58 results
Practices to prevent central line-associated bloodstream infection: A 2021 survey of infection preventionists in US hospitals
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 24 April 2024, pp. 1-5
-
- Article
- Export citation
The influence of spirituality, religiosity, and self-care on well-being among Thai infection preventionists during the COVID-19 pandemic
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue 1 / 2024
- Published online by Cambridge University Press:
- 19 February 2024, e26
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
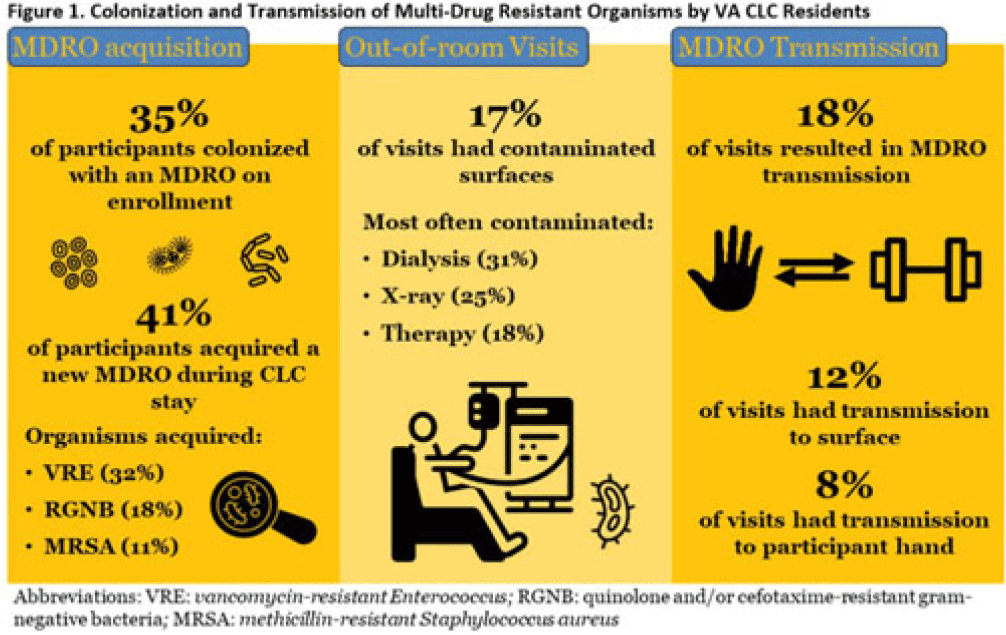
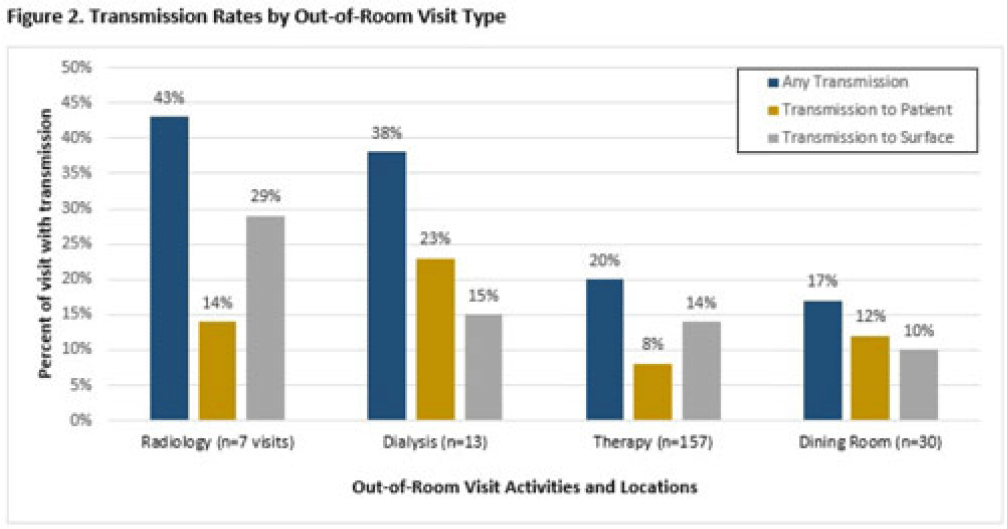
Transmission of multidrug-resistant organisms by VA CLC residents: A multisite prospective study
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s116-s117
-
- Article
-
- You have access
- Open access
- Export citation
The influence of hospital leadership support on burnout, psychological safety, and safety climate for US infection preventionists during the coronavirus disease 2019 (COVID-19) pandemic
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 3 / March 2024
- Published online by Cambridge University Press:
- 13 September 2023, pp. 310-315
- Print publication:
- March 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Strategies to prevent catheter-associated urinary tract infections in acute-care hospitals: 2022 Update
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 8 / August 2023
- Published online by Cambridge University Press:
- 25 August 2023, pp. 1209-1231
- Print publication:
- August 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Implementing strategies to prevent infections in acute-care settings
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 8 / August 2023
- Published online by Cambridge University Press:
- 11 July 2023, pp. 1232-1246
- Print publication:
- August 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
What US hospitals are doing to prevent common device-associated infections during the coronavirus disease 2019 (COVID-19) pandemic: Results from a national survey in the United States
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 12 / December 2023
- Published online by Cambridge University Press:
- 01 June 2023, pp. 1913-1919
- Print publication:
- December 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Infection prevention practices in the United States, the Netherlands, Switzerland, and Japan: Results from national surveys
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 10 / October 2021
- Published online by Cambridge University Press:
- 04 February 2021, pp. 1206-1214
- Print publication:
- October 2021
-
- Article
- Export citation
Sustainability of a program to reduce unnecessary urethral catheter use at a Veterans Affairs hospital
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 12 / December 2021
- Published online by Cambridge University Press:
- 01 February 2021, pp. 1497-1499
- Print publication:
- December 2021
-
- Article
- Export citation
Coronavirus disease 2019 (COVID-19) research agenda for healthcare epidemiology
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 2 / February 2022
- Published online by Cambridge University Press:
- 25 January 2021, pp. 156-166
- Print publication:
- February 2022
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Preventing healthcare-associated infection in Switzerland: Results of a national survey
