Epidemiological studies(Reference Brat, George and Bellamy1) have shown an association between the intake of plant foods, particularly fruit and vegetables, and a decrease in CHD and other CVD. Similarly, increased consumption of green tea is associated with a reduced risk of CVD(Reference Grassi, Aggio and Onori2), while rat studies indicate that consumption of green tea reduces the increased blood pressure induced by fructose feeding(Reference Wu, Juan and Hwang3). These beneficial effects of plant-derived foods and beverages on cardiovascular health are suggested to be due to the presence of polyphenols. Previous studies have shown that polyphenols may well be beneficial to cardiovascular health(Reference Nicholson, Tucker and Brameld4), particularly those present in red wines (e.g. resveratrol and quercetin), which are thought to account for the ‘French paradox’.
The mechanism of action for the beneficial effects of polyphenols on CVD may be due to their antioxidant activity. However, many polyphenols are metabolised in the body, and these metabolites have much lower antioxidant capacities than their parent compounds, suggesting that antioxidant activity may not be their only mechanism of action(Reference Scalbert, Manach and Morand5). Polyphenols thought to provide health benefits include resveratrol, quercetin, epicatechin gallate (ECG) and epigallocatechin gallate (EGCG), which belong to different classes of polyphenol and may therefore have differing effects dependent upon their chemical structures. Tea and polyphenols present in teas (particularly ECG and EGCG) have previously been reported to have anti-carcinogenic, anti-proliferative and anti-angiogenic properties(Reference Tang, Chiang and Shih6), as well as having effects on gene expression. These anti-angiogenic properties may relate to local expression of vascular endothelial growth factor (VEGF), a potent stimulator of angiogenesis. Red wine polyphenols have been shown to reduce expression of the vasoconstrictor, endothelin-1 (ET-1) in cultured vascular cells(Reference Corder, Douthwaite and Lees7–Reference Liu, Chen and Chan9) and to increase expression of endothelial NO synthase (eNOS), the enzyme responsible for the production of the vasodilator NO(Reference Wallerath, Deckert and Ternes10, Reference Wallerath, Li and Godtel-Ambrust11). However, cell culture-based studies investigating these effects have tended to use supraphysiological concentrations of individual polyphenols. Maximum concentrations of polyphenols in the blood after consumption of polyphenol-rich foods or beverages tend to be about 0·1–1 μm(Reference Scalbert and Williamson12), whereas previous in vitro studies have tended to treat cells with much higher concentrations (2–100 μm).
Stress is known to elevate the expression of certain genes in several cell types including human umbilical vein endothelial cells (HUVEC) and smooth muscle cells(Reference Liu, Chen and Chan9). Different methods to induce stress or to elevate gene expression include stretching cells cultured on a flexible membrane (cyclic strain)(Reference Liu, Chen and Chan9) or treatment with H2O2(Reference Liu, Chen and Chan9) or angiotensin II(Reference Chao, Juan and Liu8). Previous studies have shown that supraphysiological concentrations (10–100 μm) of resveratrol blocked the stimulatory effects(Reference Liu, Chen and Chan9) of H2O2, cyclic strain(Reference Liu, Chen and Chan9) and angiotensin II treatment(Reference Chao, Juan and Liu8) on ET-1 mRNA expression.
The present study investigated the effects of physiological concentrations of different classes of dietary polyphenol on the expression of genes important in cardiovascular health (eNOS, ET-1 and VEGF) in cultured vascular endothelial cells (HUVEC) under stress-free and oxidative stress conditions (i.e. with or without H2O2).
Materials and methods
Materials and chemicals
Unless stated otherwise, all chemicals (including polyphenols) were obtained from Sigma-Aldrich (St Louis, MO, USA). The endothelin ELISA kit was obtained from Biomedica (Vienna, Austria) via Oxford Biosystems (Oxford, UK).
Preparation of individual polyphenols
A 100 μm stock solution of each polyphenol was prepared in PBS and diluted further with PBS before being added to culture media.
