48 results
Fusion Pore: An Evolutionary Invention of Nucleated Cells
-
- Journal:
- European Review / Volume 18 / Issue 3 / July 2010
- Published online by Cambridge University Press:
- 01 July 2010, pp. 347-364
-
- Article
- Export citation
Spectroscopic follow-up of the colliding-wind binary WR 140 during the 2009 January periastron passage
-
- Journal:
- Proceedings of the International Astronomical Union / Volume 6 / Issue S272 / July 2010
- Published online by Cambridge University Press:
- 12 July 2011, pp. 501-502
- Print publication:
- July 2010
-
- Article
-
- You have access
- Export citation
Evolution and appearance of Be stars in SMC clusters
-
- Journal:
- Proceedings of the International Astronomical Union / Volume 5 / Issue S266 / August 2009
- Published online by Cambridge University Press:
- 18 January 2010, pp. 470-473
- Print publication:
- August 2009
-
- Article
-
- You have access
- Export citation
Rotational velocity distributions of A-type stars
-
- Journal:
- Proceedings of the International Astronomical Union / Volume 2004 / Issue IAUS224 / July 2004
- Published online by Cambridge University Press:
- 02 March 2005, pp. 109-114
- Print publication:
- July 2004
-
- Article
-
- You have access
- Export citation
Modelling Stellar Rotation for Optical Long Baseline Interferometry
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 187-188
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Rotation and Properties of Be Stars
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 73-82
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Differential Rotation and the v sin i Parameter
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 21-22
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
CNO abundances in Early Type Be Stars
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 222-223
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Evolutionary State of “Field” Be Stars Deduced from BCD Parameters
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 87-88
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Correlation between v sin i and the Peculiar Velocities of Be Stars
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 85-86
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Rotational Velocities of “Field” Be Stars
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 89-90
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Chemical Composition of the Pole-On Be Star HD 120991
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 224-225
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
The Initial Mass Function of Be Stars
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 83-84
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Rotational Velocity Distributions of A-Type Main-Sequence Stars
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 55-56
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Fast Rotating Stars: Effect of the Aspect Angle on Line Profiles
-
- Journal:
- Symposium - International Astronomical Union / Volume 215 / 2004
- Published online by Cambridge University Press:
- 26 May 2016, pp. 23-24
- Print publication:
- 2004
-
- Article
-
- You have access
- Export citation
Line Profile and Photometric Variations of the Be Star η Cen1
-
- Journal:
- International Astronomical Union Colloquium / Volume 185 / 2002
- Published online by Cambridge University Press:
- 12 April 2016, pp. 252-253
- Print publication:
- 2002
-
- Article
-
- You have access
- Export citation
Fundamental Parameters of Be Stars Seen Equator-On
-
- Journal:
- International Astronomical Union Colloquium / Volume 185 / 2002
- Published online by Cambridge University Press:
- 12 April 2016, pp. 244-245
- Print publication:
- 2002
-
- Article
-
- You have access
- Export citation
Spectrophotometric and Photometric Behaviours of Be Stars: the Circumstellar Envelope Formation Mechanisms
-
- Journal:
- International Astronomical Union Colloquium / Volume 175 / 2000
- Published online by Cambridge University Press:
- 12 April 2016, pp. 448-451
- Print publication:
- 2000
-
- Article
-
- You have access
- Export citation
Short-lived and Long-lived Outbursts in B and Be Stars from Hipparcos Photometry and Modelling
-
- Journal:
- International Astronomical Union Colloquium / Volume 175 / 2000
- Published online by Cambridge University Press:
- 12 April 2016, pp. 348-351
- Print publication:
- 2000
-
- Article
-
- You have access
- Export citation
The Emitting Envelope of Pleione between 1987 and 19981
-
- Journal:
- International Astronomical Union Colloquium / Volume 175 / 2000
- Published online by Cambridge University Press:
- 12 April 2016, pp. 562-565
- Print publication:
- 2000
-
- Article
-
- You have access
- Export citation



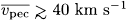 of early-type Be Stars. Further corrections of space velocities of Be Stars for systematic movements of stellar groups to which they belong, as well as identifications of coeval stars have to be performed in order to decide whether the high rotation of some of them could be the consequence of gravitational encounters.
of early-type Be Stars. Further corrections of space velocities of Be Stars for systematic movements of stellar groups to which they belong, as well as identifications of coeval stars have to be performed in order to decide whether the high rotation of some of them could be the consequence of gravitational encounters.