80 results
Impact of metagenomic next-generation sequencing on clinical decision-making at an academic medical center, a retrospective study, Iowa, 2020–2022
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue 1 / 2024
- Published online by Cambridge University Press:
- 27 March 2024, e39
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Diagnostic stewardship to improve patient outcomes and healthcare-associated infection (HAI) metrics
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 4 / April 2024
- Published online by Cambridge University Press:
- 11 January 2024, pp. 405-411
- Print publication:
- April 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Alcohol-based decontamination of gloved hands: A randomized controlled trial
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 4 / April 2024
- Published online by Cambridge University Press:
- 23 November 2023, pp. 467-473
- Print publication:
- April 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Diagnostic stewardship and the coronavirus disease 2019 (COVID-19) pandemic: Lessons learned for prevention of emerging infectious diseases in acute-care settings
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 3 / March 2024
- Published online by Cambridge University Press:
- 07 November 2023, pp. 277-283
- Print publication:
- March 2024
-
- Article
-
- You have access
- HTML
- Export citation
Improving antimicrobial use through better diagnosis: The relationship between diagnostic stewardship and antimicrobial stewardship
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 12 / December 2023
- Published online by Cambridge University Press:
- 04 September 2023, pp. 1901-1908
- Print publication:
- December 2023
-
- Article
-
- You have access
- HTML
- Export citation
Comparison of virus aerosol concentrations across a face shield worn on a healthcare personnel during a simulated patient cough
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 2 / February 2024
- Published online by Cambridge University Press:
- 23 August 2023, pp. 221-226
