22 results
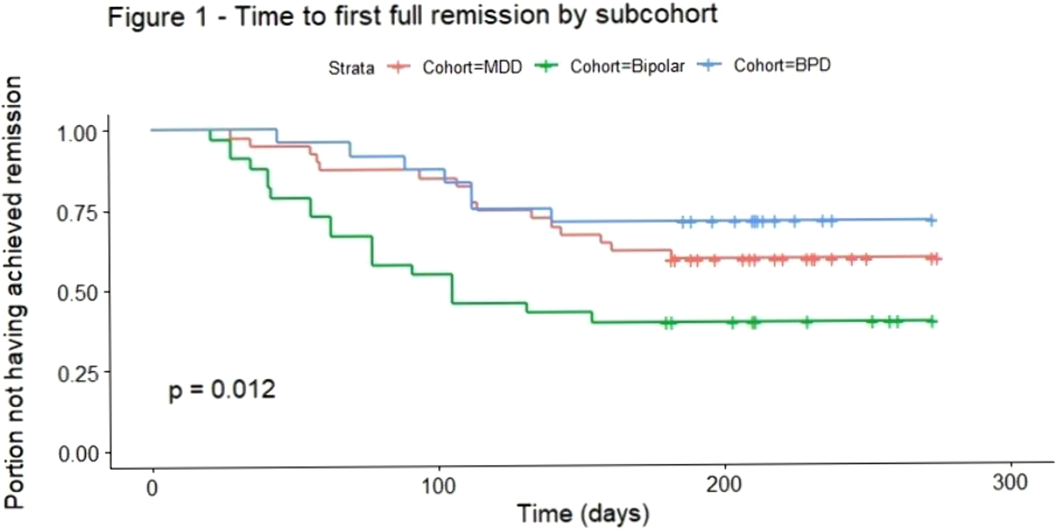
Bipolar disorder correlated to shorter remission latency and borderline personality disorder symptom severity to longer in depression – a prospective cohort study of major depressive patients
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, pp. S85-S86
-
- Article
-
- You have access
- Open access
- Export citation
Effect of pericardiotomy on exercise-induced pulmonary hypertension in the horse
-
- Journal:
- Equine and Comparative Exercise Physiology / Volume 3 / Issue 1 / February 2006
- Published online by Cambridge University Press:
- 09 March 2007, pp. 45-51
- Print publication:
- February 2006
-
- Article
- Export citation
Hydrothermal Synthesis and Structure of Neptunium(V) Oxide
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 985 / 2006
- Published online by Cambridge University Press:
- 19 October 2011, 0985-NN12-02
- Print publication:
- 2006
-
- Article
- Export citation
The Structures of Polynuclear Th(IV) Hydrolysis Products
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 986 / 2006
- Published online by Cambridge University Press:
- 26 February 2011, 0986-OO09-02
- Print publication:
- 2006
-
- Article
- Export citation
Studying Actinide Correlations in Solution using High-Energy X-ray Scattering
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 893 / 2005
- Published online by Cambridge University Press:
- 26 February 2011, 0893-JJ05-18
- Print publication:
- 2005
-
- Article
- Export citation
Presence and Persistence of Uranyl Peroxide Nanoclusters in Contact with Geological Media
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 893 / 2005
- Published online by Cambridge University Press:
- 26 February 2011, 0893-JJ09-06
- Print publication:
- 2005
-
- Article
- Export citation
Relationships between the crystal chemistry and magetic properties of Np5+ sulfates
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 893 / 2005
- Published online by Cambridge University Press:
- 26 February 2011, 0893-JJ09-02
- Print publication:
- 2005
-
- Article
- Export citation
Similarities between the Coordination of Actinide Ions in Solution and the Structures of their Related Crystalline Phases
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 893 / 2005
- Published online by Cambridge University Press:
- 26 February 2011, 0893-JJ08-02
- Print publication:
- 2005
-
- Article
- Export citation
Changing Np Redox Speciation in the Synchrotron Beam
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 802 / 2003
- Published online by Cambridge University Press:
- 01 February 2011, DD4.3
- Print publication:
- 2003
-
- Article
- Export citation
Am Doping in Copper-Oxide Superconducting Systems
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 802 / 2003
- Published online by Cambridge University Press:
- 01 February 2011, DD4.7
- Print publication:
- 2003
-
- Article
- Export citation
In Situ Actinide X-ray Absorption Spectroelectrochemistry
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 802 / 2003
- Published online by Cambridge University Press:
- 01 February 2011, DD4.5
- Print publication:
- 2003
-
- Article
- Export citation
The Influence of Desulfovibrio Desulfuricans on Neptunium Chemistry
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 590 / 1999
- Published online by Cambridge University Press:
- 10 February 2011, 27
- Print publication:
- 1999
-
- Article
- Export citation
The Structure of Actinide Ions Exchanged into Native and Modified Zeolites and Clays
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 590 / 1999
- Published online by Cambridge University Press:
- 10 February 2011, 39
- Print publication:
- 1999
-
- Article
- Export citation
The Effects of Surface Modification on the Speciation of Metal Ions Intercalated into Aluminosilicates
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 465 / 1996
- Published online by Cambridge University Press:
- 03 September 2012, 473
- Print publication:
- 1996
-
- Article
- Export citation
Uranium Speciation in Glass Corrosion Layers: an XAFS Study
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 465 / 1996
