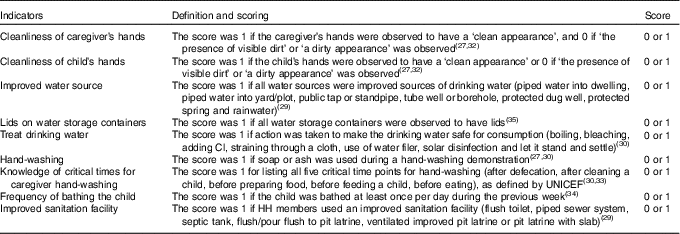
Water, sanitation and hygiene
WASH indicators used by international agencies( Reference Ram 27 – 29 ) and the 2011 Demographic and Health Survey (DHS)( 30 ) were included to capture WASH conditions and practices among participating HH. Given prior associations between cleanliness of hands and child health outcomes( Reference Luby, Halder and Huda 31 , Reference Pickering, Davis and Walters 32 ), the caregiver’s hands and the child’s hands were visually inspected for cleanliness, which included a three-point scale denoting ‘clean,’ ‘no visible dirt but unclean appearance’ and ‘visible dirt’ regarding the palms, finger pads and finger nails (later combined into two categories of clean and unclean)( Reference Ram 27 ). During data collection training, data collectors were shown several examples of proper scoring, including tests of intra- and inter-rater reliability. This training included large group demonstrations and small group exercises. A final test was administered at the end of the training to ensure that proper scoring was demonstrated.
Use of improved water sources and use of sanitation facilities were based on the WHO definitions( 29 , 30 ). Respondents were asked if they take action to treat water, according to the DHS format( 30 ). Water storage containers were assessed for having fitted lids( 33 ). Hygiene was assessed by direct observation of whether a caregiver used soap during a hand-washing demonstration( Reference Ram 27 , Reference Vujcic and Ram 28 ), knowledge regarding five critical times for hand washing( Reference Vujcic and Ram 28 , 30 , 33 ) and the frequency with which the child is bathed( Reference Khatri, Mishra and Khanal 34 ). Table 3 contains information on the definition and scoring of each WASH indicator.
Table 3 Water, sanitation and hygiene indicators from the household (HH) survey
Outcome definitions
At the end of the 12-month follow-up period, each child was classified as having ‘sustained recovery’, defined as having MUAC≥12·5 cm at every follow-up visit for 12 months; ‘relapsed to MAM’, defined as MUAC<12·5 cm and≥11·5 cm at any point during the follow-up period; ‘developed SAM’, defined as MUAC<11·5 cm and/or bipedal oedema (kwashiorkor) at any point during the follow-up period; ‘died’; or ‘lost to follow-up,’ defined as defaulting on a scheduled visit and never returning. Poor outcomes were considered to be relapsing to MAM, developing SAM, being lost to follow-up or dying. If a child experienced two such outcomes over the course of the follow-up period, the more severe category was assigned as the final outcome.
To date, there is no consensus on the definition of relapse after recovery from MAM. Our definition of relapse and the time period in which we followed up children was chosen in order to compare results with previous studies that measured relapse similarly( Reference Chang, Trehan and Wang 1 , Reference Stobaugh, Bollinger and Adams 3 ). Furthermore, in order to better understand the nutritional status following initial recovery from MAM, we chose to err on the side of inclusion, rather than exclusion, for our final definition of relapse. Therefore, we chose a 12-month follow-up time period so as to capture both children who relapse in the first few months following discharge and also those who relapse repeatedly over the course of a year. Following up children for a full year also allowed us to account for seasonality in all study participants. A relapse during the hungry season that occurs several months after initial recovery from MAM could be perceived as a ‘new episode’ of MAM, given the increased likelihood of malnutrition during this season of high food insecurity. However, it has been demonstrated that children who recover from acute malnutrition (either SAM or MAM) are at a higher risk of relapse, morbidity and mortality than those who were not previously malnourished( Reference Chang, Trehan and Wang 1 , 36 ). Therefore, because a previously malnourished child is at a higher risk than a non-previously malnourished children, we defined additional malnutrition during (or not during) the hungry season within the follow-up period as a relapse rather than a new episode.
Ethical approval
The study was conducted according to the guidelines laid down in the Declaration of Helsinki, and all procedures involving human subjects were approved by the University of Malawi’s College of Medicine Research and Ethics Committee (P. 11/12/1311), Washington University’s Human Research Protection Office (ID: 201301124) and Tufts University’s Health Sciences Campus Institutional Review Board (IRB#: 11887). Permission to conduct the study was obtained by each site’s District Health Officer and/or District Nutritionist. Written informed consent was obtained from all caregivers.
