62 results
Potential underreporting of treated patients using a Clostridioides difficile testing algorithm that screens with a nucleic acid amplification test
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 5 / May 2024
- Published online by Cambridge University Press:
- 25 January 2024, pp. 590-598
- Print publication:
- May 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Quantifying racial disparities in risk of invasive Staphylococcus aureus infection in Metropolitan Atlanta, Georgia, during the 2020–2021 coronavirus disease 2019 (COVID-19) pandemic
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 4 / April 2024
- Published online by Cambridge University Press:
- 27 December 2023, pp. 534-536
- Print publication:
- April 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
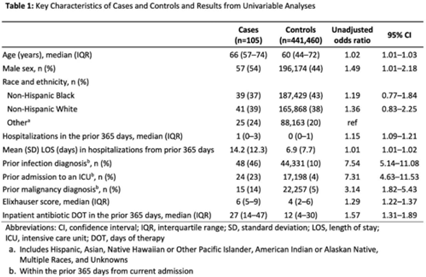
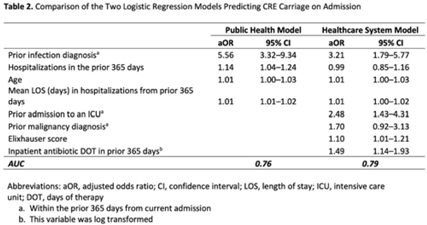
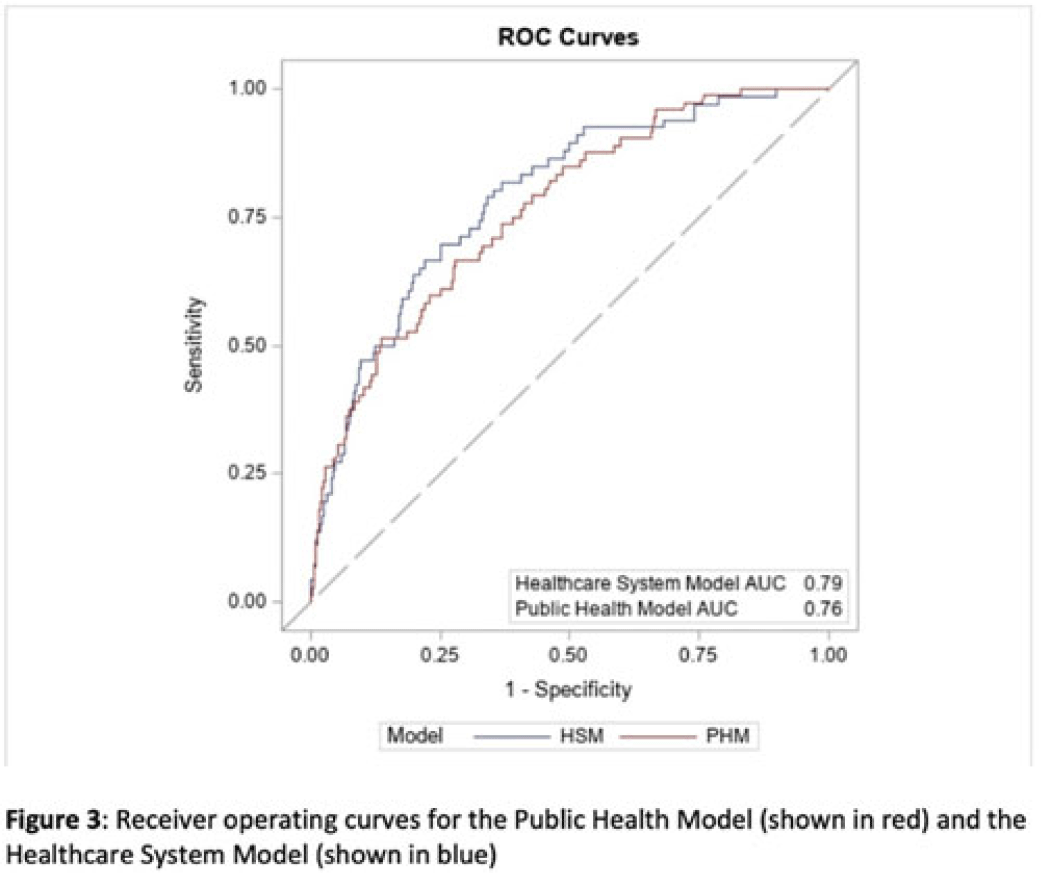
Identifying patients at high risk for carbapenem-resistant Enterobacterales carriage upon admission to acute-care hospitals
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s82-s83
-
- Article
-
- You have access
- Open access
- Export citation
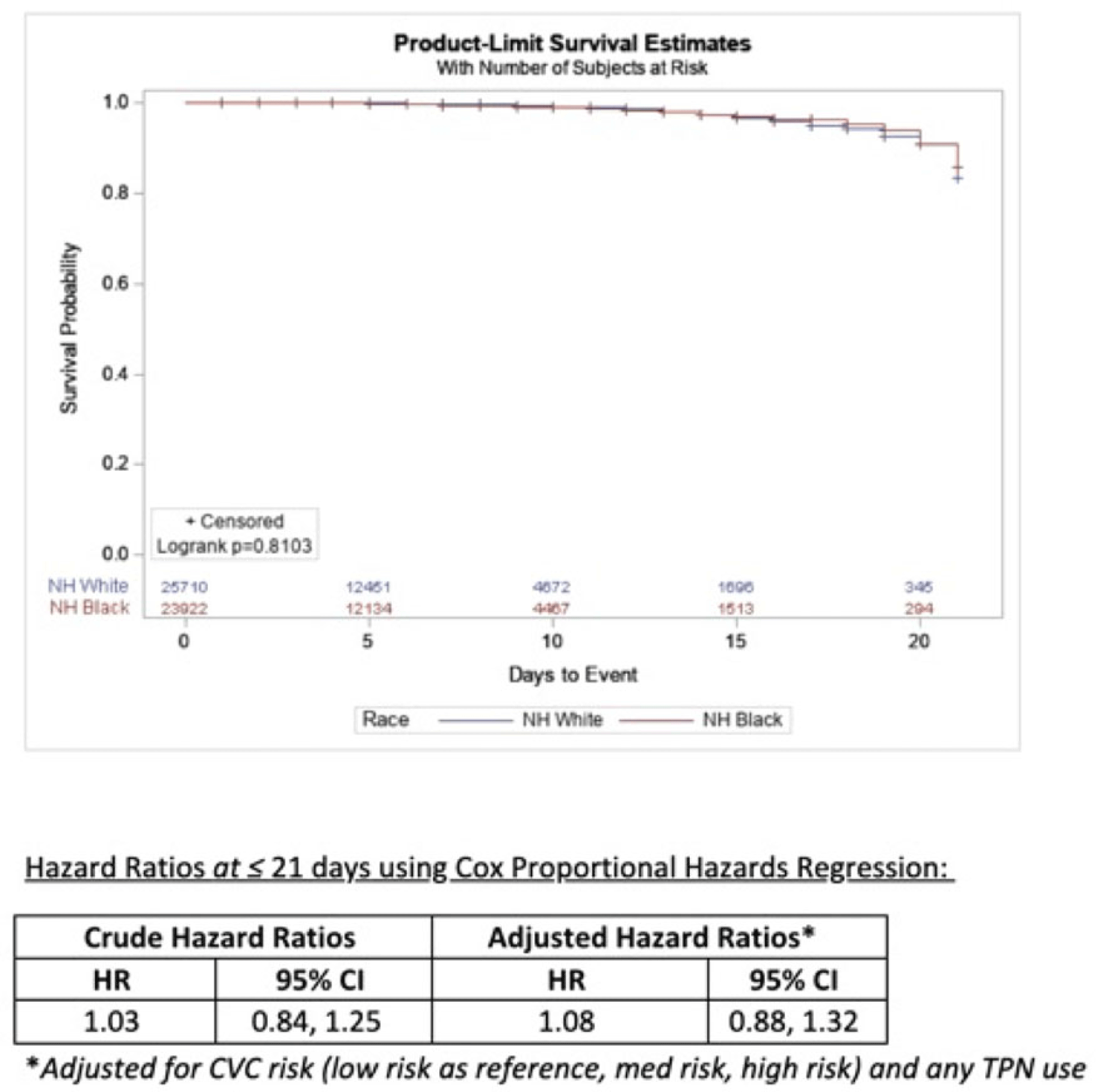
Estimating racial differences in risk for CLABSI in a large urban healthcare system
