Article contents
Nipalarsite, Ni8Pd3As4, a new platinum-group mineral from the Monchetundra Intrusion, Kola Peninsula, Russia
Published online by Cambridge University Press: 05 November 2019
Abstract
Nipalarsite, Ni8Pd3As4, is a new platinum-group mineral discovered in the sulfide-bearing orthopyroxenite of the Monchetundra layered intrusion, Kola Peninsula, Russia (67°52′22″N, 32°47′60″E). Nipalarsite forms anhedral grains (5–80 µm in size) in intergrowths with sperrylite, kotulskite, hollingworthite, isomertieite, menshikovite, palarstanide, nielsenite and monchetundtraite enclosed in pentlandite, anthophyllite, actinolite and chlorite. Nipalarsite is brittle, has a metallic lustre and a grey streak. In plane-polarised light, nipalarsite is light grey with a blue tinge. Reflectance values in air (in %) are: 46.06 at 470 nm, 48.74 at 546 nm, 50.64 at 589 nm and 54.12 at 650 nm. Values of VHN20 fall between 400.5 and 449.2 kg.mm–2, with a mean value of 429.9 kg.mm–2, corresponding to a Mohs hardness of ~4. The average result of 27 electron microprobe wavelength dispersive spectroscopy analyses of nipalarsite is (wt.%): Ni 44.011, Pd 28.74, Fe0.32, Cu 0.85, Pt 0.01, Au 0.05, As 25.42, Sb 0.05, Te 0.39, total 99.85. The empirical formula (normalised to 15 atoms per formula unit) is: (Ni8.10Fe0.06)Σ8.16(Pd2.94Cu0.18)Σ3.12(As3.68Te0.03)Σ3.71 or, ideally, Ni8Pd3As4. Nipalarsite is cubic, space group Fm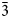 $\bar{3}$m, with a = 11.4428(9) Å, V = 1498.3(4) Å3 and Z = 8. The strongest lines in the powder X-ray diffraction pattern of synthetic Ni8Pd3As4 [d, Å (I) (hkl)] are: 2.859(10)(004), 2.623(6)(313), 2.557(6)(024), 2.334(11)(224), 2.201(35)(115,333), 2.021(100)(044), 1.906(8)(006,244) and 1.429(7)(008). The crystal structure was solved and refined from the single-crystal X-ray diffraction data of synthetic Ni8Pd3As4. The relation between natural and synthetic nipalarsite is illustrated by an electron back-scattered diffraction study of natural nipalarsite. The density calculated on the basis of the empirical formula of nipalarsite is 9.60 g.cm–3. The mineral name corresponds to the three main elements: Ni, Pd and As.
$\bar{3}$m, with a = 11.4428(9) Å, V = 1498.3(4) Å3 and Z = 8. The strongest lines in the powder X-ray diffraction pattern of synthetic Ni8Pd3As4 [d, Å (I) (hkl)] are: 2.859(10)(004), 2.623(6)(313), 2.557(6)(024), 2.334(11)(224), 2.201(35)(115,333), 2.021(100)(044), 1.906(8)(006,244) and 1.429(7)(008). The crystal structure was solved and refined from the single-crystal X-ray diffraction data of synthetic Ni8Pd3As4. The relation between natural and synthetic nipalarsite is illustrated by an electron back-scattered diffraction study of natural nipalarsite. The density calculated on the basis of the empirical formula of nipalarsite is 9.60 g.cm–3. The mineral name corresponds to the three main elements: Ni, Pd and As.
Keywords
- Type
- Article
- Information
- Copyright
- Copyright © Mineralogical Society of Great Britain and Ireland 2019
Footnotes
Associate Editor: Irina O Galuskina
References
- 6
- Cited by