Statistical analyses
Sample size was determined by the feasibility of recruiting children in the time frame during April to June 2015 of enrolment into the larger cRCT. Recruitment was limited to this time period as the inception of the study idea began after the larger cRCT was underway; therefore, the largest number of participants were recruited after ethical approval was obtained and before the close of the larger study. Given the total sample size of 312 and to achieve a statistical power of 80 % and a two-sided significance level of 0·05, minimally detectable effect sizes were calculated retrospectively to be a 12 to 16 percentage point difference between proportions for binary variables and a mean difference of 0·9, 0·3, 0·4, 0·4, 1·1 and 1·2 for years of education completed by the caregiver, number of rooms in the house, meal frequency, dietary diversity score, average HFIAS score and number of times the child was bathed during the previous week, respectively.
All data were double-entered into an Access (Microsoft Corp.) database and verified against original forms when discrepancies were identified. To compare individual indicators between HH with children who sustained recovery and those who did not, bivariate analyses were conducted using Student’s t test for continuous variables and chi-squared for binary variables with adjustment for clustering at the health clinic level. P values<0·05 were considered to be statistically significant.
Binary logistic regression was used to identify the indicators associated with sustained recovery while accounting for other factors. Cluster-adjusted robust standard errors were used to account for the clustering at the health clinic level. Variables used in the full model included sex; age at the time of admission to SFP; whether the child had fever during the 2 weeks before admission into the SFP; whether the child had diarrhoea during the 2 weeks before admission into the SFP; discharge MUAC; discharge WHZ score; years of education completed by the caregiver; number of rooms in the house; whether any livestock was owned; number of HH assets owned (out of eleven); whether the child was currently breast-feeding at the time of SFP discharge (for children under 24 months) or whether the child continued to breast-feed up to the age of 24 months (for children over 24 months); minimum dietary diversity; minimum meal frequency; average HFAIS score from all follow-up visits; whether all of the drinking water was retrieved from improved water sources; use of an improved sanitation facility; cleanliness of caregiver’s hands; cleanliness of child’s hands; and whether the child was bathed daily during the previous week. All statistical analyses were conducted using Stata Version 13.0 (StataCorp LP).
Results
Out of the 1487 children included in the parent cRCT, a total of 315 children were enrolled in this study and completed the in-depth HH survey. Three surveys were excluded because of uninterpretable data, leaving a total of 312 for final analysis. The proportion of children who sustained recovery for the duration of the 12-month follow-up period after initial recovery from MAM was 58 %.
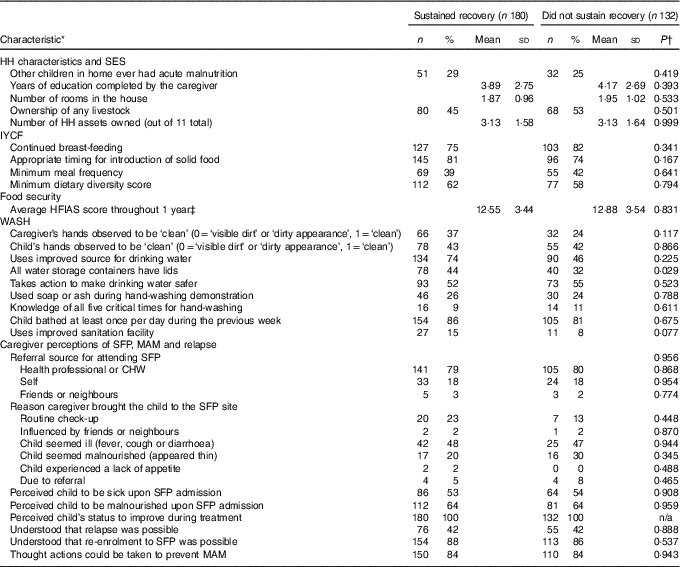
In bivariate analysis comparing HH indicators between those who sustained recovery and those who did not, few individual indicators differed (Table 4 and online Supplementary Table S1). A larger proportion of HH with a child who sustained recovery had lids on all water storage containers than those HH whose child did not sustain recovery (Table 4). Although not statistically significant, caregivers’ hands were observed by enumerators to be cleaner among children who sustained recovery. In addition, the proportions of HH that used an improved sanitation facility or improved sources for drinking water were nearly twice as high among the sustained recovery group compared with those who failed to sustain recovery (although without statistical significance). No differences were found between the two groups regarding SES indicators; IYCF practices; food security; or maternal perceptions of SFP, MAM and relapse.
Table 4 Comparison of indicators regarding household (HH) characteristics: socio-economic status (SES); infant and young child feeding (IYCF) practices; water, sanitation and hygiene (WASH) conditions; and maternal perceptions between children who sustained recovery and those who did not sustain recovery following treatment for moderate acute malnutrition (MAM) in the supplementary feeding programme (SFP)(Numbers and percentages; mean values and standard deviations)
HFIAS, Household Food Insecurity Access Scale (0–27); CHW, community health worker.
