Skeletal muscle mass (SMM) increases up to a peak in the fourth decade of life, after which it gradually declines( Reference Devries and Phillips 1 ). The age-related decrease in SMM is associated with poor health consequences like weakness, frailty( Reference Morley, Anker and von Haehling 2 ) and mortality( Reference Chuang, Chang and Lee 3 ), and is an important criterion for sarcopenia, the progressive and generalised loss of SMM and muscle strength or performance( Reference Cruz-Jentoft, Baeyens and Bauer 4 ). The prevalence of sarcopenia has reached an epidemic proportion and costed seriously in terms of both healthcare and human life. In the USA, it is estimated that cost-related health consequences of sarcopenia is over $18·5 billion in 2000( Reference Janssen, Shepard and Katzmarzyk 5 ). The prevalence of sarcopenia in older community-dwelling Taiwan adults aged 65 years and above is 3·9–7·3 % according to different definitions of sarcopenia( Reference Wu, Lin and Hsiung 6 ). Therefore, identification of modifiable risk factors for SMM decline is important to allow more effective prevention of this trend.
The Mediterranean diet (MD) is characterised by high consumption of olive oil, fruit, vegetables, legumes, nuts and cereals, low intakes of SFA and red meat, and moderate intake of alcohol( Reference Sofi, Cesari and Abbate 7 ). A large number of studies have showed the MD as a model of healthy eating due to its benefits to the prevention of various chronic diseases, such as CVD( Reference Sofi, Abbate and Gensini 8 ), Alzheimer’s dementia( Reference Scarmeas, Stern and Tang 9 ), cognitive decline( Reference Scarmeas, Stern and Mayeux 10 ), stroke, depression( Reference Psaltopoulou, Sergentanis and Panagiotakos 11 ) and cancer( Reference Couto, Boffetta and Lagiou 12 – Reference Buckland, Agudo and Lujan 15 ). A few studies have also shown favourable associations between the main components of the MD and the occurrence of sarcopenia, such as moderate consumption of ethanol( Reference Steffl, Bohannon and Petr 16 ), lower intake of SFA( Reference Liang, Oest and Jones 17 ) and higher intake of fish oil( Reference Smith, Julliand and Reeds 18 ) and fruits and vegetables( Reference Kim, Lee and Kye 19 ). Previous studies have reported that closer adherence to the MD is associated with lower concentration of inflammation markers( Reference Chrysohoou, Panagiotakos and Pitsavos 20 ) and lower oxidative stress( Reference Pastori, Carnevale and Bartimoccia 21 ), from which it might be assumed that there is a positive association between the MD pattern and SMM.
To date, favourable associations between the MD pattern (or score) and with the measures related to SMM were observed in two cross-sectional studies in UK females( Reference Kelaiditi, Jennings and Steves 22 ) and in German females (not males)( Reference Nikolov, Spira and Aleksandrova 23 ), but not in another one in Iranian females( Reference Hashemi, Motlagh and Heshmat 24 ). One prospective study showed no significant cross-sectional nor longitudinal association between the MD score (MDS) and sarcopenia in Hong Kong older adults( Reference Chan, Leung and Woo 25 ). Among these studies, percent fat-free mass was used in the UK study( Reference Kelaiditi, Jennings and Steves 22 ), whereas a ratio of appendicular lean mass:BMI was used in the German study( Reference Nikolov, Spira and Aleksandrova 23 ), and the Iranian study only analysed a posteriori pattern (a MD-like pattern) rather than the MDS. Therefore, the association between MDS and SMM remains speculative. In this cross-sectional study, we examined the hypothesis that higher alternate Mediterranean diet score (aMDS) is associated with greater SMM in a middle-aged and older population.
