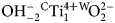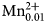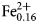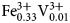A multi-methodological study of the (K,Ca)-variety of the zeolite merlinoite from Fosso Attici, Sacrofano, Italy was carried out on the basis of electron microprobe analysis in wavelength dispersive mode, singlecrystal X-ray diffraction (at 100 K), Raman and infrared spectroscopy. Thechemical formula of the merlinoite from Fosso Attici is (Na0.37K5.69)Σ=6.06(Mg0.01Ca1.93Ba0.40)Σ=2.34(Fe0.023+Al10.55Si21.38)Σ=31.9O64·19.6H2O,compatible with the ideal chemical formula K6Ca2[Al10Si22O64]·20H2O.
Anisotropic structure refinements confirmed the symmetry and the framework model previously reported (space group Immm, a = 14.066(5),b = 14.111(5), c = 9.943(3) Å at 100 K). Refinement converged with four cationic sites and six H2O sites; refined bond distances of the framework tetrahedra suggest a highly disordered Si/Al-distribution. The Raman spectrum of merlinoite (collected between 100and 4000 cm–1) is dominated by a doublet of bands between 496–422 cm–1, assigned to tetrahedral T–O–T symmetric bending modes. T–O–T antisymmetric stretching is also observed; stretching and bending modes of the H2Omolecules are only clearly visible when using a blue laser. The single-crystal near-infrared spectrum shows a very weak band at 6823 cm–1, assigned to the first overtone of the O–H stretching mode, and a band at 5209 cm–1, due to the combination of H2Ostretching and bending modes. Avery broad and convoluted absorption, extending from 3700 to 3000 cm–1 occurs in the H2O stretching region, while the ν2 bending mode of H2O is found at 1649 cm–1. The powder mid-infraredspectrum of merlinoite between 400–1300 cm–1 is dominated by tetrahedral T–O–T symmetric and antisymmetric stretches. Raman and Fourier-transform infrared spectroscopy spectra of merlinoite and phillipsite provide a quick identification tool for these zeolites,which are often confused due to their close similarity.