* Missing values ranged from 0 to 7 for all indicators with the exception of perceived child to be malnourished upon SFP admission; used soap or ash during hand-washing demonstration; continued breast-feeding; and perceived the child to be sick upon SFP admission, which had 11, 13, 15 and 31 missing values, respectively.
† P values derived using Student’s t test or χ 2 with adjustment for clustering. Minimally detectable effect sizes were calculated retrospectively for proportions to be a 12 to 16 percentage point difference for all main binary variables and a mean difference of 0·9, 0·3, 0·4, 0·4, 1·1 and 1·2 for years of education completed by the caregiver, number of rooms in the house, meal frequency, dietary diversity score, average HFIAS score and number of times the child was bathed during the previous week, respectively. For example, we have 80 % power to detect a mean HFIAS difference of 1·1. Although the mean difference in HFIAS score is <1·1 (at 0·33 HFIAS score), as seen here, this translates to extremely similar HH food insecurity situations between the two groups and we can confidently conclude no statistical or practical difference.
‡ HFIAS ranging from 0 to 27 was assessed at 1, 3, 6 and 12 months of follow-up. These scores were averaged to provide the food security situation at the household level at multiple points throughout the year. Changes in participants’ HFIAS scores between visits ranged from 0 to 23 and averaged a ten-point difference between the lowest and highest HFIAS scores across follow-up visits.
Similar results were found when controlling for other factors in binary logistic regression (Table 5). HH that had fitted lids on all water storage containers were more likely to have a child who sustained recovery than those who did not have lids on all storage containers. Caregivers whose hands appeared to be clean and HH that used an improved sanitation facility also showed strong trends to having a child sustain recovery. HH SES, food security and IYCF practices were not significantly associated with whether a child sustained recovery. Children with a higher MUAC upon discharge from initial MAM treatment in an SFP were more likely to sustain recovery than those with a lower discharge MUAC.
Table 5 Factors associated with children who sustained recovery for 12 months following recovery from moderate acute malnutrition in multivariate logistic regression modelFootnote * (Adjusted odds ratios and 95 % confidence intervals)
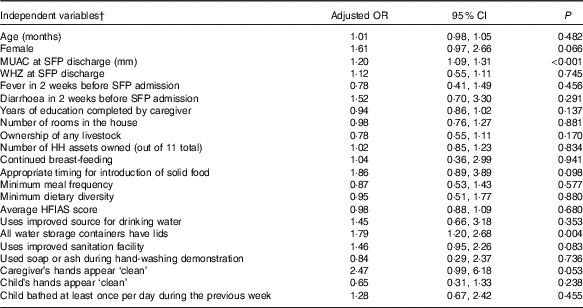
MUAC, mid-upper-arm circumference; SFP, supplementary feeding programme; WHZ, weight-for-height z score; HH, Household; HFIAS, Household Food Insecurity Access Scale.
* Logistic regression model included cluster robust standard errors to account for clustering at the health clinic level.
Discussion
These results show that improved conditions associated with WASH, including HH having fitted lids on water storage containers, caregivers having clean hands and HH using an improved sanitation facility, were each associated with sustained recovery after MAM. Interestingly, factors related to SES, food security and IYCF practices were not significant. Given these results, we hypothesise that improved WASH conditions in tandem with management of MAM through supplemental feeding programmes have the possibility to decrease relapse following recovery from MAM.
Although many studies have been conducted regarding the impact of WASH on child health outcomes, such as diarrhoea( Reference Pruss-Ustun, Bartram and Clasen 37 ) and helminth infection( Reference Strunz, Addiss and Stocks 38 ), fewer and less rigorous studies have been used regarding nutritional outcomes and show little evidence of impact. A recent Cochrane review identified five cRCT that measured the effect of WASH interventions on nutritional status and found no evidence that WASH interventions have an impact on weight-for-age z score or WHZ and only a small effect on height-for-age z score( Reference Victora, Requejo and Barros 24 , Reference Dangour, Watson and Cumming 39 ). That said, the interventions were short in duration; no study considered the effect of a complete package of WASH interventions; and no study examined the impact on relapse rates following initial recovery from MAM. A prospective, randomised clinical trial that examines the impact of a package of WASH interventions on relapse among children following recovery from MAM or SAM is warranted.