Methods
Study population
This study was based on cross-sectional data from the Guangzhou Nutrition and Health Study (GNHS), a community-based prospective cohort study designed to investigate the determinants of major health problems. The cohort study recruited a total of 3169 apparently healthy participants aged 40–75 years old by advertisement and referral between 2008 and 2010 in Guangzhou, China. In the first follow-up survey conducted between 2011 and 2013, 649 participants dropped out and another 879 new participants were recruited in the same way in 2013. Information on habitual diet and covariates were collected in both 2008–2010 and 2011–2013, and SMM was measured by dual energy X-ray absorptiometry (DXA) during the 2011–2013 survey. Details of the study population have been described previously( Reference Zhang, He and Liu 26 ). Overall, 110 individuals were excluded due to (I) core data missing (sixty-seven subjects); (II) a history of malignancy (thirty subjects), hepatic cirrhosis (three subjects), and renal insufficiency (six subjects); (III) extreme energy intake( Reference Zhang, He and Liu 26 ) (three subjects); and (IV) extreme BMI (one subject). Finally, the study included 3289 participants with at least cross-sectional diet and SMM data collected during 2011–2013. All of the participants provided written informed consent. The study was approved by Sun Yat-Sen University’s School of Public Health Ethics Committee and was conducted according to the Declaration of Helsinki.
Data collection
Information about socio-demographic characteristics (e.g. age, sex, household income, education,), lifestyle (physical activity, smoking and drinking status), use of oral oestrogen and years since menopause (YSM) for females, and personal and family history of chronic diseases and medications was collected by means of face-to-face interviews using a structured questionnaire. Physical activity was specified as activities of daily life, work time and leisure time (excluding sleeping and sitting) and was calculated as the metabolic equivalent (h/d) as described previously( Reference Zhang, Deng and He 27 ). Height and weight were measured with the participant in a standing position and wearing light clothes and no shoes.
Assessment of dietary intake
Participants were asked about their food intake at five possible frequencies (never, per year, per month, per week and per day) over the past year using a seventy-nine-item quantitative FFQ. The Chinese Food Composition Table, 2004 was used to calculate the average daily intake of nutrients and total energy( Reference Zeng, Xue and Cao 28 ). The interview methods have been described in detail elsewhere( Reference Zhang, He and Liu 26 ). For the 2520 subjects, who provided dietary data at both baseline and the first follow-up, we calculated the average intake, which is commonly used to assess long-term dietary habits and reduce within-subject variation as did in previous studies( Reference Hu, Stampfer and Rimm 29 , Reference Larsson, Bergkvist and Wolk 30 ); for the 879 new participants, only one set of dietary data was available and used in further analyses.
Calculation of alternate Mediterranean diet score
The traditional MDS was developed by Trichopoulou et al. ( Reference Trichopoulou, Kouris-Blazos and Wahlqvist 31 ) to indicate the adherence to traditional MD in a Greek population, in which eight items were included and classified adherence by assigning 0 to 5 points for each food component: high MUFA:SFA ratio; moderate consumption of ethanol; high consumption of cereals, fruits, vegetables, legumes; low consumption of milk and dairy products, meat and meat products. Fung et al.’s aMDS( Reference Fung, Rexrode and Mantzoros 32 ) included vegetables, fruits, nuts, whole grains, legumes, fish, ratio of MUFA:SFA, red and processed meats and alcohol. The aMDS is calculated by summing the dichotomous points (0 or 1) for the items of higher intakes of whole grain, vegetables (excluding potatoes), fruits, legumes, nuts, fish and ratio of MUFA:SFA, lower red meat according to their sex-specific group medians, and moderate ethanol consumption (5–15 g/d in females and 15–25 g/d in males)( Reference Fung, Rexrode and Mantzoros 32 ) (online Supplementary Table S1). The possible score range for aMDS was 0 to 9, with a higher score representing closer adherence to the MD( Reference Fung, Rexrode and Mantzoros 32 ). As previous studies suggested that SMM might be associated with fish oil( Reference Murphy, Mourtzakis and Chu 33 ) and nut( Reference Jacques, Leblanc and Papineau 34 ), we finally chose to use Fung et al.’s aMDS to evaluate the diet quality in our study.