- Published online by Cambridge University Press:
- 03 September 2012, 229
- Print publication:
- 1996
-
- Article
- Export citation
Observations of Cef-Split Intermultiplet Transitions in Optically Opaque EuBa2Cu3O7 Using Inelastic Neutron Scattering
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 376 / 1994
- Published online by Cambridge University Press:
- 22 February 2011, 535
- Print publication:
- 1994
-
- Article
- Export citation
The Oxidation State and Magnetic Behaviour of Tb in High-Tc Related Materials
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 376 / 1994
- Published online by Cambridge University Press:
- 22 February 2011, 529
- Print publication:
- 1994
-
- Article
- Export citation
Synthesis and Properties of Lanthanide-Exchanged Preyssler's Heteropolyanions
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 368 / 1994
- Published online by Cambridge University Press:
- 15 February 2011, 223
- Print publication:
- 1994
-
- Article
- Export citation
The Use of Curium as a Rare Earth Substitute in High Tc‐Related Compounds
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 169 / 1989
- Published online by Cambridge University Press:
- 28 February 2011, 73
- Print publication:
- 1989
-
- Article
- Export citation
Polarized X-Ray Absorption Studies of Oxide Superconductors
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 143 / 1988
- Published online by Cambridge University Press:
- 21 February 2011, 97
- Print publication:
- 1988
-
- Article
- Export citation

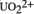 ) cations exchanged into two classes of microporous aluminosilicate minerals: zeolites and smectite clays. XAS is also employed to examine the fate of the exchanged cations after modification of the mineral surface using self-assembled organic films and/or exposure to hydrothermal conditions. These treatments serve as models for the forces that ultimately determine the chemical fate of the actinide cations in the environment. The speciation of the cations depends on the pore size of the aluminosilicate, which is fixed for the zeolites and variable for the smectites.
) cations exchanged into two classes of microporous aluminosilicate minerals: zeolites and smectite clays. XAS is also employed to examine the fate of the exchanged cations after modification of the mineral surface using self-assembled organic films and/or exposure to hydrothermal conditions. These treatments serve as models for the forces that ultimately determine the chemical fate of the actinide cations in the environment. The speciation of the cations depends on the pore size of the aluminosilicate, which is fixed for the zeolites and variable for the smectites.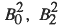 and the spinorbit coupling parameter were obtained by fitting the splitting of the J=l multiplet and the energy separation between the J=0 and 1 multiplets. The J=0 → 1 splitting observed here is smaller than previously seen by optical spectroscopic studies on a variety of transparent, ionic compounds, necessitating fitting of the free-ion parameter. Additional spectroscopic information on the J=2 multiplet indicates that additional fitting of free ion parameters must be included to adequately model the observed low energy separation between the two lowest J-multiplets. Preliminary calculation on the Q-dependence of the CEF split J=0 → 1 transitions and the comparison with observations are presented.
and the spinorbit coupling parameter were obtained by fitting the splitting of the J=l multiplet and the energy separation between the J=0 and 1 multiplets. The J=0 → 1 splitting observed here is smaller than previously seen by optical spectroscopic studies on a variety of transparent, ionic compounds, necessitating fitting of the free-ion parameter. Additional spectroscopic information on the J=2 multiplet indicates that additional fitting of free ion parameters must be included to adequately model the observed low energy separation between the two lowest J-multiplets. Preliminary calculation on the Q-dependence of the CEF split J=0 → 1 transitions and the comparison with observations are presented.