Introduction
With the emergence of the pandemic, wearing face masks became one of the most prominent interventions against COVID-19 Reference Kreuter, Stewart, Garcia, Li and O’Brien1 . In the absence of protective immunity, many countries made mask wearing mandatory for the general population in crowded areas where social distancing could not be maintained (such as public transport), and recommended wearing them even outdoors Reference Czypionka, Greenhalgh, Bassler and Bryant2 . Before the COVID-19 pandemic, wearing a medical face mask was recommended for healthcare professionals (HCPs) when caring for patients with respiratory symptoms as part of standard precaution measures and for patients with respiratory symptoms as part of the respiratory etiquette. Transmission-based precautions were also recommended for HCPs in close contact with symptomatic patients as part of droplet precautions.
Early in the pandemic, transmission of SARS-CoV-2 by asymptomatic and presymptomatic COVID-19 cases appeared critical in the epidemiology and control of the disease Reference He, Lau and Wu3 . Universal face masking in hospitals is usually defined as the requirement to wear a medical or surgical mask by all staff (clinical and nonclinical), patients, and visitors at any time. In the early phase of the pandemic, universal masking was part of guidelines on respiratory protection, issued by leading organizations in infection prevention and control (IPC) Reference Birgand, Mutters and Otter4 . Following Hong Kong, Singapore, and other regions of Asia, the United States and Germany introduced universal masking in March 2020 as part of a range of non-pharmaceutical measures to control the spread of SARS-CoV-2 in healthcare settings. This change was unprecedented since, prior to the COVID-19 pandemic, studies had not shown an advantage of masks to prevent acquiring or spreading of respiratory pathogens in HCPs by the use of medical face masks. Three years later, the lower impact of COVID-19 on morbidity and mortality, fatigue of HCPs to wear personal protective equipment (PPE), cost, and ecological considerations have challenged the benefits of universal masking, and called for discussions on de-implementation.
In this context of evolving knowledge and recommendations, we reflected on the factors to consider de-escalating universal masking in healthcare centers.
Within-hospital SARS-CoV-2 risk of transmission
Our understanding of within-hospital transmission of respiratory viruses remains imperfect, despite increased attention over the last three years. Using viral genomics to investigate sources of SARS-CoV-2 transmission in HCPs, the majority of infections (57.9%) could not be linked to a patient or coworker Reference Braun, Moreno and Buys5 . Only 10.5% and 4.2% of infections were traced to a coworker or a patient, respectively. A UK study combined viral genomic and epidemiological data in 2181 patients and HCPs Reference Lindsey, Villabona-Arenas and Campbell6 . During the first wave with an increasingly strict masking policy (“when in contact with suspected cases”, followed by “all patient contact”, and finally by “universal masking”), transmissions were staff-to-staff in 31.6%, patient-to-patient in 27.1%, patient-to-staff in 25.5%, and staff-to-patient in 15.5%. During the second wave with universal masking from the beginning, transmissions were patient-to-patient in 52.1%, patient-to-staff in 21.2%, staff-to-patient in 13.5%, and staff-to-staff in 12.9%. A staff member was identified as the index case in 50.6% of the transmission chains in the first wave but only in 31.3% in the second wave. The authors concluded that intensified control measures during the pandemic likely reduced transmission between healthcare workers but not between patients. The analysis of data from 11,290 patients admitted to shared rooms revealed that almost 40% of exposed patients were getting infected Reference Karan, Klompas and Tucker7 .
Current evidence on the effectiveness of universal masking to prevent the transmission of COVID-19 and other respiratory viral infections
The effectiveness of universal masking on the transmission of respiratory viruses was studied already before the pandemic. Wearing a medical mask or N95 respirator throughout work shifts by HCPs significantly reduced self-reported clinical respiratory illness (risk ratio [RR] = 0.59; 95% CI: 0.46–0.77) and influenza-like illness (RR = 0.34; 95% CI: 0.14–0.82) Reference Offeddu, Yung, Low and Tam8 . Systematic masking of all individuals in direct contact with stem cell transplant recipients significantly reduced infections due to respiratory infections in patients Reference Partridge, Sori and Green9 . A universal masking policy in a neonatal unit reduced the number of infections due to respiratory viruses from 38 during 2011–2015 to 5 during 2017–2019, with respiratory syncytial viruses (RSV), parainfluenza viruses, and rhinoviruses representing two-thirds of the cases Reference Tong, Yung and Chiew10 .