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s50-s51
-
- Article
-
- You have access
- Open access
- Export citation
Determinates of Clostridioides difficile infection (CDI) testing practices among inpatients with diarrhea at selected acute-care hospitals in Rochester, New York, and Atlanta, Georgia, 2020–2021
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 7 / July 2023
- Published online by Cambridge University Press:
- 14 September 2022, pp. 1085-1092
- Print publication:
- July 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
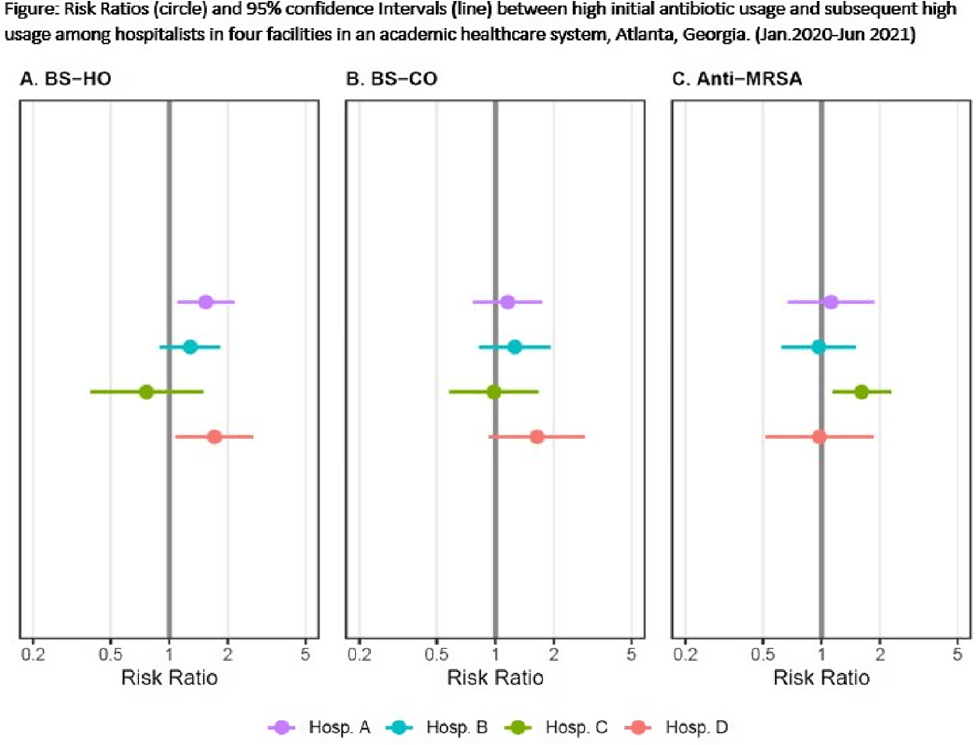
Do hospitalists who prescribe high (risk-adjusted) rates of antibiotics do so repeatedly?
