The Wisconsin Twin Project began in the early 1990s in the state of Wisconsin, which is north-centrally located in the United States. The design of the Wisconsin Twin Project accommodates a range of research on emotional and cognitive development, with a strong emphasis on clinical outcomes. Assessments for some participants began in the prenatal period, and the oldest age of follow-up was approximately 24 years.
The broad themes of our results span psychophysiology, temperament development, gene–environment interplay and other aspects of mental and physical health. Many studies incorporate observational behavior and laboratory temperament assessments that were partially developed in our lab (Buss & Goldsmith, Reference Buss and Goldsmith2000; Gagne et al., Reference Gagne, Van Hulle, Aksan, Essex and Goldsmith2011; Goldsmith et al., Reference Goldsmith, Lemery-Chalfant, Schmidt, Schmidt, Chow and Justen2010; Planalp et al., Reference Planalp, Van Hulle, Gagne and Goldsmith2017). Several of our neuroimaging studies incorporate the monozygotic (MZ) difference design (Adluru et al., Reference Adluru, Luo, Van Hulle, Schoen, Davidson, Alexander and Goldsmith2017; Alisch et al., Reference Alisch, Van Hulle, Chopra, Bhattacharyya, Zhang, Davidson and Goldsmith2017; Burghy et al., Reference Burghy, Fox, Cornejo, Stodola, Sommerfeldt, Westbrook and Birn2016; Carroll et al., Reference Carroll, Planalp, Van Hulle and Goldsmith2019). Recent empirical results from more than 30 papers are summarized in Schmidt et al. (Reference Schmidt, Brooker, Carroll, Gagne, Luo, Planalp and Goldsmith2019).
Sample, Recruitment, and Retention
The Wisconsin Twin Project sample is based on birth-record-based cohorts of twins born in the state of Wisconsin during the years 1989–2004 (Goldsmith et al., Reference Goldsmith, Lemery-Chalfant, Schmidt, Arneson and Schmidt2007; Lemery-Chalfant et al., Reference Lemery-Chalfant, Goldsmith, Schmidt, Arneson and Van Hulle2006; Schmidt et al., Reference Schmidt, Van Hulle, Brooker, Meyer, Lemery-Chalfant and Goldsmith2013; Van Hulle et al., Reference Van Hulle, Lemery-Chalfant and Goldsmith2002). According to US Census Bureau and other data, of the 50 states, Wisconsin ranks 20th in total population, 25th in physical size, 23rd in median household income, 20th in gross domestic product, 11th in percentage of people who are high school graduates, 26th in percentage of people who are college graduates and 37th in percentage of people who identify as racial or ethnic minorities. Although differences in state ranks in these categories often reflect small differences, Wisconsin is clearly about average in many characteristics among US states. As a result, data from Wisconsin are likely to represent the US population reasonably well.
After nearly 30 years, the research program encompasses a series of longitudinal studies that span infancy to early adulthood (see Figure 1), with another currently planned longitudinal follow-up in early adulthood. Families entered the Wisconsin Twin Project through two primary mechanisms: (1) multimethod recruiting (e.g., advertising, recruiting from twin groups); and (2) state birth records, both depicted at the top of Figure 1. No new recruiting occurred after the infant–toddler years except for subsets of participants for Studies 3 and 5. The arrows in Figure 1 depict sequences of subsequent testing occasions across development. The boxes in Figure 1 represent studies that each comprise one or more testing occasions. A given study typically yields several published papers, some of which span studies. Each study includes a target age (e.g., birth, age 12–18 years). The arrows also depict what earlier and later data exist. For instance, the sample in Study 2c was tested previously in either Study 1 or Study 2a. A portion of the Study 2c sample was also tested in Study 4. Study 2c encapsulated three separate testing occasions at ages 7, 12 and 15 years. Detailed descriptions of early assessment were reported in Schmidt et al. (Reference Schmidt, Van Hulle, Brooker, Meyer, Lemery-Chalfant and Goldsmith2013).