In March 2020, a large US hospital implemented a prevention strategy combining systematic testing of symptomatic HCPs and universal masking for both HCPs and patients Reference Wang, Ferro, Zhou, Hashimoto and Bhatt11 . Before universal masking, the SARS-CoV-2 positivity ratio increased exponentially from 0% to 21.3%, with an average increase of 1.2% per day. During universal masking by HCPs and patients, the positivity rate decreased linearly from 14.7% to 11.5%, with an average decline of 0.5% per day. Contact tracing of 226 patients exposed to HCPs with confirmed COVID-19 identified only one possible transmission by an unmasked HCP prior to universal masking Reference Baker, Fiumara and Rhee12 . In the same study population, only 1 of 12 hospitalized patients testing positive on hospital day 3 or later was considered to have hospital-acquired COVID-19 Reference Rhee, Baker and Vaidya13 . The authors concluded that there was no convincing evidence of in-hospital transmission after the implementation of universal masking for both staff and patients. During the first winter season of the COVID-19 pandemic, Europe and the US experienced a quasi-absence of seasonal influenza Reference Adlhoch, Mook and Lamb14 . Epidemiological investigations and ecological studies provided convincing evidence that universal non-pharmaceutical interventions in the general population, including universal masking, were associated with relevant reductions of non-COVID-19 RVIs Reference Brooks and Butler15 . The impact of universal masking on the transmission of COVID-19 and non-COVID-19 RVIs was confirmed by other studies Reference Seidelman, Smith, Akinboyo and Lewis16–Reference Temkin, Schwaber and Vaturi20 .
Together, these findings suggest that universal masking impacts on overall hospital transmission of respiratory viruses, particularly during periods of high community prevalence Reference Richterman, Meyerowitz and Cevik21 . However, most of these studies used a before-after design, with limited control for confounders and without sensitivity analysis on time periods.
Factors to consider for de-escalating or maintaining universal masking in healthcare centers
Lessons learned in healthcare centers during the COVID-19 pandemic, and particularly the evolving understanding of respiratory virus transmission, should drive the way medical face masks are used in the future. Importantly, universal masking is only one measure to reduce the risk of transmission of respiratory viruses among other non-pharmaceutical interventions such as hand hygiene, room ventilation, testing strategies, distancing, and patient/visitor restrictions. Some factors support de-escalating universal masking but others are in favor of maintaining it (Table 1).
Table 1. Factors for and against De-escalating or Maintaining Universal Masking in Healthcare Centers
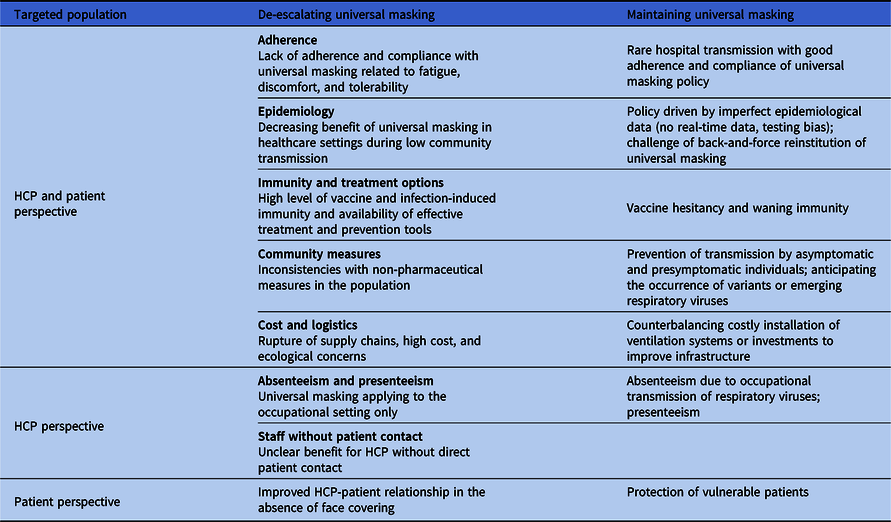
Note. HCPs: healthcare professionals.
