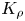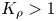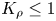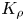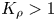The buoyancy-driven instabilities triggered by neutralization reaction were studied experimentally in a miscible two-layer system placed in a vertically oriented Hele-Shaw cell. The initial density stratification was always set to exclude the development of the Rayleigh–Taylor instability. The problem was examined for a few reactant pairs formed by a strong acid and a strong base. To classify the numerous experimental observations we introduced a dimensionless parameter, namely, a reaction-induced buoyancy number 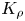 ${K}_{\rho }$, which defines the density of the reaction zone relative to that of the upper layer. We show that, depending on the value of this parameter, one of two global scenarios develops in the system right after the layers came into contact. If
${K}_{\rho }$, which defines the density of the reaction zone relative to that of the upper layer. We show that, depending on the value of this parameter, one of two global scenarios develops in the system right after the layers came into contact. If 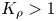 ${K}_{\rho }>1$, the process is governed mainly by diffusion, which results later on in the development of relatively weak convective motion caused by a differential-diffusion effect. Besides the irregular finger-type flow structures, reported earlier in numerous studies, we found a new type of instability, called the concentration-dependent diffusion instability, which is characterized by the formation of a regular cell-type convective pattern. In the case
${K}_{\rho }>1$, the process is governed mainly by diffusion, which results later on in the development of relatively weak convective motion caused by a differential-diffusion effect. Besides the irregular finger-type flow structures, reported earlier in numerous studies, we found a new type of instability, called the concentration-dependent diffusion instability, which is characterized by the formation of a regular cell-type convective pattern. In the case 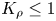 ${K}_{\rho }\le 1$, the unstable density stratification above the reaction front leads to the development of vigorous convection in the upper layer, forcing the reaction front to move downwards fast, so that it takes just a few minutes for reagents to burn out. We show that a new parameter can be used to plot the stability maps, which allows us to predict reaction–diffusion–convection processes in similar systems prior to experiment.
${K}_{\rho }\le 1$, the unstable density stratification above the reaction front leads to the development of vigorous convection in the upper layer, forcing the reaction front to move downwards fast, so that it takes just a few minutes for reagents to burn out. We show that a new parameter can be used to plot the stability maps, which allows us to predict reaction–diffusion–convection processes in similar systems prior to experiment.