Cell culture
HUVEC were purchased from Clonetics (Cambrex, Inc., Walkersville, MD, USA) as a frozen stock (passage 3), defrosted and cultured in endothelial basal medium 2 growth media plus bullet kit (Cambrex, Inc.) containing 2 % fetal calf serum, antibiotics and growth factor supplements at 37°C and 5 % CO2. The cells were passaged three times to provide a pool of cells to be used for experiments. Experiments were performed in six-well plates (Corning, Inc., Corning, NY, USA) when the cells were 80–85 % confluent, at which stage the endothelial basal medium 2 growth media (2 % foetal calf serum, with growth factors) was replaced with endothelial basal medium 2 control media (0·8 % foetal calf serum, no growth factors) and incubated for 24 h (37°C, 5 % CO2). Treatments were prepared in endothelial basal medium 2 containing 0·8 % fetal calf serum and antibiotics but no other supplements. This consisted of 70 % media and 30 % polyphenol and/or H2O2 in PBS, with PBS alone used as control. Final concentrations of individual polyphenols in the culture media ranged from 0·001 to 1·0 μm, while concentrations of H2O2 ranged from 6·25 to 50 μm. After 24 h incubation with treatments, culture media were stored at − 40°C for measurement of endothelin secretion using an ELISA kit and total RNA was extracted from the cells using TRIzol (Invitrogen, Carlsbad, CA, USA), both according to the manufacturer's instructions.
Quantitative real-time PCR
Total RNA was DNase-treated before quantifying RNA yields using a Nanodrop spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA) and checking RNA integrity by running on 1 % agarose gels. Complementary DNA was synthesised using Moloney murine leukaemia virus reverse transcriptase and buffer (Promega, Fitchburg, WI, USA). Gene- and species-specific primers and probes for real-time PCR (Table 1) were all purchased from Applied Biosystems (Foster City, CA, USA). Real-time PCR was carried out on triplicate complementary DNA samples from control or treated HUVEC following the standard curve method on the Roche Lightcycler 480 diagnostic system using 384-well plates and TaqMan Universal PCR master mix (Applied Biosystems). As there were no significant treatment effects on glyceraldehyde 3-phosphate dehydrogenase mRNA (Tables 2–6), glyceraldehyde 3-phosphate dehydrogenase was selected as the housekeeping gene to allow quantification of the expression of eNOS, ET-1 and VEGF.
Table 1 Real-time PCR primers and probes
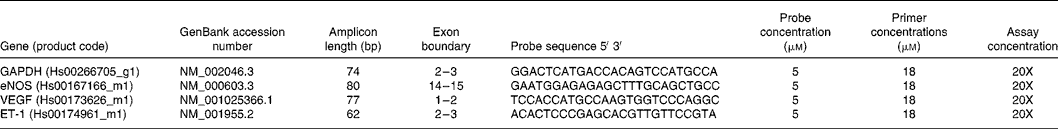
GAPDH, glyceraldehyde 3-phosphate dehydrogenase; eNOS, endothelial NO synthase; VEGF, vascular endothelial growth factor; ET-1, endothelin-1.
Table 2 Dose-dependent effects of hydrogen peroxide on human umbilical vein endothelial cells gene expression
(Mean values with their standard error of the differences)

GAPDH, glyceraldehyde 3-phosphate dehydrogenase; eNOS, endothelial NO synthase; VEGF, vascular endothelial growth factor; ET-1, endothelin-1.
* Gene expression normalised to GAPDH.
† Mean values were significantly different from those of the control group (P < 0·05, post hoc Dunnett's test).
Table 3 Dose-dependent effects of resveratrol (R) in the absence or presence of hydrogen peroxide (50 μm) on human umbilical vein endothelial cells gene expression
(Mean values with their standard error of the differences)
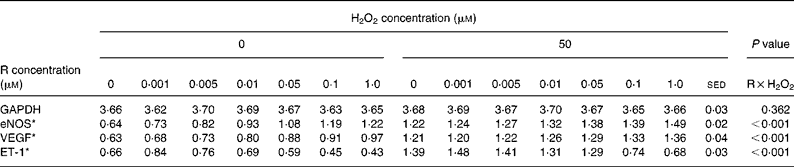
GAPDH, glyceraldehyde 3-phosphate dehydrogenase; eNOS, endothelial NO synthase; VEGF, vascular endothelial growth factor; ET-1, endothelin-1.