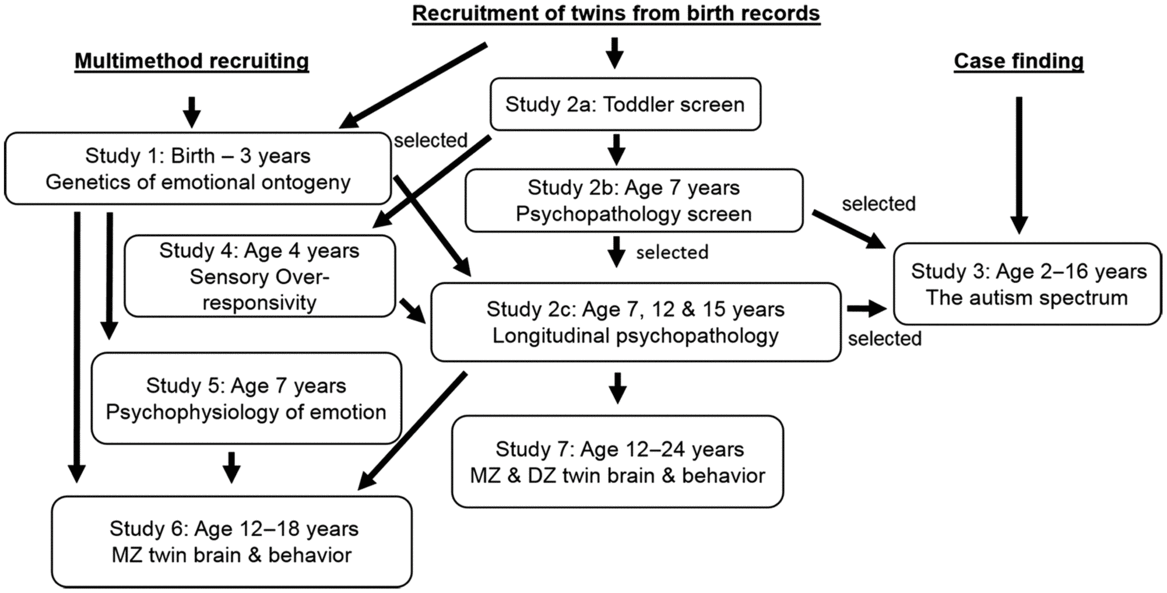
Figure 1. Wisconsin twin research project overview.
As an illustration, consider a twin pair recruited from state birth records (top center of Figure 1). Generally, the twins would have still been infants when we accessed the birth records. The parents were sent an invitation letter and subsequently agreed to be contacted for future research participation. The first parent interview and survey about toddler temperament, parenting and challenging behaviors occurred in Study 2a, when the twins were approximately 24 months old. After the toddler screen, they were assessed again via parental report at age 7 years in Study 2b. If either twin’s scores met selection criteria in Study 2b (e.g., mild enrichment for psychopathology), they were tested up to three times in Study 2c at ages 7, 12 and 15 years, and a final time around age 17 years in Study 6, with neuroimaging and behavioral assessment. The arrows show other pathways to Study 6.
As another example, consider a different twin pair located under the multimethod recruitment (e.g., local advertisement) shortly after birth, at an age before state birth records were available. This twin pair was first tested in Study 1 and tested with lab-based observational assessment on multiple occasions between birth and age 3 years. This twin pair was tested again in Study 5 with laboratory measurement of psychophysiology and then a final time in Study 6, the same final study as for the twin pair described in the previous paragraph.
Twin family recruitment and early results were covered in prior overviews of the project (Goldsmith et al., Reference Goldsmith, Lemery-Chalfant, Schmidt, Arneson and Schmidt2007; Schmidt et al., Reference Schmidt, Van Hulle, Brooker, Meyer, Lemery-Chalfant and Goldsmith2013). Briefly, initial contact was attempted with a mailed letter and contact form. Contact was maintained with multiple phone numbers, email addresses, a toll-free phone number and secondary contact information from a family friend or relative. Sample retention efforts included newsletters and a website devoted to participant communication. We attempted to recapture twin families at later testing occasions, where possible. We used Web-based tracing methods (e.g., public court records) to locate families with whom we had lost contact. The University of Wisconsin Survey Center also provided tracing services. All of these procedures helped maintain the research sample longitudinally.