Provider’s adherence and compliance with universal masking
High compliance and correct use of masks are important to ensure the effectiveness of universal masking. Experiments simulating the transmission of SARS-CoV-2 between two individuals demonstrated that source control alone is more effective than protection of the exposed individual alone, but that protection is most effective when both sides wear a mask, which makes the case of universal masking Reference Cheng, Ma and Witt22 . The main factor affecting the risk of transmission in the universal masking scenario is the leakage between the mask and the face Reference Bagheri, Thiede, Hejazi, Schlenczek and Bodenschatz23 . Correct donning of face masks was 88% and 70% in COVID-19 and non-COVID-19 wards of a German University hospital, respectively Reference Neuwirth, Mattner and Otchwemah24 . Although the COVID-19 wards performed better, deficits of correct use of respirators were important. During a 4-week period after the implementation of a mandated universal masking policy in a tertiary-care center in the US, the median weekly face-mask compliance was 82.2% (range: 80.8–84.4%) Reference Datta, Glenn and Pellegrino25 . Compliance increased to 92.6% (84.6%-97.9%) after implementing audits, passive feedback, active discussion, and better communication by leadership.
Adherence with face mask use by HCPs relies on organizational (communication, support from managers, workplace culture, training, and access to face masks) and individual factors (trust in face masks, desire to deliver good patient care, and feelings of safety from exposure to COVID-19 with and without vaccination) Reference Houghton, Meskell and Delaney26 . In a survey among 1,109 HCPs from an academic medical center with universal masking, only 33% of participants reported face masks being comfortable Reference Alzunitan, Perencevich and Edmond27 . The main reported disadvantages of face mask wearing were breathing difficulties (80%), heat sensation (68%), and skin irritation (46.2%). In addition, only 17.1% felt that speech was sufficiently audible by others. During observations performed across the vaccination campaigns, HCPs reported feeling safe from exposure to COVID-19 when being around their colleagues Reference O’Donohue, Fletcher-Gutowski, Sidhu, Verma, Phillips and Misra28 . Concerns about the negative impact of face masks on the physician-patient relationship were raised repeatedly. A study in surgery reported that patients appreciated to see the face of their surgeons when using transparent face masks, improving communication and trust, and giving a perception of increased empathy Reference Kratzke, Rosenbaum, Cox, Ollila and Kapadia29 .
Impact of community transmission on the efficacy of universal masking in healthcare centers
A point of attention is the level of exposure during both occupational and nonoccupational activities. Nonoccupational exposure plays a critical role in the risk of COVID-19 among HCPs. A meta-analysis showed a higher risk of COVID-19 for HCPs exposed outside the workplace (14%–32%) in comparison with exposure inside (6%–13%) Reference Gholami, Fawad and Shadan30 . Using survey data on self-reported risk factors, staff with confirmed household contacts were at highest risk of COVID-19 (adjusted Odds Ratio [aOR] 4.82, 95% CI: 3.45–6.72) Reference Eyre, Lumley and O’Donnell31 . The authors concluded that the moments most at risk for HCPs may be outside the healthcare system (e.g., during home-to-work transport). A large case-control study in France identified that infected persons outside work are much more likely to play a role in SARS-CoV-2 transmission (aOR 19.9, 95% CI: 12.4–31.9), compared to infected colleagues (aOR 2.26, 95% CI: 1.53–3.33) or COVID-19 patients (aOR 2.37, 95% CI: 1.66–3.40) Reference Belan, Charmet and Schaeffer32 . HCPs alternate between not wearing a mask (e.g., at home or during meals) and wearing a mask (e.g., during commute or at work), with varying degrees of compliance and exposure toward community transmission. During phases of high community transmission, the risk of infectious staff coming to work is high. Consequently, the benefit of the universal masking strategy in healthcare centers depends on the number of infected staff coming to work and also on the degree of compliance with mask-wearing by HCPs during their daily activities.
