Article contents
Determination of adsorption-controlled growth windows of chalcogenide perovskites
Published online by Cambridge University Press: 12 February 2018
Abstract
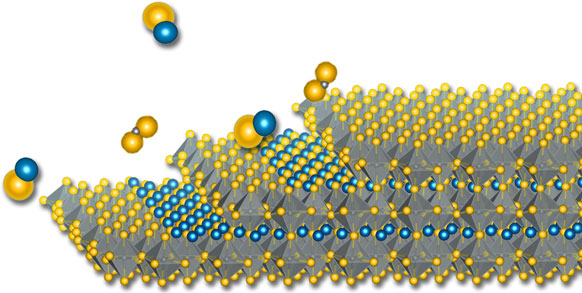
Ternary sulfides and selenides in the distorted-perovskite structure (“chalcogenide perovskites”) are predicted by theory to be semiconductors with a band gap in the visible-to-infrared and may be useful for optical, electronic, and energy conversion technologies. Here we use computational thermodynamics to predict the pressure–temperature phase diagrams for select chalcogenide perovskites. Our calculations incorporate formation energies calculated by density functional theory, and empirical estimates of heat capacities. We highlight the windows of thermodynamic equilibrium between solid chalcogenide perovskites and the vapor phase at high temperature and very low pressure. These results can guide the adsorption-limited growth of ternary chalcogenides by molecular beam epitaxy.
- Type
- Research Letters
- Information
- Copyright
- Copyright © Materials Research Society 2018
References
A correction has been issued for this article:
- 5
- Cited by
Linked content
Please note a has been issued for this article.