The research is conducted at the University of Wisconsin–Madison’s Waisman Center and the Department of Psychology (https://goldsmithtwins.waisman.wisc.edu/). Procedures in studies under the Wisconsin Twin Project were approved by University of Wisconsin–Madison Internal Review Boards and comply with the Helsinki Accords of 1975, as revised in 2008.
The Wisconsin Twin Project’s signature approach is its depth and breadth of phenotyping (see Table 1). Sample sizes shown in Table 1 depict longitudinal data collection. For instance, 100% of the sample of 311 twin pairs with neuroimaging were tested in the earlier waves of data collection.
Table 1. Research modality and sample sizes by study
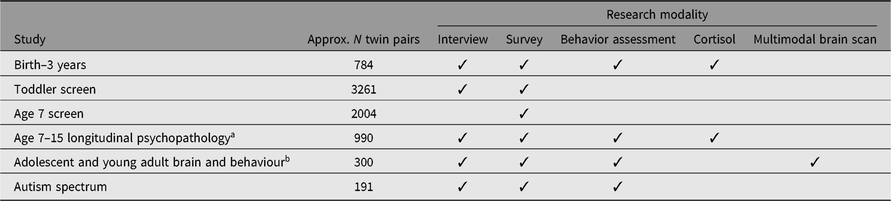
Note: Interview and written survey measures of sensory over-responsivity were included in all studies. Medical record data from the twin pregnancy and birth exist for most twins.
a Genotyping was done with the longitudinal psychopathology study.
b Neuroimaging studies comprise a pair of studies, Studies 6 and 7 from Figure 1. The vast majority (85%) of twins were participants of the RDoC neuroimaging study.
Twin zygosity was classified with multiple methods across testing occasions. Initially, zygosity was classified with the Zygosity Questionnaire for Young Children (Goldsmith, Reference Goldsmith1991). Observational ratings of zygosity were collected during in-person assessments. Ambiguous zygosity was resolved by genotyping.
Twin Neuroimaging Studies
Twin neuroimaging was a focus of the project during the past decade. The neuroimaging studies assessed brain structure and function with multimodal magnetic resonance imaging (MRI). Although MRI methods have varied with improvements in technology, we use T1- and T2-weighted structural scans, diffusion-weighted imaging, arterial spin labeling, resting state functional magnetic resonance imaging (fMRI) and task-related fMRI. The combined neuroimaging sample comprises 600 twins (300 pairs), all of whom have been studied previously, some on seven prior testing occasions. We oversampled MZ twins (66%). Some of our early neuroimaging and epigenetic studies use the MZ difference design (Adluru et al., Reference Adluru, Luo, Van Hulle, Schoen, Davidson, Alexander and Goldsmith2017; Alisch et al., Reference Alisch, Van Hulle, Chopra, Bhattacharyya, Zhang, Davidson and Goldsmith2017; Burghy et al., Reference Burghy, Fox, Cornejo, Stodola, Sommerfeldt, Westbrook and Birn2016). The twin neuroimaging data were also used to illustrate novel computational MRI methods (Chung, Luo, Adluru et al., Reference Chung, Luo, Adluru, Alexander, Davidson and Goldsmith2018; Chung, Luo, Leow et al., Reference Chung, Luo, Leow, Alexander, Davidson, Goldsmith, Frangi, Schnabel, Davatzikos, Alberola-López and Fichtinger2018; Huang et al., Reference Huang, Chung, Carroll and Goldsmith2019).