Protection of vulnerable and non-vulnerable patients
Patients in healthcare facilities are often vulnerable due to their age, comorbidities, and immunosuppression. Transmission from individuals with COVID-19 (patients, staff members, or visitors) is a relevant risk, especially during high prevalence of COVID-19 in the community. Before August 1, 2020, 11.3% (95% CI: 11.1–11.6) of hospitalized patients with COVID-19 in 314 UK hospitals got infected during hospital stay Reference Read, Green and Harrison33 . The risk of mortality was estimated to be 1.3 times higher in patients with hospital- compared to community-acquired COVID-19 (95% CI: 1.005–1.683) Reference Ponsford, Ward and Stoneham34 . Among patients, vaccination largely reduces the risk of serious COVID-19. However, the risk associated with HA-RVI persists for unvaccinated individuals, with vaccine escape variants, or with specific underlying medical conditions Reference Havers, Pham and Taylor35 . Presenteeism, defined as working while being ill, is common among HCPs, even among those who work in high-risk settings Reference Wilson, Wood and Schaecher36 . HCPs coming to work with mild symptoms potentially expose patients and staff members, sometimes leading to clusters of infections among HCPs and patients. This risk increases with the virus activity in the community. Universal masking by all staff in the hospital might have a relevant benefit during high prevalence periods.
Protection of HCPs, prevention of absenteeism, and preservation of the hospital activity
RVIs are a major driver of absenteeism in the healthcare workforce during the winter season, when demand for healthcare services also peaks Reference Gianino, Politano and Scarmozzino37 . Absenteeism decreases the quality by limiting the capacity of care and by exposing the remaining professionals to extra workload and stress Reference Gohar, Larivière and Nowrouzi-Kia38 . In a period of major staff shortages, absenteeism is a critical point to consider when reflecting on the use of PPE and recommending vaccination. Universal masking may represent a cost-effective strategy to address absenteeism, presenteeism, medical care, and mortality. However, maintaining HCPs with RVI at work with appropriate PPE in acute phases of staff shortage remains a concern.
The level of immune protection among HCPs is a critical factor to include in the evaluation of universal masking strategies. Although vaccination had an effect on transmission initially, now this effect has become limited. Still, vaccination prevents severe disease, and thus, has a benefit to the functioning of the healthcare system by shortening staff absence, allowing HCPs with mild symptoms coming to work, and keeping COVID-19 patients out of the hospital Reference Hollmeyer, Hayden, Mounts and Buchholz39 . In a study performed in Greece, vaccination of HCPs reduced absenteeism from 11.8 to 4.7 episodes per 100 HCPs, and the duration of absence from 11.9 to 6.9 days during a period of high SARS-CoV-2 activity in the community Reference Maltezou, Panagopoulos and Sourri40 . Vaccination of HCPs against SARS-CoV-2 may protect patients from acquiring COVID-19 Reference Roberts, Aniskiewicz, Choi, Pettker and Martinello41 . However, the effectiveness of current COVID-19 vaccines at preventing healthcare-associated transmission remains uncertain, with some inconsistency across studies Reference Stokel-Walker42 . The viral load (which is linked to risk of transmission) of fully vaccinated and unvaccinated individuals was similar, suggesting comparable efficiencies of SARS-CoV-2 transmission Reference Singanayagam, Hakki and Dunning43 . In other reports, fully vaccinated individuals had a shorter duration of shedding of viable virus and a lower rate of secondary transmission than partially vaccinated or unvaccinated individuals Reference Jung, Kim and Park44 .
Despite long-term recommendations for influenza vaccination of HCPs, uptake remains low in most countries Reference Jorgensen, Mereckiene, Cotter, Johansen, Tsolova and Brown45 . Regarding COVID-19 vaccination, on September 15, 2021, among 3 millions of HCPs in 2086 facilities in the US, 70.0% were fully vaccinated Reference Reses, Jones, Richardson, Cate, Walker and Shapiro46 . However, vaccine hesitancy in HCPs also occurred for COVID-19 vaccination before the winter season 2022/2023, and for the bivalent COVID-19 booster vaccine Reference Sinclair, Taylor, Weitz, Beckett and Samanez-Larkin47 . This may compromise the protection conferred at both individual and organizational levels. Offering influenza vaccination to staff translated into a 4.4% reduction in all-cause mortality in hospitals Reference Ahmed, Lindley, Allred, Weinbaum and Grohskopf48 . A modeling estimated that 100% vaccination coverage of HCPs could result in 43% reduction in the risk of infection in hospital patients and 60% reduction in nursing home patients Reference van den Dool, Bonten, Hak and Wallinga49 . According to these criteria, requiring unvaccinated HCPs to wear a mask when working in patient areas, and vaccine mandates for HCPs against COVID-19 and influenza must be discussed.
