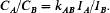Volume 51 - Issue 359 - March 1987
Electron Microscopy in Mineralogy and Petrology
Electron Microscopy in Mineralogy and Petrology
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1-2
-
- Article
- Export citation
Atomic number and crystallographic contrast images with the SEM: a review of backscattered electron techniques
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 3-19
-
- Article
- Export citation
Mineralogical applications of the analytical SEM in archaeology
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 21-31
-
- Article
- Export citation
Convergent beam electron diffraction
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 33-48
-
- Article
- Export citation
Quantitative X-ray microanalysis of thin specimens in the transmission electron microscope; a review
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 49-60
-
- Article
- Export citation
Iron ore sinter in the analytical transmission electron microscope
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 61-69
-
- Article
- Export citation
Composition-induced microstructures in rhombohedral carbonates
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 71-86
-
- Article
- Export citation
A study of the thickness of ferrihydrite coatings on kaolinite
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 87-92
-
- Article
- Export citation
A natural example of the disequilibrium breakdown of biotite at high temperature: TEM observations and comparison with experimental kinetic data
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 93-106
-
- Article
- Export citation
Transmission electron microscopy of pyrometamorphic breakdown of phengite and chlorite
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 107-121
-
- Article
- Export citation
Analytical transmission electron microscopy in the study of diagenetic clay minerals
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 123-124
-
- Article
- Export citation
Electron optical studies of experimentally deformed Tennessee Sandstone and quartz + kaolinite gouge
-
- Published online by Cambridge University Press:
- 05 July 2018, p. 125
-
- Article
- Export citation
A TEM investigation of optical variations in sphalerite
-
- Published online by Cambridge University Press:
- 05 July 2018, p. 127
-
- Article
- Export citation
The Hallimond Lecture
Minerals and the electron microscope
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 129-138
-
- Article
- Export citation
Mineralogy
Palagonites of the Red Sea: a new occurrence of hydroxysulphate
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 139-143
-
- Article
- Export citation
High-pressure, synthetic loveringite-davidite and its rare earth element geochemistry
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 145-149
-
- Article
- Export citation
Sector-zoned kaersutite in camptonites from Morocco
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 151-156
-
- Article
- Export citation
The lattice dynamics of forsterite
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 157-170
-
- Article
- Export citation
Short Communications
Estimation equations for F in fractional crystallisation and partial melting
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 171-172
-
- Article
- Export citation
Hetaerolite from Eastern Cliff, Kennack Sands, Cornwall: the first British occurrence
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 172-173
-
- Article
- Export citation