* Gene expression normalised to GAPDH.
Table 4 Dose-dependent effects of quercetin (Q) in the absence or presence of hydrogen peroxide (50 μm) on human umbilical vein endothelial cells gene expression
(Mean values with their standard error of the differences)
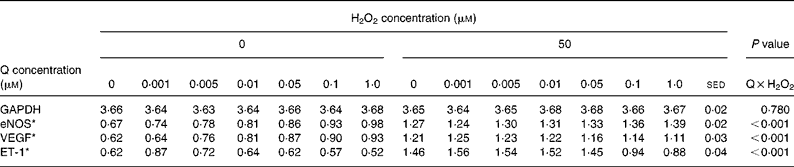
GAPDH, glyceraldehyde 3-phosphate dehydrogenase; eNOS, endothelial NO synthase; VEGF, vascular endothelial growth factor; ET-1, endothelin-1.
* Gene expression normalised to GAPDH.
Table 5 Effects of polyphenols (Poly) with differing structures (all 0·1 μm) in the absence or presence of hydrogen peroxide (50 μm) on human umbilical vein endothelial cells gene expression
(Mean values with their standard error of the differences)
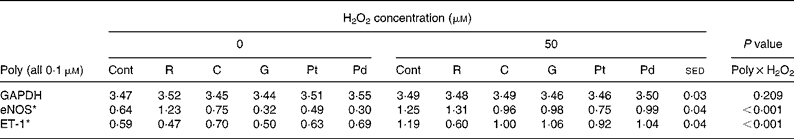
Cont, control; R, resveratrol; C, caffeic acid; G, genistein; Pt, phloretin; Pd, phloridzin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; eNOS, endothelial NO synthase; ET-1, endothelin-1.
* Gene expression normalised to GAPDH.
Table 6 Effects of individual polyphenols (Poly; all 0·1 μm) in the absence or presence of hydrogen peroxide (50 μm) on human umbilical vein endothelial cells gene expression
(Mean values with their standard error of the differences)
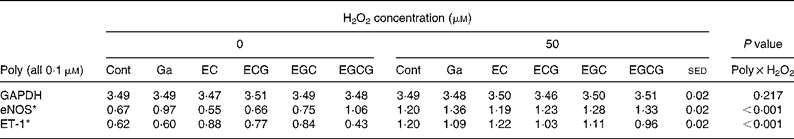
Cont, control; Ga, gallic acid; EC, epicatechin; ECG, epicatechin gallate; EGC, epigallocatechin; EGCG, epigallocatechin gallate; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; eNOS, endothelial NO synthase; ET-1, endothelin-1.
* Gene expression normalised to GAPDH.
Statistical analyses
Relative mRNA expression data are expressed as means, with the standard error of the differences of the means shown in tables or the standard deviations shown in figures. All the data were analysed by one- or two-way ANOVA using Genstat version 9.1 (2007). P < 0·05 was considered as statistically significant. A post hoc Dunnett's test was used to identify significant differences between the control and treatment groups, but only when there were no significant interactions.
Results
There were no effects (P>0·1) of any of the treatments on the expression of glyceraldehyde 3-phosphate dehydrogenase (Tables 2–6), so glyceraldehyde 3-phosphate dehydrogenase was used as the control gene to normalise the expression of eNOS, ET-1 and VEGF mRNA.
Effects of hydrogen peroxide on human umbilical vein endothelial cells gene expression
The dose-dependent effects of H2O2 (6·25–50 μm) on the expression of eNOS, ET-1 and VEGF mRNA were determined. Table 2 shows a dose-dependent increase in the expression of all the three genes with H2O2 treatment with maximum increases of about 220 % (relative to controls) observed with 50 μm-H2O2 (P < 0·001 for all).
