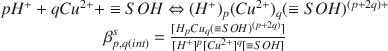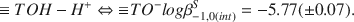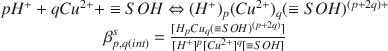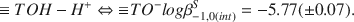The interaction of H+- and Cu2+-ions with Ca-montmorillonite was investigated in 0.1 mol/dm3 solutions of Ca(CIO4)2 at 298.2 K by Potentiometrie titrations using both glass electrodes (for H+) and ion specific electrodes (for Cu2+ ). The experimental data were interpreted on the basis of the surface complexation model. The calculations were performed with the least-squares program FITEQL (Westall, 1982) using the constant capacitance approximation. The best fit was obtained with a set of equilibria of the general form