Muscle mass assessment
During the 2011–2013 survey, the SMM (kg) of the whole body and the arms and legs was measured by DXA (Discovery W; Hologic Inc.) using a default model and analysed with Hologic Discovery software version 3.2. The DXA scanner was calibrated daily. The in vivo coefficients of variation of the duplicated measurements in thirty participants after repositioning were 6·1 % for the whole body, 3·1 % for the limbs, 2·2 % for the arms and 4·0 % for the legs. The skeletal muscle mass index (SMI) was defined as SMM/height2 (in kg/m2)( Reference Cruz-Jentoft, Baeyens and Bauer 4 ), and SMI were calculated for the whole body, four limbs, arms and legs, respectively.
Statistical analysis
Analyses were performed using SPSS (version 17.0; SPSS Inc.), and two-sided P values below 0·05 were considered significant. The participants were divided into three categories (low (0–3), middle (4, 5) and high (6–8)) according to the aMDS. The common characteristics of the participants were presented as frequencies and percentages for categorical variables, and mean values and standard deviations for continuous variables by sex. The t test, χ 2 test and Wilcoxon rank sum test were used to compare the differences between females and males for the continuous, categorical and ordinal variables, respectively.
Multivariate ANCOVA were used to compare the covariate-adjusted mean SMI between the three aMDS categories in females and males separately. Model 1 adjusted for age and physical activity. Model 2 adjusted for age, physical activity, smoking status, passive smoking status, dairy foods, daily energy intake and dietary protein intake in males, and further adjusted for YSM and oral oestrogen use in females based on theoretical relevance and significance in univariate analysis (P<0·2). Furthermore, we carried out age stratified analyses according to the group medians of age in males (62·2 years) and females (59·2 years) using model 2. Bonferroni tests were used for multiple comparisons between the aMDS categories. Our study could achieve a statistical power of 86 % to detect a non-zero contrast of the means of the total body SMI based on the our data in total subjects using PASS 2011( Reference Hintze 35 ).
In sensitive analyses, regression analysis was used to test whether there was a significant linear association when the aMDS was considered as a continuous variable. Moreover, we compared the SMI by dichotomous categories of each component in model 2 to assess which components of the aMDS were responsible for the associations of aMDS. We also performed discriminant classification analysis using Fisher’s linear discriminant function and the calculation of Wilks’ λ to explore the discriminating ability of the individual components of the aMDS. To explore whether the association between aMDS and SMI was modified by age, sex, physical activity or dietary intake of energy and protein, the potential interactions were assessed in model 2.
Results
The characteristics of the study population are shown in Table 1. Analyses included 2225 females and 1064 males. Sex, BMI, energy intake, smoking status, physical activity and food intakes were significantly different across the three aMDS categories.
Table 1 Characteristics of the participants by categories of the alternate Mediterranean diet score (aMDS) (Mean values and standard deviations; numbers and percentages)
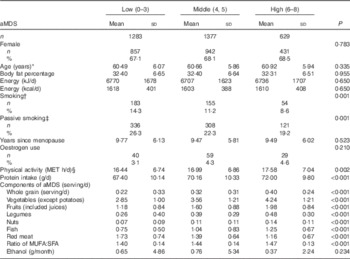
MET, metabolic equivalent.
* Calculated by the time of dual-energy X-ray absorptiometry examination.
† Smoking was defined if they had smoked at least five packages of cigarettes during the last year.
‡ Passive smoking was defined if they had been exposed to others’ tobacco smoke for at least 5 min or one cigarette per day for the past year.
§ Physical activities (MET h/d) included daily activities in occupation, leisure-time and household-chores apart from the lying and sitting.
