Original Articles
Effect of a disinfectant udder wash and a post-milking teat dip on the bacterial population of the teat end and on the rate of new intramammary infection
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 253-258
-
- Article
- Export citation
Studies on the motility of smooth muscles of the teats in lactating cows
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 259-275
-
- Article
- Export citation
Effect of oil-enriched diets on the milk yield and composition, and on the composition and physical properties of the milk fat, of dairy cows receiving a basal ration of grass silage
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 277-285
-
- Article
- Export citation
Effect on milk lipolysis of restricted feeding with and without supplementation with protected rape seed oil
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 287-294
-
- Article
- Export citation
Detection and quantification of non-milk fat in mixtures of milk and non-milk fats
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 295-303
-
- Article
- Export citation
Characterization and response to mitogens of mammary lymphocytes from the bovine dry-period secretion
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 305-311
-
- Article
- Export citation
Heat-induced association of β-lactoglobulin and casein micelles
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 313-325
-
- Article
- Export citation
Effect of concentration by ultrafiltration on the heat stability of skim-milk
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 327-335
-
- Article
- Export citation
Factors influencing the stability of milk dialysate
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 337-342
-
- Article
- Export citation
Light scattering investigation of the stability of bovine casein micelles to dilution
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 343-350
-
- Article
- Export citation
Determination of the proportion of κ-casein hydrolysed by rennet on coagulation of skim-milk
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 351-358
-
- Article
- Export citation
The effect of the chemical structure of additives on the coagulation of casein micelle suspensions by rennet
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 359-369
-
- Article
- Export citation
Influence of storage of milk on casein distribution between the micellar and soluble phases and its relationship to cheese-making parameters
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 371-382
-
- Article
- Export citation
Factors influencing casein distribution in cold-stored milk and their effects on cheese-making parameters
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 383-391
-
- Article
- Export citation
Influence of elevated somatic cell count on casein distribution and cheese-making
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 393-400
-
- Article
- Export citation
Multiple modification/restriction systems in lactic streptococci and their significance in defining a phage-typing system
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 401-409
-
- Article
- Export citation
Effect of ripening cream with selected lactic acid bacteria on the quality of ghee
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 411-415
-
- Article
- Export citation
Short Communications
Use of a pH-stat to indicate the end-point in titration of calcium plus magnesium in milk and whey
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 417-419
-
- Article
- Export citation
Specific action of milk-clotting enzymes on water buffalo caseins: I. Effect of chymosin on β-casein
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 421-426
-
- Article
- Export citation
Reviews of the Progress of Dairy Science
Hormones in milk: their physiological significance and value as diagnostic aids
-
- Published online by Cambridge University Press:
- 01 June 2009, pp. 427-449
-
- Article
- Export citation

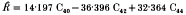
 of 0·7088 where C
of 0·7088 where C