No CrossRef data available.
Article contents
The crystal structure of charmarite – the first case of a 11 × 11 Å superstructure mesh in layered double hydroxides
Published online by Cambridge University Press: 08 March 2024
Abstract
Charmarite, Mn4Al2(OH)12CO3⋅3H2O, is a hydrotalcite supergroup member (or layered double hydroxide, LDH) with a previously unknown crystal structure and a Mn2+-analogue of quintinite (commonly erroneously reported as ‘2:1 hydrotalcite’). The single-crystal X-ray diffraction (XRD) data were obtained from the specimen from Mont Saint-Hilaire, Québec, Canada and are best processed in the space group P$\bar{3}$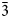 , a = 10.9630(4), c = 15.0732(5) Å and V = 1568.89(12) Å3. The crystal structure has been solved by direct methods and refined to R1 = 0.0750 for 3801 unique reflections with Fo > 2σ(Fo). The charmarite structure has long-range periodicity in the xy plane due to $2\sqrt 3$
, a = 10.9630(4), c = 15.0732(5) Å and V = 1568.89(12) Å3. The crystal structure has been solved by direct methods and refined to R1 = 0.0750 for 3801 unique reflections with Fo > 2σ(Fo). The charmarite structure has long-range periodicity in the xy plane due to $2\sqrt 3$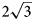 a’ × $2\sqrt 3$
a’ × $2\sqrt 3$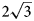 a’ scheme (or 11 × 11 Å) determined for LDHs for the first time (where a’ is a subcell parameter ≈ 3.2 Å). This periodicity is produced by the combination of two superstructures formed by: (1) Mn2+ and Al3+ ordering in the metal-hydroxide layers [Mn4Al2(OH)12]2+ according to the $\sqrt 3$
a’ scheme (or 11 × 11 Å) determined for LDHs for the first time (where a’ is a subcell parameter ≈ 3.2 Å). This periodicity is produced by the combination of two superstructures formed by: (1) Mn2+ and Al3+ ordering in the metal-hydroxide layers [Mn4Al2(OH)12]2+ according to the $\sqrt 3$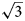 a’ × $\sqrt 3$
a’ × $\sqrt 3$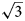 a’ pattern and (2) the (CO3)2– ordering according to the 2a’ × 2a’ pattern in the [CO3(H2O)3]2– interlayer sheet in order to avoid close contacts between adjacent carbonate groups. The $2\sqrt 3$
a’ pattern and (2) the (CO3)2– ordering according to the 2a’ × 2a’ pattern in the [CO3(H2O)3]2– interlayer sheet in order to avoid close contacts between adjacent carbonate groups. The $2\sqrt 3$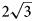 a’ × $2\sqrt 3$
a’ × $2\sqrt 3$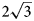 a’ superstructure is an example of the adaptability of the components of the interlayer space to the charge distribution of the metal-hydroxyl layers. The Mn2+ and Al3+ cations have a large difference in size, which apparently leads to the considerable degree of their order as di- and trivalent cations resulting in a higher degree of statistical order of the interlayer components. Both powder and single-crystal XRD data show that the samples studied belong to the hexagonal branch of two-layer polytypes (2T or 2H) with d00n ≈ 7.57 Å. The chemical composition of the samples studied is close to the ideal formula. The Raman spectrum of charmarite is reported and the band assignment is provided.
a’ superstructure is an example of the adaptability of the components of the interlayer space to the charge distribution of the metal-hydroxyl layers. The Mn2+ and Al3+ cations have a large difference in size, which apparently leads to the considerable degree of their order as di- and trivalent cations resulting in a higher degree of statistical order of the interlayer components. Both powder and single-crystal XRD data show that the samples studied belong to the hexagonal branch of two-layer polytypes (2T or 2H) with d00n ≈ 7.57 Å. The chemical composition of the samples studied is close to the ideal formula. The Raman spectrum of charmarite is reported and the band assignment is provided.
Keywords
- Type
- Article
- Information
- Copyright
- Copyright © The Author(s), 2024. Published by Cambridge University Press on behalf of The Mineralogical Society of the United Kingdom and Ireland
Footnotes
Associate Editor: G. Diego Gatta