Comparisons of the covariate-adjusted mean values with their standard errors of the SMI by aMDS categories are presented in Table 2. Generally, there is a favourable association between aMDS and SMI. However, the favourable associations tended to be more pronounced in males (all P trend<0·05) than in females at all measured sites in both models 1 and 2. In males, graded and positive associations were found between the aMDS and SMI at all measured sites (all P trend<0·05). After adjusting for multiple covariates in model 2, the favourable associations tended to be slightly strengthened (all P trend: 0·002–0·013). The SMI means were 2·70 % (whole body), 2·65 % (limbs), 2·50 % (arms) and 2·70 % (legs) higher in the high (v. low) aMDS category (all P<0·05), respectively. In females, the corresponding values were 1·35 % (whole body), 1·05 % (limbs), 0·52 % (arms) and 1·20 % (legs), but not statistically significant except the whole body (P trend=0·03). Age-stratified results showed that the favourable associations tended to be more substantial in younger participants than in the older subjects in both males and females,. However, no statistically significant interaction was observed in males (P: 0·516–0·821) and in females (P: 0·109–0·706) (Table 3).
Table 2 Comparisons of covariate-adjusted skeletal muscle mass index (SMI) by alternate Mediterranean diet score (aMDS) categories in males and females (Mean values with their standard errors)

* P<0·05; ** P<0·01, compared with ‘low category’ by Bonferroni test, α=0·05.
† Percentage difference=(high−low)/low×100 %.
‡ P value for linear trend.
§ Model 1: controlled for age and physical activity.
|| Model 2: controlled for age, physical activity, smoking status, passive smoking status, daily energy intake and dietary protein intake, and years since menopause and oral oestrogen (in females only) based on theoretical relevance and significance in univariate analysis (P<0·2).
Table 3 Age and sex stratified comparisons of covariate-adjusted skeletal muscle mass index (SMI) by categories of alternate Mediterranean diet score (aMDS)* (Mean values with their standard errors)
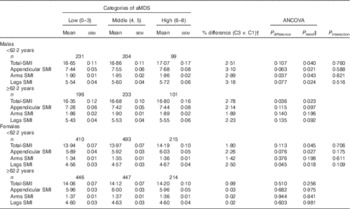
* All analyses were controlled for age, physical activity, smoking status, passive smoking status, daily energy intake and dietary protein intake, and years since menopause and oral oestrogen (in females only) based on theoretical relevance and significance in univariate analysis (P<0·2).
† Percentage difference=(high−low)/low×100 %.
‡ P value for linear trend.
Table 4 showed the associations between food components of the aMDS and SMI in male and female participants (model 2). In males, food components with significant associations included vegetables (all sites, P trend: 0·003–0·030), fruits (all sites, P trend: 0·014–0·022), red and processed meats (all sites, P trend: 0·001–0·011), fish (total body, P trend: 0·011) and ratio of MUFA:SFA (arms, P trend: 0·017). In female, it is vegetables and fruits (all sites except arms, P trend: <0·001–0·005). In consistent with the results in Table 4, discriminant classification analysis showed that the components with significant discriminating ability were mainly those of vegetable, fruit and ratio of MUFA:SFA (online Supplementary Table S2).
Table 4 Comparisons of covariate-adjusted skeletal muscle mass index (SMI) by groups of food components of the alternate Mediterranean diet score (aMDS) in males and femalesFootnote * (Mean values with their standard errors)
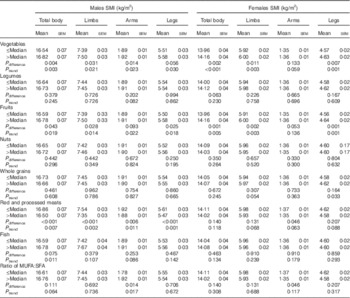
* All analyses were controlled for age, physical activity, smoking status, passive smoking status, daily energy intake and dietary protein intake, and years since menopause and oral oestrogen (in females only).
Multiple regression analyses showed significant linear relationships between the aMDS (as a continuous variable) and SMI at all studied sites in males (P: <0·001–0·036), and at all studied sites (except for the arms) in females (P: <0·001–0·302) (online Supplementary Table S3). We examined whether the association between aMDS and SMI was modified by smoking, passive smoking, physical activity, dietary intake of total energy and protein. No significant interactions were observed (P interaction: 0·092–0·982) (online Supplementary Table S4).
Discussion
In this cross-sectional study of a middle-aged and elderly Chinese population, a favourable association was found between aMDS and SMI after adjusting for potential confounding variables. This association tended to be more pronounced in males (v. females) and in younger (v. older) subjects with ages less than the median age of 59·2 and 62·2 years in females and males.
Previous studies have suggested favourable associations between some components of the MD diet (e.g. moderate consumption of alcohol( Reference Steffl, Bohannon and Petr 16 ), lower intake of SFA( Reference Liang, Oest and Jones 17 ) and higher intake of fish oil( Reference Smith, Julliand and Reeds 18 ) and fruits and vegetables( Reference Kim, Lee and Kye 19 )) and the risk of sarcopenia. Analysing dietary data as a whole rather than single nutrient or single food has been viewed as an important method to nutritional epidemiologists. As foods were eaten together, dietary pattern analysis or quality scores could be easier for making recommendations and to avoid the complex interactions among different kinds of food. Furthermore, eating patterns seem more efficient in the association analyses between dietary factors and nutrition-related diseases( Reference Slattery 36 ). To date, however, few studies have examined the association between the MDS and muscle mass or sarcopenia risk.
One cross-sectional study reported significant positive associations between the MDS and fat-free mass% in 2570 healthy females aged 18–79 years from the Twins UK study( Reference Kelaiditi, Jennings and Steves 22 ). The Berlin Aging Study II, a cross-sectional study including 1509 participants (51 % females, 68·2 (sd 3·7) years), showed that greater adherence to the MD was associated with a higher ratio of appendicular lean mass:BMI in females (P=0·004), but not in males( Reference Nikolov, Spira and Aleksandrova 23 ). Another cross-sectional study showed greater MD-like pattern score was associated with a lower presence of sarcopenia (tertile 3 v. tertile 1: OR=0·42; 95 % CI 0·18, 0·97) in 300 elderly Iranian males and females( Reference Hashemi, Motlagh and Heshmat 24 ). However, null associations of the MDS were found with the odds or the 4-year incidence of sarcopenia (low SMI, low grip strength and low gait speed) were not found in either the cross-sectional analysis (P>0·05) or the longitudinal analysis (P>0·05) in a sample of Hong Kong Chinese males (n 1979) and females (n 1978) aged 65 years or older, possibly because of the limited number of sarcopenic subjects (n 290) at baseline and follow-up (264 new cases)( Reference Chan, Leung and Woo 25 ). In this community-based study, a favourable association between aMDS and SMI was found in a middle-aged and elderly Chinese population, particular in males and younger subjects (age<median). These findings and some previous results highlight the importance and practical value of the MD pattern (or higher MDS) in the maintenance of muscle mass.
Higher aMDS may prevent muscle mass reduction through several mechanisms. First, better adherence to the MD pattern is inversely associated with some pro-inflammatory cytokines, such as C-reactive protein and IL-6. Chronic systemic inflammation was suggested to be one of the key contributors of SMM declination( Reference Lightfoot, McCormick and Nye 37 ). Besides, people with higher aMDS have high intake of fruit and vegetables, which are rich in antioxidants, as oxidative stress is the major mechanism of sarcopenia implicated in aging adults. Sensitivity analysis of aMDS components in our study also showed significant favourable associations between intakes of fruit and vegetables, fruits and SMI, suggesting fruit and vegetables as important explanatory variables.
This study found a more pronounced relationship between aMDS and SMI in males than in females. Similar trends were also found when we consider the aMDS as a continuous variable. Favourable associations between the intake of fruit and vegetables and sarcopenia (defined as the lowest quintile of lean mass) in older adults were similarly more prominent in males than in females in the fourth Korea National Health and Nutrition Examination( Reference Kim, Lee and Kye 19 ). Previous studies have also reported that chronic periods of negative energy balance tend to have less of an effect on growth and loss of body tissue in females than in males( Reference Cortright and Koves 38 ). Other studies( Reference Hughes, Frontera and Roubenoff 39 – Reference Fantin, Di Francesco and Fontana 41 ) have shown much higher rates of reduction in appendicular lean mass in males than in females. Therefore, males appear to be more sensitive than females in terms of muscle mass. The difference in sex hormones may also play a role, as oestrogen could contribute to the antioxidant defense ability in female muscles( Reference Fano, Mecocci and Vecchiet 42 ). In the age-stratified analysis, we found that the favourable associations were more pronounced in the younger group than in the older one. The cumulative effect of a long-term adherence to the healthy dietary pattern seemed to be attenuated at the elderly stage. Our findings suggested that the middle aged (but not elderly) period might be the ‘window period’ for the intervention of dietary pattern on body SMM. Further longitudinal studies are needed to clarify this issue.
This study had several strengths. First, it examined the association between aMDS and SMM in a large community-based population, adding to the limited evidence. Second, average food intake data were used for analyses, which should provide a better estimation of habitual intake before muscle mass was measured in the majority of participants. Third, SMM was measured by DXA rather than bioelectric impedance. Previous studies have shown the excellent validity of DXA compared with the golden methods of MRI and computed tomography (CT) (R 2 0·96–0·98)( Reference Kim, Wang and Heymsfield 43 , Reference Visser, Fuerst and Lang 44 ). Lastly, SMI was assessed at multiple sites. Total body SMI provide us an overall evaluation of participants’ SMM, but when it comes to sarcopenia, we always focus on appendicular muscle mass rather than trunk muscle mass( Reference Cruz-Jentoft, Baeyens and Bauer 4 ). Assessment of multiple sites enabled a comprehensive understanding of the association.
Several limitations of this study should be mentioned. First, the associations derived from a cross-sectional study do not necessarily indicate causality, although adults generally maintain stable long-term dietary habits, and the mean dietary value of the data from two surveys administered about 3 years apart was used for most of the participants. Moreover, as with any observational studies, despite attempts to control major potential confounders, the effect of residual confounding or unmeasured factors could not be completely ruled out. Third, volunteers were recruited to the study, which might have meant that the study population led a healthier lifestyle than the general population. However, the favourable associations did not significantly change according to age, education, income or dietary intake of energy and protein, suggesting that the generalisability of the results should not be greatly affected by these factors. Fourth, thought the aMDS are relatively easier to use, and is comparable among different populations, but is not targeted to our study population. Further modifications might be needed to improve its specificity in the Chinese people. Finally, the 649 withdrew subjects had longer YSM and lower physical activity at baseline than those 2520 participants followed-up (online Supplementary Table S5). We therefore could not exclude the possibility of lost-to-follow-up bias in this study.
Conclusions
The findings of this study show that aMDS positively associated with SMI in Chinese adults, particular males and younger subjects aged less than the medians of 59·2 and 62·2 years in females and males, suggesting that increasing the aMDS might be of great utility in the maintenance of muscle mass at middle ages. Additional longitudinal studies, particularly interventional studies, are needed to confirm these findings.
Acknowledgements
The authors thank the voluntary blood donors that participated in this study and are grateful for the help of other staff, postgraduates and undergraduates involved in data collection in this study.
This study was jointly supported by the National Natural Science Foundation of China (nos 81273049 and 81472965) and by the 5010 Program for Clinical Researches (no. 2007032) of the Sun Yat-Sen University (Guangzhou, China). The funding sources had no role in the design, analysis or writing of this article.
Y.-m. C. conceived and designed the study, and critically revised the paper; H.-y. T. analysed the data and wrote the paper; H.-y. T., R. Q., L.-p. J., Z.-y. C. and G.-d. C. collected the data; Y.-m. C. had primary responsibility for the final content. All the authors read and approved the final version of the manuscript.
The authors declare that there are no conflicts of interest.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114517001118