Dose–response effects of resveratrol and quercetin on human umbilical vein endothelial cells gene expression
The dose-dependent effects of resveratrol and quercetin (0·001–1·0 μm) on the expression of eNOS, ET-1 and VEGF mRNA were investigated in the absence or presence of H2O2 (50 μm). Both the polyphenols had similar effects, but resveratrol was more potent than quercetin at equimolar concentrations (Tables 3 and 4).
As before, addition of 50 μm-H2O2 increased the eNOS, ET-1 and VEGF mRNA expression (Tables 3 and 4), but the effects of the polyphenols were gene specific. Resveratrol induced dose-dependent increases in the expression of eNOS and VEGF mRNA in the absence of H2O2 (Table 2), but the two treatments were not additive (P < 0·001 for H2O2 × resveratrol interactions). In contrast, resveratrol (at 0·1 and 1 μm) reduced the ET-1 mRNA expression in the absence of H2O2 (Table 3) and reduced or blocked the effect of 50 μm-H2O2 (P < 0·001 for both H2O2 × resveratrol interaction).
Similarly, quercetin induced dose-dependent increases in the expression of eNOS and VEGF mRNA in the absence of H2O2 (Table 4), but there was no additive effect with H2O2 on eNOS mRNA expression, while higher concentrations of quercetin reduced VEGF expression induced by 50 μm-H2O2 (P < 0·001 for both H2O2 × quercetin interactions). Like resveratrol, quercetin (at 0·1 and 1 μm) reduced ET-1 mRNA expression compared with controls (Table 4) and reduced or blocked the effect of 50 μm-H2O2 (P < 0·001 for both H2O2 × quercetin interaction).
Effects of resveratrol, quercetin and hydrogen peroxide on endothelin secretion
Since both quercetin and resveratrol (at 0·1 μm) reduced basal- and H2O2 (50 μm)-induced expression of ET-1 mRNA, their effects on the secretion of endothelin by HUVEC were examined. The effects were very similar to the effects on mRNA expression: in the absence of H2O2, both quercetin and resveratrol (0·1 μm) reduced the secretion of endothelin relative to controls (Fig. 1). In the absence of quercetin and resveratrol, H2O2 (50 μm) significantly increased the secretion of endothelin, but this was blocked by the addition of either resveratrol or quercetin (0·1 μm), with resveratrol being slightly more potent (P < 0·001, H2O2 × polyphenol interaction).

Fig. 1 Effects of resveratrol and quercetin (0·1 μm) in the presence or absence of hydrogen peroxide(50 μm) on secretion of endothelin by cultured human umbilical vein endothelial cells. Means and standard deviations (n 3) are shown. □, Minus hydrogen peroxide (50 μm); ▧, plus hydrogen peroxide (50 μm).
Effects of other dietary polyphenols on human umbilical vein endothelial cells gene expression
In order to investigate structure–function relationships in the observed effects, a variety of polyphenols (caffeic acid, genistein, phloretin, phloridzin, gallic acid, epicatechin, ECG, epigallocatechin and EGCG) were compared with resveratrol (all at 0·1 μm) in the absence or presence of 50 μm-H2O2 (Tables 5 and 6). These studies were carried out in two batches (each table represents a batch) but were performed at exactly the same time, therefore allowing direct comparisons. Although previous studies had shown a significant effect of resveratrol and H2O2 on VEGF expression, the effects of the different polyphenols on VEGF expression were not examined here. As in previous studies, significant interactions (P < 0·001) were observed between polyphenol and H2O2 treatments on both the eNOS and ET-1 mRNA expression. As before, H2O2 treatment (50 μm) increased eNOS and ET-1 mRNA expression relative to control treatment (Tables 5 and 6) and resveratrol (0·1 μm) increased eNOS mRNA expression in the absence of H2O2, but there was no additive effect of resveratrol and H2O2 (Table 5). Similar to resveratrol, gallic acid and EGCG (Table 6) had stimulatory effects on eNOS, with both appearing to be additive to the effect of H2O2. The effects of the catechins on eNOS mRNA appeared to increase in the order epicatechin < ECG < epigallocatechin < EGCG (Table 6), which might indicate a structure–function relationship, although only epigallocatechin and EGCG appeared to have significant effects. In contrast, genistein, phloretin and phloridzin (Table 5) all reduced eNOS expression in the presence and absence of H2O2 (50 μm), opposite to the effect of resveratrol. Both resveratrol (Table 5) and EGCG (Table 6) reduced ET-1 expression compared with controls and blocked the stimulatory effect of H2O2 on ET-1 mRNA. None of the other polyphenols studied appeared to have significant effects on ET-1 expression, although there was some variability in expression.
Discussion
H2O2 alone was found to significantly increase the expression of eNOS, VEGF and ET-1 mRNA in a dose-dependent manner (6·25–50 μm) after 24 h treatment. Previous studies have shown similar effects, with 25 μm-H2O2 increasing ET-1 expression in HUVEC(Reference Liu, Chen and Chan9) and 100 μm-H2O2 increasing eNOS expression in bovine aortic endothelial cells(Reference Drummond, Cai and Davis13).
Quercetin and resveratrol were found to significantly increase the expression of eNOS in a dose-dependent manner after 24 h. This agrees with previous studies using supraphysiological concentrations of resveratrol, where 33 μm and 10–100 μm up-regulated eNOS mRNA expression in HUVEC and HUVEC-derived EA.hy 926 cells, respectively(Reference Wallerath, Deckert and Ternes10). In the present study, the combination of 50 μm-H2O2 and quercetin or resveratrol (0·001–1·0 μm) increased eNOS expression, but the two effects were not completely additive, suggesting that the effects are either via similar mechanisms or have simply reached a maximum level. Although eNOS protein or activity levels were not measured, a recent study(Reference Appeldoorn, Venema and Peters14) showed that both resveratrol and quercetin (at 100 μm) increased NO synthesis by HUVEC-derived EA.hy 926 cells, indicating that the effects on mRNA expression described here are likely to be matched at the protein level.
In contrast, treatment of HUVEC with either quercetin or resveratrol (at 0·1 or 1·0 μm) resulted in decreased expression of ET-1 mRNA, particularly in the presence of 50 μm-H2O2. These findings are consistent with previous studies in HUVEC showing that resveratrol (100 μm) inhibited the expression of ET-1 mRNA induced by H2O2 (25 μm) or cyclic strain(Reference Liu, Chen and Chan9). Similarly, resveratrol (at 1–100 μm) reduced ET-1 mRNA expression and gene promoter activity induced by angiotensin II in rat aorta smooth muscle cells(Reference Chao, Juan and Liu8). Therefore, resveratrol's ability to block stress-induced ET-1 mRNA expression is not specific to treatment with H2O2, but importantly the present studies demonstrate the effects at physiologically relevant concentrations.
As well as increasing ET-1 mRNA expression, H2O2 (50 μm) also significantly increased secretion of endothelin protein into the media. A similar effect on endothelin protein levels was observed in human aortic vascular smooth muscle cells treated with 200 μm-H2O2 for 8 h(Reference Ruef, Moser and Kubler15). Quercetin and resveratrol (0·1 μm) significantly reduced the secretion of endothelin and blocked the stimulatory effects of H2O2 (50 μm); again in agreement with Ruef et al. (Reference Ruef, Moser and Kubler15), who showed that supraphysiological concentrations of resveratrol and quercetin (100 and 50 μm, respectively) blocked the effect of H2O2 (200 μm) on ET-1 protein content in human aortic vascular smooth muscle cells.