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue 5 / May 2020
- Published online by Cambridge University Press:
- 13 April 2020, pp. 597-600
- Print publication:
- May 2020
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Use of and patient-reported complications related to midline catheters and peripherally inserted central catheters
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue 5 / May 2020
- Published online by Cambridge University Press:
- 04 March 2020, pp. 608-610
- Print publication:
- May 2020
-
- Article
- Export citation
Antibiotic stewardship teams and Clostridioides difficile practices in United States hospitals: A national survey in The Joint Commission antibiotic stewardship standard era
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue 2 / February 2020
- Published online by Cambridge University Press:
- 11 December 2019, pp. 143-148
- Print publication:
- February 2020
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Reducing unnecessary urethral catheter use in Japanese intensive care units: A multicenter interventional study
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 11 / November 2019
- Published online by Cambridge University Press:
- 27 September 2019, pp. 1272-1274
- Print publication:
- November 2019
-
- Article
- Export citation
Assessing a National Collaborative Program To Prevent Catheter-Associated Urinary Tract Infection in a Veterans Health Administration Nursing Home Cohort
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 39 / Issue 7 / July 2018
- Published online by Cambridge University Press:
- 10 May 2018, pp. 820-825
- Print publication:
- July 2018
-
- Article
- Export citation
Reducing Inappropriate Urinary Catheter Use in the Emergency Department: Comparing Two Collaborative Structures
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 39 / Issue 1 / January 2018
- Published online by Cambridge University Press:
- 18 December 2017, pp. 77-84
- Print publication:
- January 2018
-
- Article
- Export citation
National Survey of Environmental Cleaning and Disinfection in Hospitals in Thailand
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 38 / Issue 10 / October 2017
- Published online by Cambridge University Press:
- 30 August 2017, pp. 1250-1253
- Print publication:
- October 2017
-
- Article
- Export citation
Comparing Catheter-Associated Urinary Tract Infection Prevention Programs Between Veterans Affairs Nursing Homes and Non–Veterans Affairs Nursing Homes
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 38 / Issue 3 / March 2017
- Published online by Cambridge University Press:
- 05 December 2016, pp. 287-293
- Print publication:
- March 2017
-
- Article
- Export citation
Infection Prevention and Antimicrobial Stewardship Knowledge for Selected Infections Among Nursing Home Personnel
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 38 / Issue 1 / January 2017
- Published online by Cambridge University Press:
- 04 October 2016, pp. 83-88
- Print publication:
- January 2017
-
- Article
- Export citation
Response to Allen-Bridson and Pollock
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 37 / Issue 9 / September 2016
- Published online by Cambridge University Press:
- 15 July 2016, p. 1122
- Print publication:
- September 2016
-
- Article
-
- You have access
- HTML
- Export citation