A majority of our neuroimaging work incorporates Research Domain Criteria (RDoC) as a framework for the behavioral assessments. Specifically, the RDoC twin study used longitudinal and quantitative genetic approaches to establish developmental antecedents and neural substrates for the RDoC positive valence systems (e.g., anticipatory positive affect and contentment) and negative valence systems (e.g., acute fear, potential threat/anxiety, frustrative nonreward and loss; Cuthbert, Reference Cuthbert2014). The RDoC twin study aimed to (a) establish distinctiveness, stability and external validity of each RDoC construct during childhood and adolescence; (b) investigate the relationship between brain structure and function (via MRI) and concurrent and longitudinal RDoC measures; and (c) utilize the MZ difference design to highlight early environmental contributions to later brain structure and function. In the MRI analyses, we focused more on white matter microstructure and on resting state and task-related functional measures (i.e., circuitry and networks) than on gray matter structure.
The RDoC twin study enrolled 518 MZ (56%) and dizygotic (DZ) twin individuals (mean age = 17.4 years, SD = 2.2 years). Approximately 70% were under 18 years of age at the time of assessment. Parents (88% mothers) of adolescents completed surveys. Data collection concluded in early 2019. Tables 2 and 3 list the principal twin-individual-level instruments and family-level instruments collected concurrently with neuroimaging data for the RDoC sample.
Table 2. Principal individual-level instruments for the adolescent and young adult RDoC brain and behaviour study

Note: N = 518 MZ and DZ twins (68% were adolescents; 56% MZ).
a Co-twin ratings also collected.
b Adult twin ratings collected.
Table 3. Principal family-level instruments for the adolescent and young adult brain and behaviour study
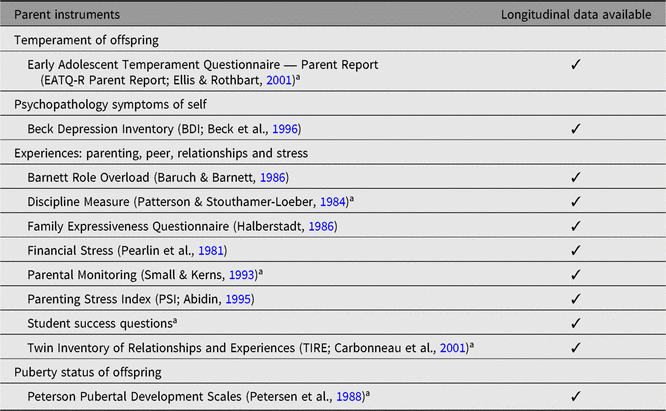
Note: We collected parent ratings for twins under 18 years of age at the time of MRI testing.
a Completed separately for each twin.
Future Directions
Although the panel is no longer being expanded, we maintain contact with the full panel of twin families through biannual newsletters and a website. These data could support diverse empirical investigations. The sample sizes afford the possibility to examine selected subsamples (e.g., clinical cases, the MZ twin difference sample). Planned research includes new assessments with early adulthood outcomes, deeper assessment of experiential differences using ecological momentary assessment and a greater focus on general health outcomes. We welcome interest in collaboration. A portion of our data, including the neuroimaging, resides in the National Data Archive (all data related to the study named Validating RDoC for Children and Adolescents: A Twin Study with Neuroimaging, collection #2105; https://ndar.nih.gov/edit_collection.html?id=2105&source=RDoCdb&funding=NIH+-+Extramural).
Acknowledgments
This work was supported by the National Institute of Mental Health (R01-MH101504, R01-MH059785, R01-MH069793 and R37-MH050560), the Wisconsin Center for Affective Science (P50-MH069315), two Conte Neuroscience Centers (P50-MH084051 and P50-MH100031), the Wallace Research Foundation and the National Alliance for Autism Research. Infrastructure support was provided by the Waisman Center via core support from the National Institute of Child Health and Human Development (P30-HD003352 and U54-HD090256). The authors owe special gratitude to Wisconsin twins and their families for their generous time for research participation. We also thank a large team of scientific collaborators, graduate students, postdoctoral trainees, research staff and undergraduate student research assistants.