Studies on the effects of equimolar concentrations of resveratrol and nine other dietary polyphenols showed that resveratrol, gallic acid and EGCG all increased eNOS mRNA expression in the presence and absence of H2O2 (50 μm), but only resveratrol and EGCG decreased ET-1 expression in the absence and presence of H2O2 (50 μm). In contrast, genistein, phloridzin and phloretin reduced the expression of eNOS, particularly in the absence of H2O2 (50 μm) but had little or no effect on ET-1 mRNA expression. The stimulatory effects of gallic acid on eNOS mRNA expression agree with a previous study(Reference Wallerath, Li and Godtel-Ambrust11) showing that 1 μm-gallic acid increased eNOS expression in HUVEC-derived EA.hy 926 cells, although another study(Reference Appeldoorn, Venema and Peters14) showed no effect of gallic acid on NO synthesis by EA.hy 926 cells. Interestingly, Appeldoorn et al. (Reference Appeldoorn, Venema and Peters14) also demonstrated the stimulatory effects of resveratrol, quercetin, EGCG and ECG (all at 100 μm) on NO synthesis, with no effects of epicatechin, epigallocatechin, phloretin, caffeic acid and a number of other polyphenols. The stimulatory effects of resveratrol, quercetin and EGCG were matched by increases in eNOS mRNA expression(Reference Appeldoorn, Venema and Peters14), whereas the effect of ECG on eNOS mRNA expression was not statistically significant, agreeing with the data described here using lower concentrations of individual polyphenols. Although the observed inhibitory effects of phloretin on eNOS mRNA expression contrast with its lack of effect on NO synthesis(Reference Appeldoorn, Venema and Peters14), inhibitory effects on NO synthesis would be difficult to measure in the culture system used, since the basal level of NO synthesis was already low(Reference Appeldoorn, Venema and Peters14). The inhibitory effects of genistein on eNOS mRNA expression in HUVEC described here are in contrast to a previous study(Reference Si and Liu16), where genistein increased the expression of eNOS mRNA and protein (at 1–10 and 0·1–10 μm, respectively) in human aortic endothelial cells, possibly through activation of eNOS transcription.
Recently, Appeldoorn et al. (Reference Appeldoorn, Venema and Peters14) demonstrated low recovery ( < 50 %) of several polyphenols, including quercetin, from the culture medium after 24 h incubation, suggesting that some polyphenols may be unstable or being metabolised in the culture system. Using HPLC, we observed similar losses of quercetin in cell culture medium (without cells) during incubation under the same conditions used for gene expression studies (pH 7·4, 37°C, 100 % humidity) with an apparent half-life of approximately 6 h. Hence, it is unclear whether the observed effects are due to quercetin per se or a breakdown product, but the data are still physiologically relevant since the conditions used for cell culture would match those encountered when quercetin is absorbed from the gut into the blood stream.
In conclusion, resveratrol and quercetin increased eNOS mRNA expression and decreased ET-1 mRNA expression in HUVEC in a dose-dependent manner in the absence and presence of H2O2. Similar to their effects on ET-1 mRNA expression, resveratrol and quercetin decreased endothelin secretion, again in the absence and presence of H2O2 (50 μm). Of the nine other polyphenols tested, only EGCG had similar effects to resveratrol and quercetin on both eNOS and ET-1 mRNA expression but to a lesser extent than resveratrol at an equimolar concentration (0·1 μm). Whether these effects are dependent upon structure and/or antioxidant capacities is not clear, although the three active polyphenols (resveratrol, quercetin and EGCG) had the highest antioxidant capacities as measured by ferric reducing antioxidant power assay (data not shown). The observed effects on gene expression would be expected to result in a potent vasodilation and thereby reduced blood pressure. Indeed, quercetin is suggested to reduce diastolic blood pressure in hypertensive human subjects(Reference Edwards, Lyon and Litwin17); although closer inspection indicates that the statistical significance is due to the quercetin group having a higher baseline blood pressure but reaching a similar endpoint blood pressure to controls(Reference Edwards, Lyon and Litwin17). Further in vivo studies investigating the effects of individual polyphenols on blood pressure in human subjects or animals are therefore required.
Acknowledgements
S. K. N. was funded by a Biotechnology and Biological Sciences Research Council (BBSRC) committee PhD studentship. J. M. B., G. A. T. and S. K. N. planned the work; S. K. N. carried out the laboratory analyses; S. K. N. and J. M. B. statistically analysed the data; J. M. B., S. K. N. and G. A. T. wrote the manuscript. The authors have no conflict of interest to declare.