Availability, logistics, costs, and waste associated with face mask usage
The availability of good quality material, procurement, cost, and waste associated with the use of face masks are important factors to consider when deciding on universal masking as a prevention strategy. At the early stage of the pandemic, serious shortages of face masks were due to a combined increase in demand, shortage of production, and supply-chain ruptures. Production and supply-chain issues are now solved and most healthcare centers in high-income countries do not suffer from shortages anymore. However, the vulnerability of the system demands for stocking face masks in adequate storage conditions.
During the first year of the pandemic, the cost to procure PPE in hospitals in the US was estimated at more than $3 billion 50 . In 2019, hospitals spent around $7 per patient per day on PPE, $20.40 during spring 2020, and $12.45 during the height of the pandemic. Additionally, approximately 87,000 tons of PPE procured between March 2020 and November 2021 ended up as waste 51 . The COVID-19 waste story and the urgency to address sustainability also in healthcare ask for systems to reduce and manage healthcare waste including novel strategies of recycling and repurposing material.
Alternative strategies for permanent universal masking in healthcare centers
Table 2 lists advantages and disadvantages of alternative strategies to permanent universal masking. The first level is symptom-based and includes transmission-based precautions as defined by standard precaution measures and respiratory etiquette. Face masks are used by HCPs during droplet exposure and by patients with respiratory symptoms. Symptomatic HCPs should be discouraged or not allowed coming to work. This strategy is promoted by groups of IPC specialists in US Reference Shenoy, Babcock and Brust52 . Asymptomatic and presymptomatic transmission is not addressed. The second level aims to protect patients and includes face masks in direct patient contact, either for all patients or for vulnerable patients only. HCPs wear masks during any activity with the patient. Patient-to-staff and staff-to-staff transmission is not addressed. The third level is epidemiology-driven and includes universal masking when disease activity in the community is high. The main gap in this strategy is the need for epidemiological data, either in form of a sentinel network or wastewater surveillance. The strategy is also challenged by the back-and-forth institution of universal masking depending on the epidemiology, which requires significant operational flexibility. The fourth level is a seasonal strategy, defining a time period for universal masking. Adherence with this policy by HCPs may be affected in case disease activity is still low in the community. The fifth level targets patient care and includes universal masking for all HCPs during their entire shift in all areas with patient care Reference Kalu, Henderson, Weber and Haessler53 . Adherence could be a real challenge outside periods of disease activity in the community. It also would generate a significant overuse of face masks raising cost-efficiency and ecological concerns. The last level aims at maximising risk reduction and includes permanent mask use by HCP, patients and visitors.
Table 2. Example of Advantages and Disadvantages of Alternative Strategies to Permanent Universal Masking in Healthcare Centers
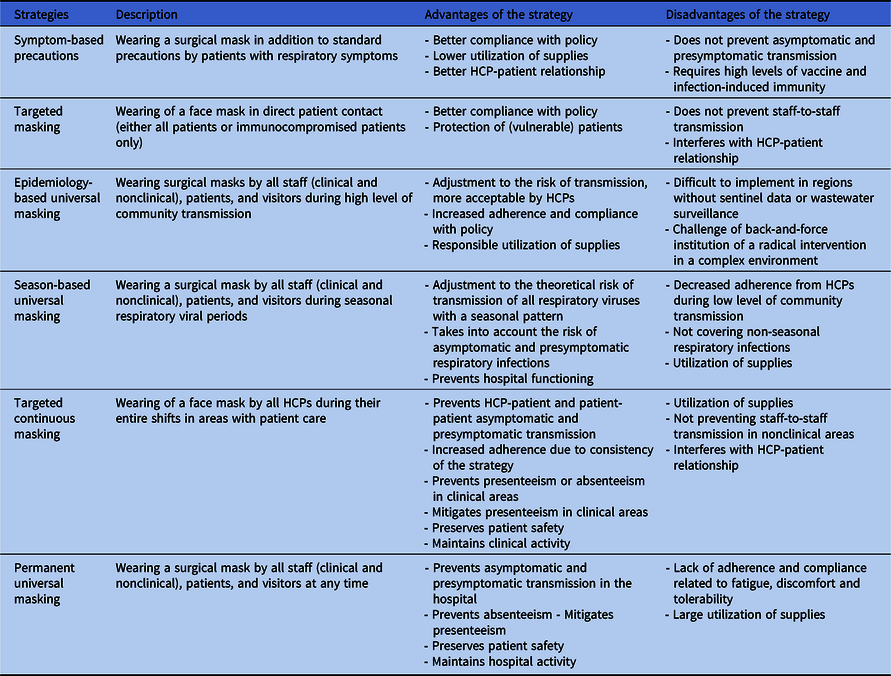
Note. HCPs: healthcare professionals.