Further studies are needed to generate a better understanding of the potential causal pathways between WASH conditions and sustained recovery from MAM. These include in-depth biological analyses regarding diarrhoeal diseases, intestinal parasite infections and environmental enteric dysfunction (EED) during and following the state of moderate malnutrition. A recent study shows a high proportion of subclinical inflammation in children at the time of MAM diagnosis( Reference Cichon, Fabiansen and Yaméogo 40 ), a likely sign of EED. Diarrhoea and intestinal parasites have been shown to exacerbate acute malnutrition( Reference Caulfield, de Onis and Blössner 41 – Reference O’Lorcain and Holland 44 ) and, if gone untreated at the time of discharge from SFP, could leave children susceptible to relapse. However, results from the larger cRCT, in which this current study was embedded, found that providing a single dose of deworming medication and a 14-d course of Zn supplementation at the time of SFP discharge (as part of a larger package of services) did not improve relapse-free survival curves for 12 months following recovery from MAM( Reference Stobaugh, Bollinger and Adams 3 ).
Although evidence from this study points to the potential for improved HH WASH conditions to reduce relapse rates following recovery from MAM, improved HH WASH condition is likely only one of many factors that must be explored to improve MAM treatment so that recovery is better sustained. For example, our study shows that a higher MUAC at SFP discharge is predictive of sustaining recovery during the following year. Higher WHZ and MUAC at the time of admission and discharge from MAM treatment have also been observed in other studies to be more predictive of sustained recovery than a longer duration of treatment( Reference Chang, Trehan and Wang 1 , Reference Trehan, Banerjee and Murray 7 ). Therefore, the combination of improved WASH condition, as well as improved anthropometrics at the time of SFP discharge, may have a significant impact on reducing relapse rates following SFP.
There are several limitations of the study design to be considered. First, the lack of variability in the results is reflective of a relatively homogeneous study population. The indicators chosen in the study may not have been specific enough to this study population to identify true variations that did exist. Second, although the WASH indicators included in the HH survey are standard indicators commonly used for assessments and programme evaluations, they do not fully capture all aspects of WASH conditions. For example, although a HH may have access to an improved sanitation facility, other members in the community may still practice open defecation, which reduces the positive effects of that individual HH’s use of the improved sanitation facility. Third, the sampling of the study population was based on convenience at the end of the study, as opposed to a random selection throughout the duration of the larger cRCT in which this sub-study was embedded. This may have led to some biases in the selection of the study population.
With the exception of a few WASH indicators, our results show that the vast majority of HH characteristics and caregiving practices in this context did not greatly differ between HH whose children relapsed and those who sustained recovery. In this homogeneous low-income population, factors relating to SES, IYCF practices and food security that are commonly associated with malnutrition( Reference Ma, Zhou and Hu 11 – Reference Humphrey 14 , Reference Moursi, Arimond and Dewey 17 – 20 ) do not distinguish between children who sustain MAM recovery and those who do not. This suggests that what distinguishes those who relapse and those who do not is related less to HH conditions and relatively more to individual determinants of a child’s health and nutrition. Similar findings were also demonstrated in a recent follow-up study of children after recovery from SAM, whereby clinical and anthropometric indicators were predictive of death during the follow-up period, yet social or economic factors were not( Reference Khatoon, Mollah and Choudhury 10 ). Certainly, HH factors are important to the overall well-being of children, and improvements in the environment and caregiving practices should remain goals in public health interventions and development programmes. Yet, on the basis of these results, increasing nutrition-sensitive programming alone is not likely to reduce markedly the large percentage of children experiencing relapse following MAM. Rather, these programmes should be considered an important complement to improved SFP treatment and follow-up protocols that address risk factors of relapse at the level of the individual child.
Acknowledgements
We thank the Malawian nurses at the St. Louis Nutrition Project for their dedication to collecting high-quality data throughout the course of this study.
This publication was supported by US Agency for International Development (USAID) (H. C. S., C. T., K. M. M., I. T., M. J. M., Cooperative Agreement/grant no. AID-OAA-A-12-0005); the Children’s Discovery Institute of Washington University and St. Louis Children’s Hospital (I. T.); and the Office of Food for Peace (FFP) of the Bureau for Democracy, Conflict and Humanitarian Assistance (B. L. R., P. W., I. H. R., Cooperative Agreement no. AID-OAA-C-16-00020). USAID had no role in the design, analysis or writing of this article. The contents are the responsibility of the authors and do not necessarily reflect the views of USAID or the US government, nor does any mention of trade names, commercial products or organisations imply endorsement by USAID or the US government.
H. C. S., B. L. R., P. W., I. H. R., M. J. M. and I. T. formulated the research and designed the study. H. C. S. and I. T. enrolled participants and conducted the study. C. T., K. M. M., M. J. M. and I. T. supervised the implementation of the study and all study protocols. H. C. S. cleaned and analysed the data, wrote the first draft of the manuscript and has primary responsibility for the paper’s final content. All authors edited the manuscript and approved its final contents.
The authors declare that there are no conflicts of interest. This manuscript does not include animal studies, in vitro studies or probiotics.
Supplementary Material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114518000363










