Article contents
First-principles study on mechanical properties and electronic structures of Ti–Al intermetallic compounds
Published online by Cambridge University Press: 04 February 2019
Abstract
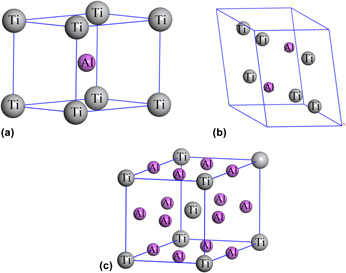
In this study, we investigated the elastic constants, moduli, hardness, and electronic structures of Ti–Al intermetallic compounds (TiAl, Ti3Al, and TiAl3) using first-principles calculations. The cohesive energy and formation enthalpy of these compounds are negative, which indicates that they are thermodynamically stable. We calculated the elastic constants and moduli using the stress–strain method and Voigt–Reuss–Hill approximation, respectively. We evaluated the mechanical anisotropy of these compounds using the anisotropic index and found that the results are in good agreement with other experimental and theoretical data. We evaluated the chemical bonding of these compounds by calculating their density of states, the results of which revealed that the bonding behavior of all Ti–Al intermetallic compounds involved a mixture of metallic and covalent bonds. We also estimated the Debye temperature and sound velocities of these Ti–Al intermetallic compounds.
- Type
- Article
- Information
- Journal of Materials Research , Volume 34 , Issue 7: Focus Section: Interconnects and Interfaces in Energy Conversion Materials , 15 April 2019 , pp. 1112 - 1120
- Copyright
- Copyright © Materials Research Society 2019
References
- 8
- Cited by