Current masking strategies adopted in national and international guidelines
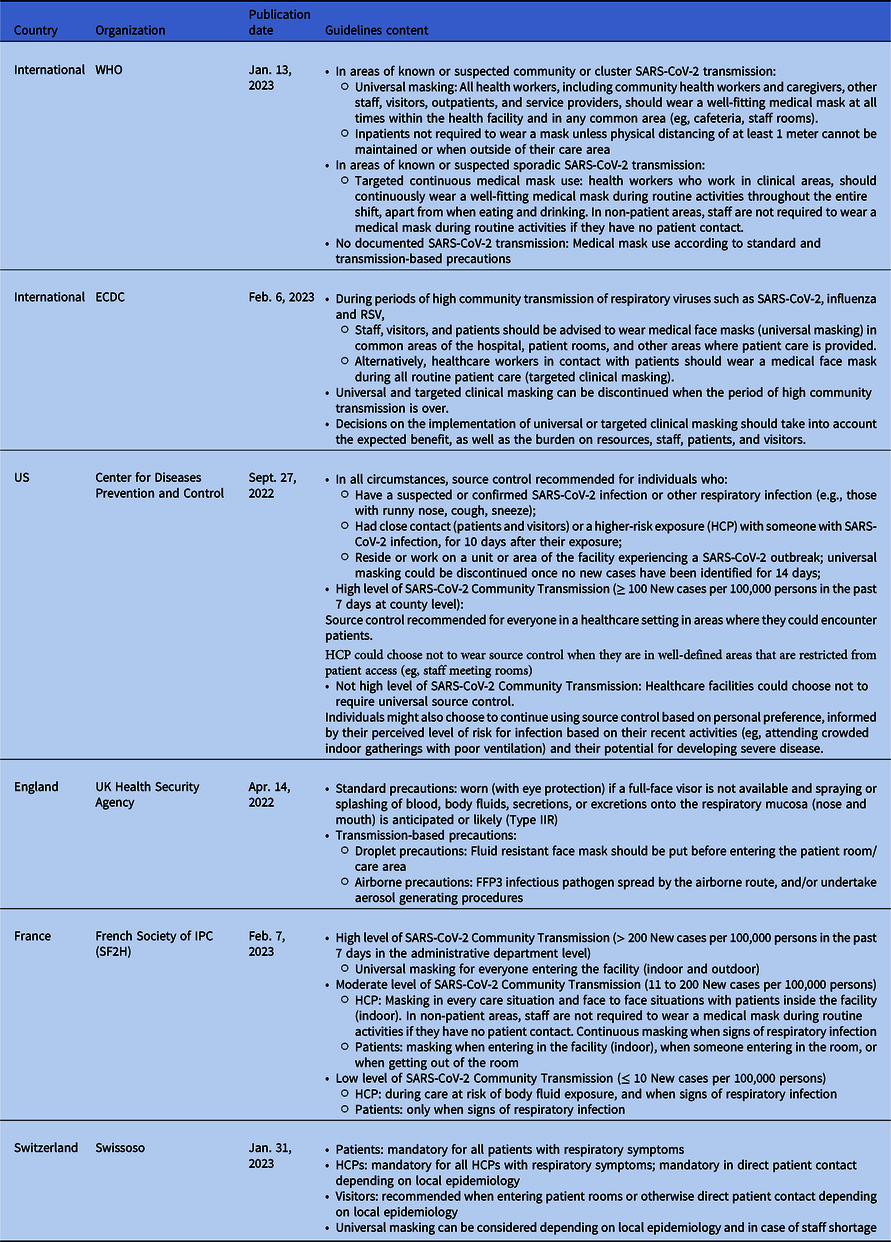
The US CDC adopted an epidemiology-based, targeted continuous masking strategy on September 23, 2022 (Table 3). Healthcare facilities in areas with low levels of community spread can “choose not to mandate” all clinicians, patients, and visitors to wear a face mask. Even if masking is not universally required, if a provider works in an area experiencing a COVID-19 outbreak, or if HCPs care for immunocompromised patients, they should wear a mask. When transmission levels are high, masking is recommended for everyone in a health care setting in areas with patients, but HCPs can choose not to wear masks when they are in “well-defined areas” that are restricted from patient access, like staff meeting rooms. The US CDC guidance shift created a controversy in the IPC world with professional societies urging to maintain mandatory mask requirement policies for healthcare employees in all patient care areas 54 . Among 44 healthcare epidemiologists in the US participating in a survey, 33 (97%) reported that their facility was maintaining universal masking despite CDC guidance, mostly (90%) for preventing non-SARS-CoV-2 seasonal viruses and to maintain staffing capacities (73%) Reference Snyder, Passaretti and Stevens55 .
Table 3. Description of Masking Strategies for Source Control Adopted in National and International Guidelines
Note. RSV, respiratory syncytial virus; HCP, healthcare professionals.
On January 13, 2023, WHO released an update of the living IPC guideline taking into account the evolving epidemiological trends for COVID-19. The masking strategy was adapted to the SARS-CoV-2 transmission with three levels 56 . When any SARS-CoV-2 transmission is documented in the area, medical masks can be used according to standard and transmission-based precautions. In areas of known or suspected sporadic SARS-CoV-2 transmission, the strategy is based on a targeted continuous medical mask use. In areas of known or suspected community or cluster SARS-CoV-2 transmission, universal masking applies for HCPs but not for patients unless physical distancing of at least one meter cannot be maintained.
The ECDC guidelines, published on February 6, 2023, recommend universal masking for staff, visitors, and patients in common areas of the hospital, patient rooms, and other areas where patient care is provided, during periods of high community transmission of respiratory viruses such as SARS-CoV-2, influenza, and RSV 57 . Alternatively, targeted clinical masking is recommended for HCP in contact with patients during all routine patient care.
In Switzerland, the strategy is epidemiologically-driven with mandatory masking for all HCPs in direct patient contact or universal masking depending on local epidemiology and in case of staff shortage 58 .
Since February 7, 2023, SARS-CoV-2 community transmission is guiding the masking policy in France 59 . Universal masking is mandatory for every individual entering the facility (indoor and outdoor) when >200 new cases per 100,000 population have been reported during the past seven days in the administrative department. During phases of moderate transmission (11 to 200 new cases per 100,000 population), a targeted continuous masking is adopted in patients-areas, with a flexibility to not wear the mask in non-patient areas. Standard and transmission-based precautions are applied ≤ 10 new cases per 100,000 population. In England, since May 27, 2022, the national IPC manual reverts back from universal masking to standard and transmissions-based precautions 60 .
Conclusion
In conclusion, three years after the emergence of the COVID-19 pandemic and the improved understanding of the transmission of RVI, the IPC community should consider revisiting the current masking policies. Specifically, de-escalation of the universal masking strategy implemented in healthcare centers since the first wave of COVID-19 must be revisited. The current strategies by most national and international organizations are based on the level of community transmission of SARS-CoV-2. Actions are now required to establish a list of the most burdensome RVI to consider, implement reliable local surveillance of RVI, and define thresholds for activating masking strategies. Considering the epidemiological context (measured via existing sentinel networks or innovative ways such as wastewater analysis), and if not available considering a time period (winter season) may guide to three gradual levels of masking: (i) standard and transmission-based precautions and respiratory etiquette, (ii) consistent face mask wearing when in direct contact with patients, and (iii) universal masking. Cost-effectiveness analyses are warranted in the coming years to refine recommendations. The face mask is only one element among other preventive measures in healthcare settings such as staff and patient immunization, and efficient ventilation.
Acknowledgments
None.
Author contribution
All authors contributed to the thematic, literature review and writing. The views expressed in this publication are those of the author(s) and not necessarily those of their organization.
Financial support
None
Competing interests
None to declare from all authors.
Ethical standard
Not applicable.