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s2
-
- Article
-
- You have access
- Open access
- Export citation
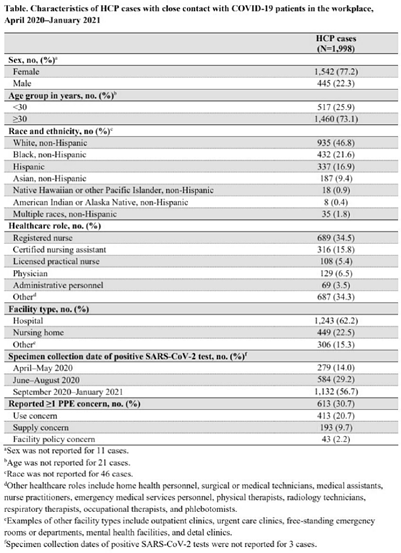
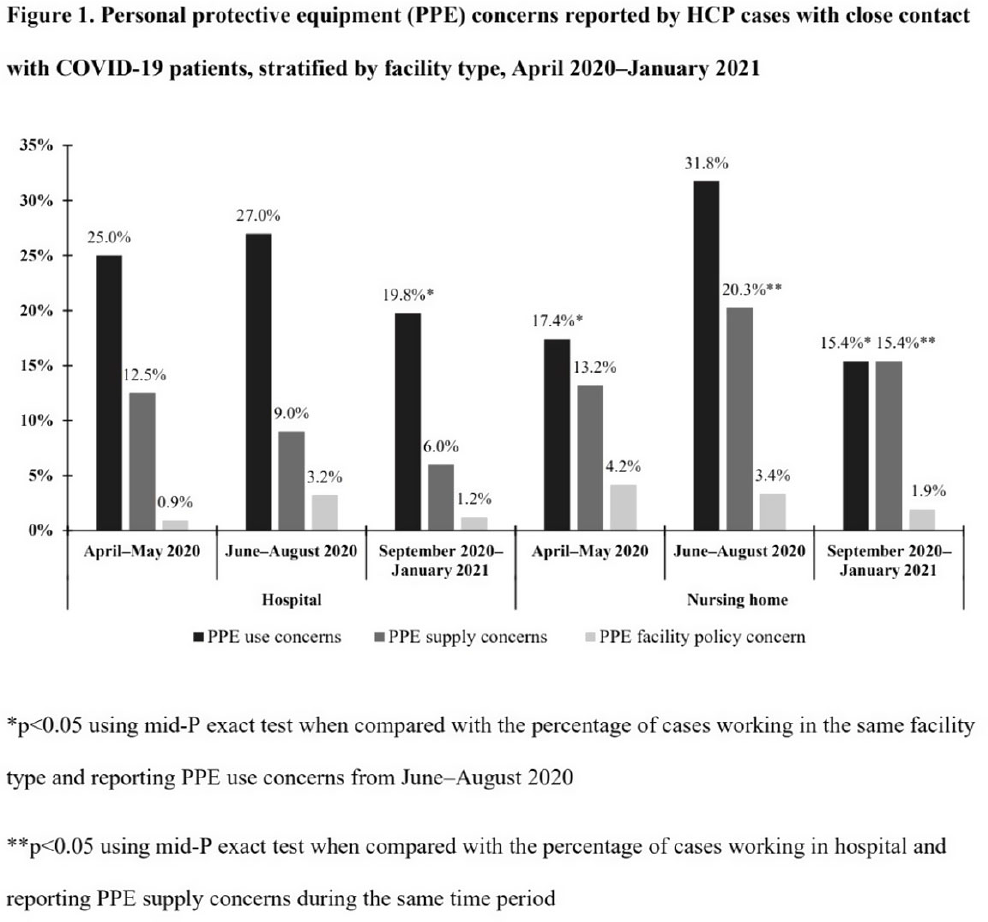
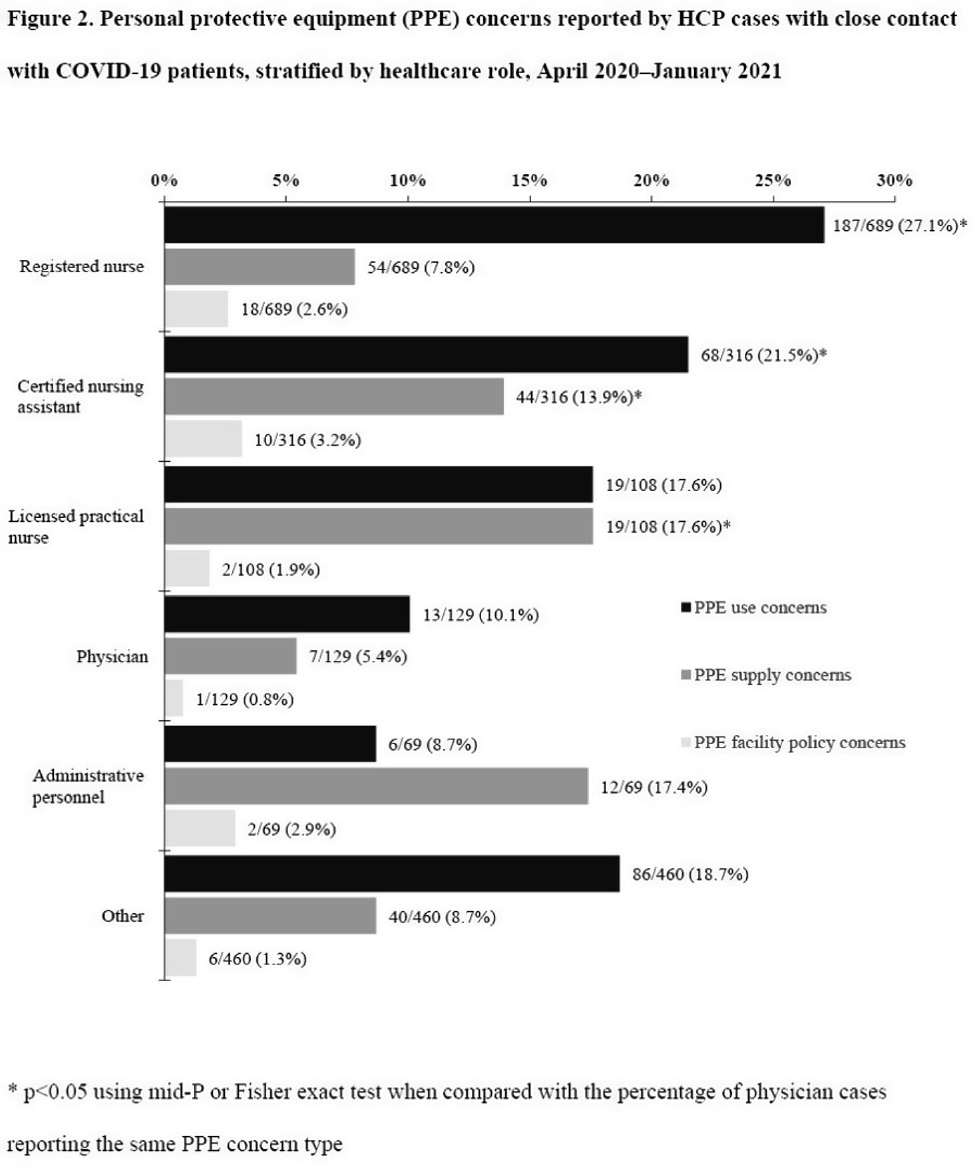
Characteristics of healthcare personnel who reported concerns related to PPE use during care of COVID-19 patients
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s8-s9
-
- Article
-
- You have access
- Open access
- Export citation
Which nursing home workers were at highest risk for SARS-CoV-2 infection during the November 2020–February 2021 winter surge of COVID-1?