- Print publication:
- February 2024
-
- Article
- Export citation
A review of extended coronavirus disease 2019 (COVID-19) isolation duration among inpatients in a tertiary-care hospital—Iowa, 2020–2022
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 1 / January 2024
- Published online by Cambridge University Press:
- 02 August 2023, pp. 110-113
- Print publication:
- January 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Reconsidering the routine use of contact precautions in preventing the transmission of severe acute respiratory coronavirus virus 2 (SARS-CoV-2) in healthcare settings
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 04 May 2023, pp. 1035-1037
- Print publication:
- June 2023
-
- Article
-
- You have access
- HTML
- Export citation
Principles of diagnostic stewardship: A practical guide from the Society for Healthcare Epidemiology of America Diagnostic Stewardship Task Force
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 2 / February 2023
- Published online by Cambridge University Press:
- 14 February 2023, pp. 178-185
- Print publication:
- February 2023
-
- Article
-
- You have access
- HTML
- Export citation
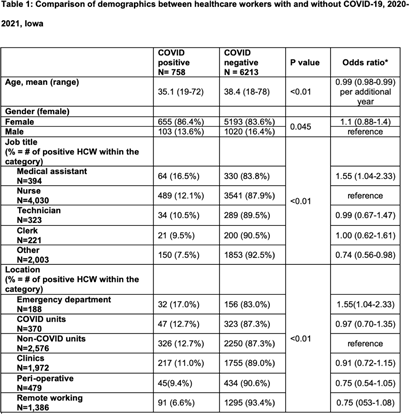
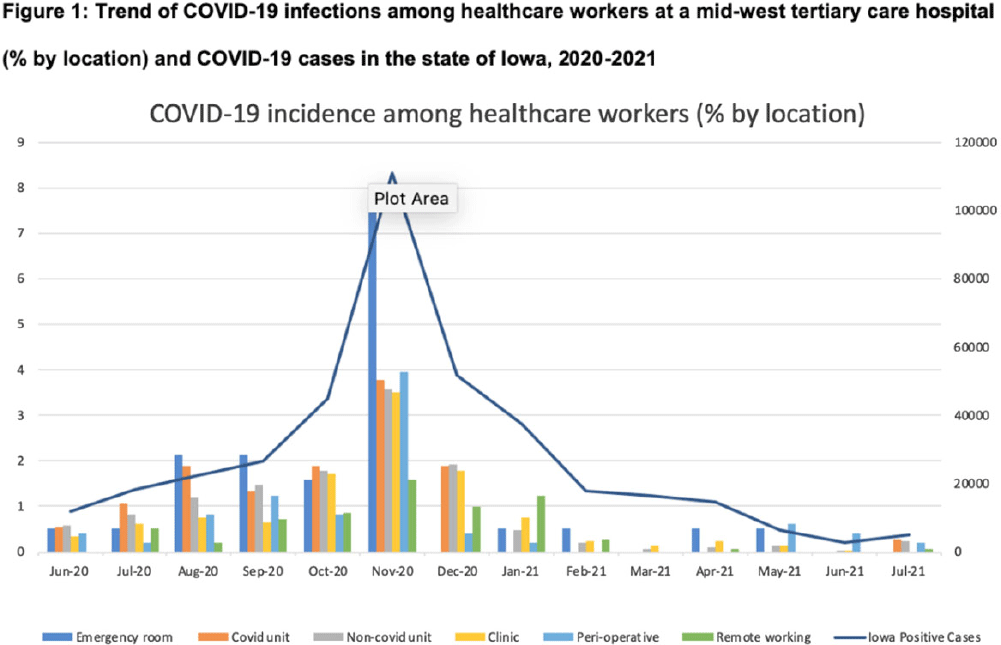
Association between job role and coronavirus disease 2019 (COVID-19) among healthcare personnel, Iowa, 2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 01 December 2022, e188
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Effectiveness and acceptability of intranasal povidone-iodine decolonization among fracture fixation surgery patients to reduce Staphylococcus aureus nasal colonization
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 15 June 2022, pp. 982-984
- Print publication:
- June 2023
-
- Article
- Export citation
Coronavirus disease 2019 (COVID-19) among nonphysician healthcare personnel by work location at a tertiary-care center, Iowa, 2020–2021
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 8 / August 2023
- Published online by Cambridge University Press:
- 02 June 2022, pp. 1351-1354
- Print publication:
- August 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Feasibility and acceptability of intranasal povidone iodine decolonization among orthopedic trauma surgery patients
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s62-s63
-
- Article
-
- You have access
- Open access
- Export citation
COVID-19 incidence among nonphysician healthcare workers at a tertiary care center–Iowa, 2020–2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s6-s7
-
- Article
-
- You have access
- Open access
- Export citation
Blood-culture ordering practices in patients with a central line at an academic medical center–Iowa, 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s30
-
- Article
-
- You have access
- Open access
- Export citation
Blood culture practices in patients with a central line at an academic medical center—Iowa, 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 13 April 2022, e64
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
The long-term effectiveness of coronavirus disease 2019 (COVID-19) vaccines: A systematic literature review and meta-analysis
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 14 February 2022, e22
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
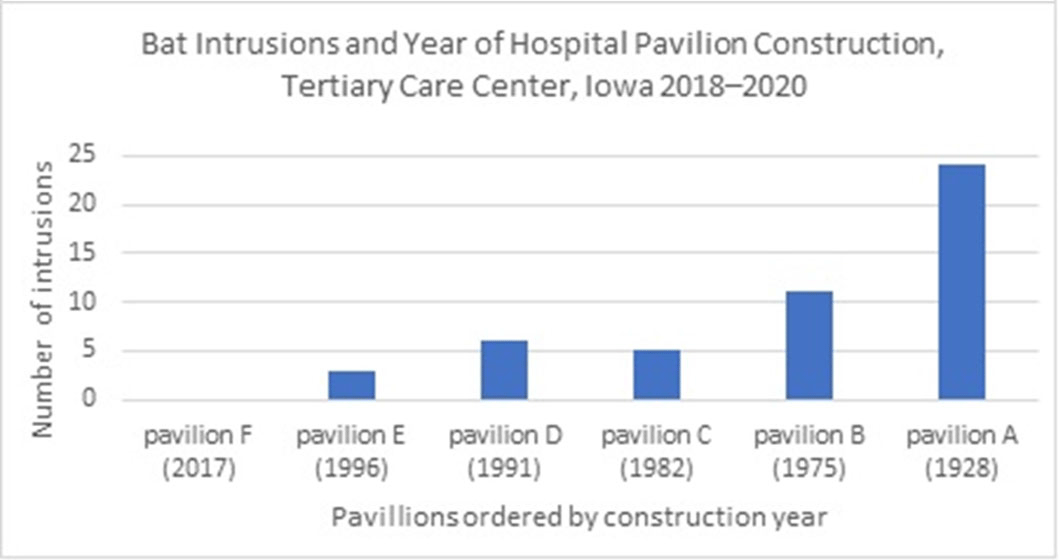
Bat intrusions at a tertiary care center in Iowa, 2018–2020
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 12 / December 2022
- Published online by Cambridge University Press:
- 16 August 2021, pp. 1948-1950
- Print publication:
- December 2022
-
- Article
- Export citation
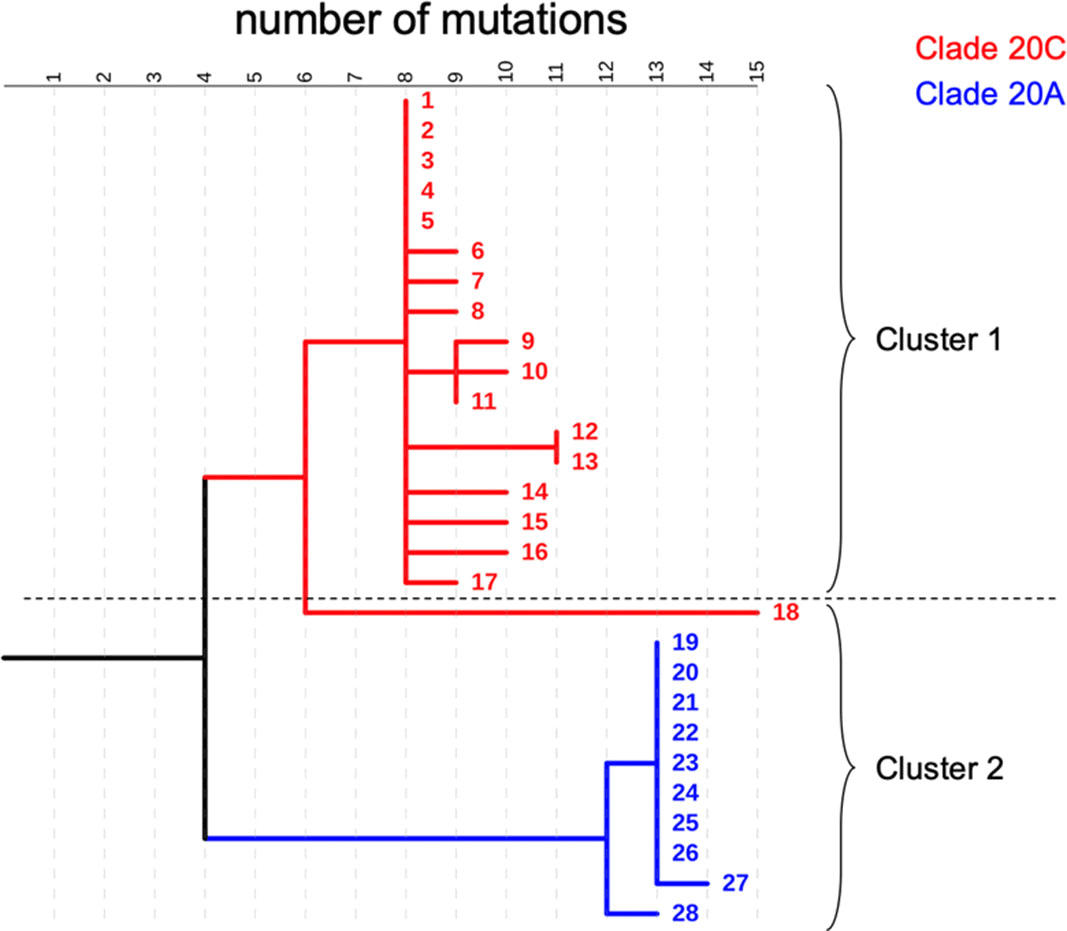
Molecular Epidemiology of Large COVID-19 Clusters at an Academic Medical Center, March–October 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s10-s11
-
- Article
-
- You have access
- Open access
- Export citation
Bat Intrusions at a Tertiary Care Center, Iowa 2018–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s16
-
- Article
-
- You have access
- Open access
- Export citation