Article contents
Effects of the geothermal gradient on the convective dissolution in CO2 sequestration
Published online by Cambridge University Press: 19 May 2023
Abstract
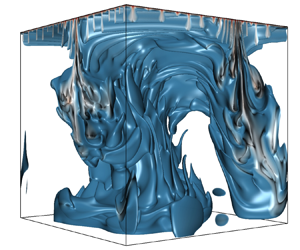
Convective dissolution is an important mechanism for long-term CO $_2$ sequestration in deep saline aquifers. The presence of an unstable geothermal gradient can affect the process of dissolution. In this paper, we present direct numerical simulations in a three-dimensional porous medium at three different concentration Rayleigh numbers
$_2$ sequestration in deep saline aquifers. The presence of an unstable geothermal gradient can affect the process of dissolution. In this paper, we present direct numerical simulations in a three-dimensional porous medium at three different concentration Rayleigh numbers  $Ra_S$ with a set of thermal Rayleigh numbers
$Ra_S$ with a set of thermal Rayleigh numbers  $Ra_T$. Simulations reveal that the flow structures alter when
$Ra_T$. Simulations reveal that the flow structures alter when  ${\textit {Ra}}_T$ increases for a fixed
${\textit {Ra}}_T$ increases for a fixed  ${\textit {Ra}}_S$. Strong thermal gradient can yield large-scale convection rolls which change the horizontal distribution and motions of concentration fingers. The time evolution of fluxes also has different responses to different
${\textit {Ra}}_S$. Strong thermal gradient can yield large-scale convection rolls which change the horizontal distribution and motions of concentration fingers. The time evolution of fluxes also has different responses to different  ${\textit {Ra}}_T$. A theoretical model is developed and successfully describes the evolution of concentration flux and volume averaged concentration during the final shutdown stage. We further calculate the dissolved CO
${\textit {Ra}}_T$. A theoretical model is developed and successfully describes the evolution of concentration flux and volume averaged concentration during the final shutdown stage. We further calculate the dissolved CO $_2$ into the interior over time, which shows non-monotonic variations as
$_2$ into the interior over time, which shows non-monotonic variations as  ${\textit {Ra}}_T$ increases. At the end of our simulations, the maximum increment of dissolved CO
${\textit {Ra}}_T$ increases. At the end of our simulations, the maximum increment of dissolved CO $_2$ occurs when density ratio is around unity for all three concentration Rayleigh numbers we have explored. We apply our results to a typical geological reservoir and discuss their implications.
$_2$ occurs when density ratio is around unity for all three concentration Rayleigh numbers we have explored. We apply our results to a typical geological reservoir and discuss their implications.
JFM classification
- Type
- JFM Papers
- Information
- Copyright
- © The Author(s), 2023. Published by Cambridge University Press
References
- 1
- Cited by