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s7
-
- Article
-
- You have access
- Open access
- Export citation
Occupational risk factors for severe acute respiratory coronavirus virus 2 (SARS-CoV-2) infection among healthcare personnel: A 6-month prospective analysis of the COVID-19 Prevention in Emory Healthcare Personnel (COPE) Study
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 11 / November 2022
- Published online by Cambridge University Press:
- 14 February 2022, pp. 1664-1671
- Print publication:
- November 2022
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
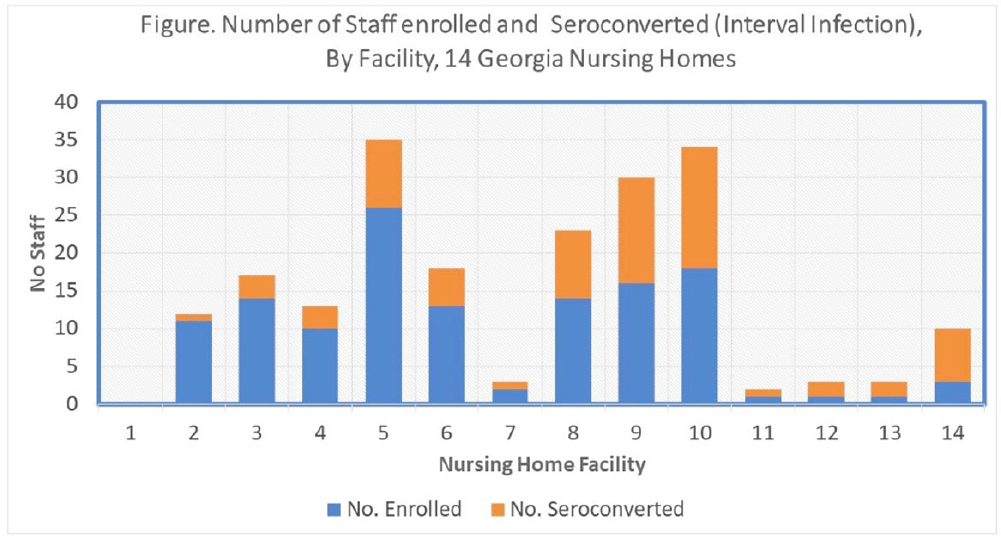
Risk factors for severe acute respiratory coronavirus virus 2 (SARS-CoV-2) seropositivity among nursing home staff
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 28 October 2021, e35
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
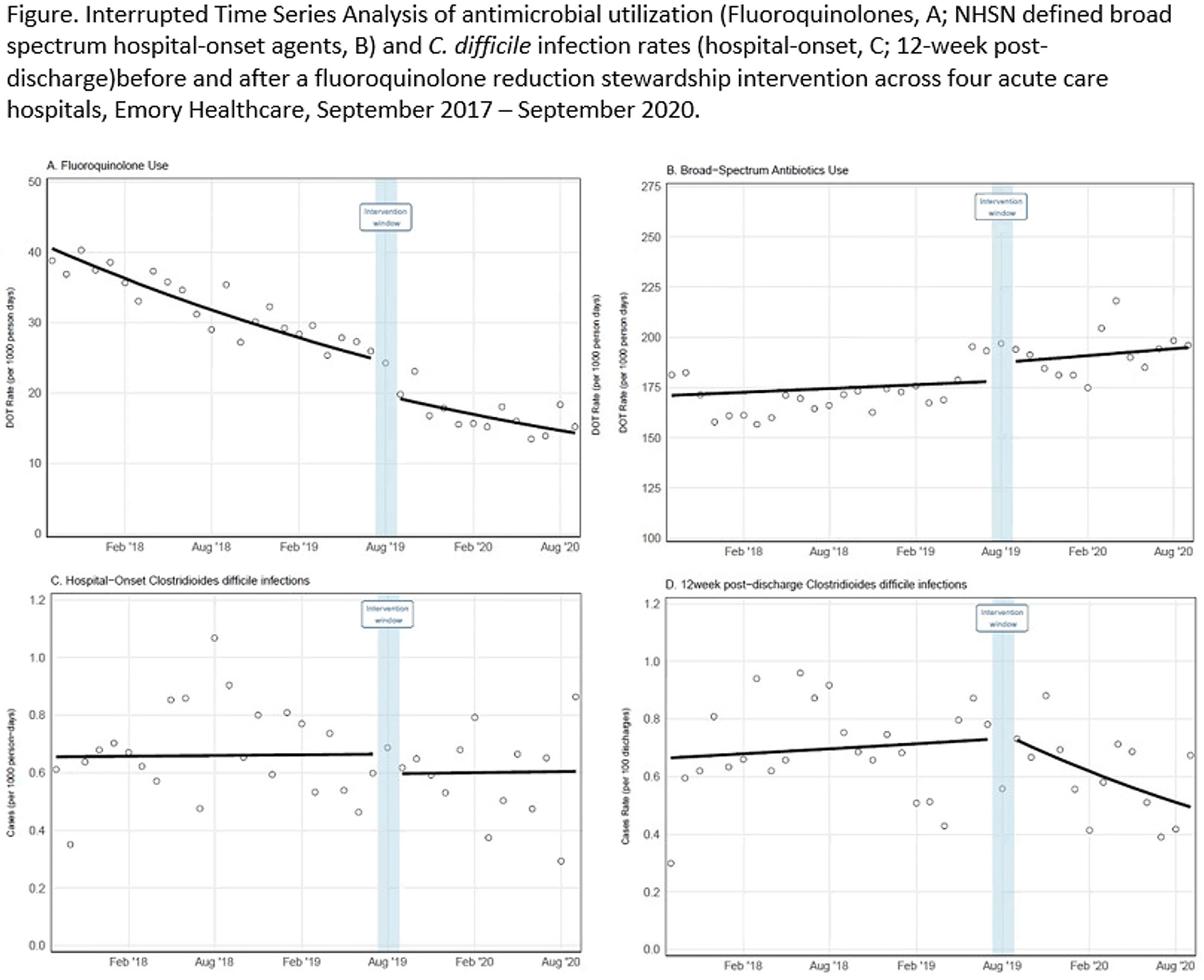
Reductions in inpatient fluoroquinolone use and postdischarge Clostridioides difficile infection (CDI) from a systemwide antimicrobial stewardship intervention
- Part of
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 22 October 2021, e32
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Reductions in Postdischarge Clostridioides difficile Infection after an Inpatient Health System Fluoroquinolone Stewardship
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s4
-
- Article
-
- You have access
- Open access
- Export citation
Reductions in positive Clostridioides difficile events reportable to National Healthcare Safety Network (NHSN) with adoption of reflex enzyme immunoassay (EIA) testing in 13 Atlanta hospitals
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 7 / July 2022
- Published online by Cambridge University Press:
- 08 July 2021, pp. 935-938
- Print publication:
- July 2022
-
- Article
- Export citation
Occupational risk factors for severe acute respiratory coronavirus virus 2 (SARS-CoV-2) infection among healthcare personnel: A cross-sectional analysis of subjects enrolled in the COVID-19 Prevention in Emory Healthcare Personnel (COPE) study
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 3 / March 2022
- Published online by Cambridge University Press:
- 09 February 2021, pp. 381-386
- Print publication:
- March 2022
-
- Article
-
- You have access
- HTML
- Export citation
A Portable, Easily Deployed Approach to Measure Healthcare Professional Contact Networks in Long-Term Care Settings
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s455
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Variation in Hospitalist-Specific Antibiotic Prescribing at Four Hospitals: A Novel Tool for Antibiotic Stewardship
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s56-s57
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Racial Differences in Incidence of Staphylococcus aureus Joint Infections in Metropolitan Atlanta, 2016–2018
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s495
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Evaluation of Care Interactions Between Healthcare Personnel and Residents in Nursing Homes Across the United States
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s36-s38
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Validation of Administrative Codes for Identification of Staphylococcus aureus Infections Among Electronic Health Data
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s507-s509
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
The Second Central Line Increases Central-Line–Associated Bloodstream Infection Risk by 80%: Implications for Inpatient Quality Reporting Programs
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s499-s500
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation