15 results
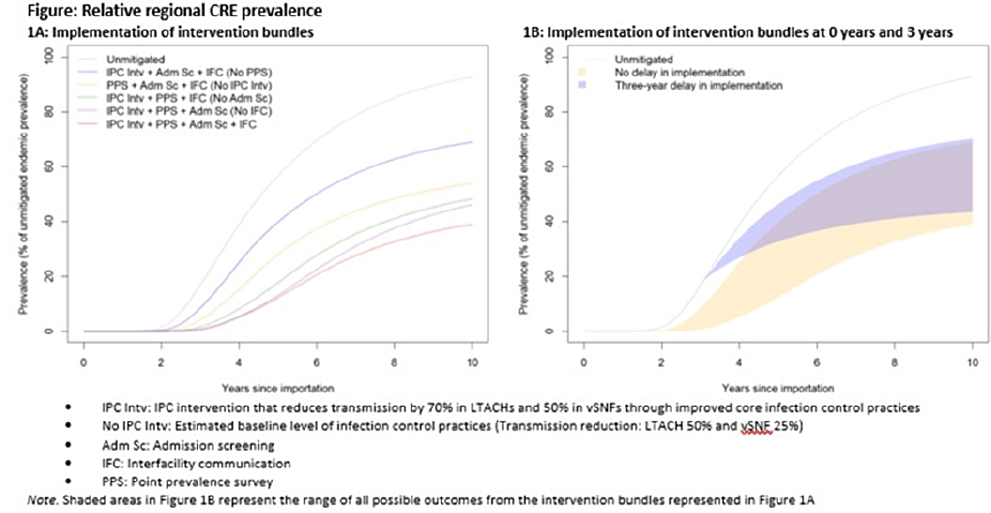
Regional impact of multidrug-resistant organism prevention bundles implemented by facility type: A modeling study
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 28 February 2024, pp. 1-8
-
- Article
- Export citation
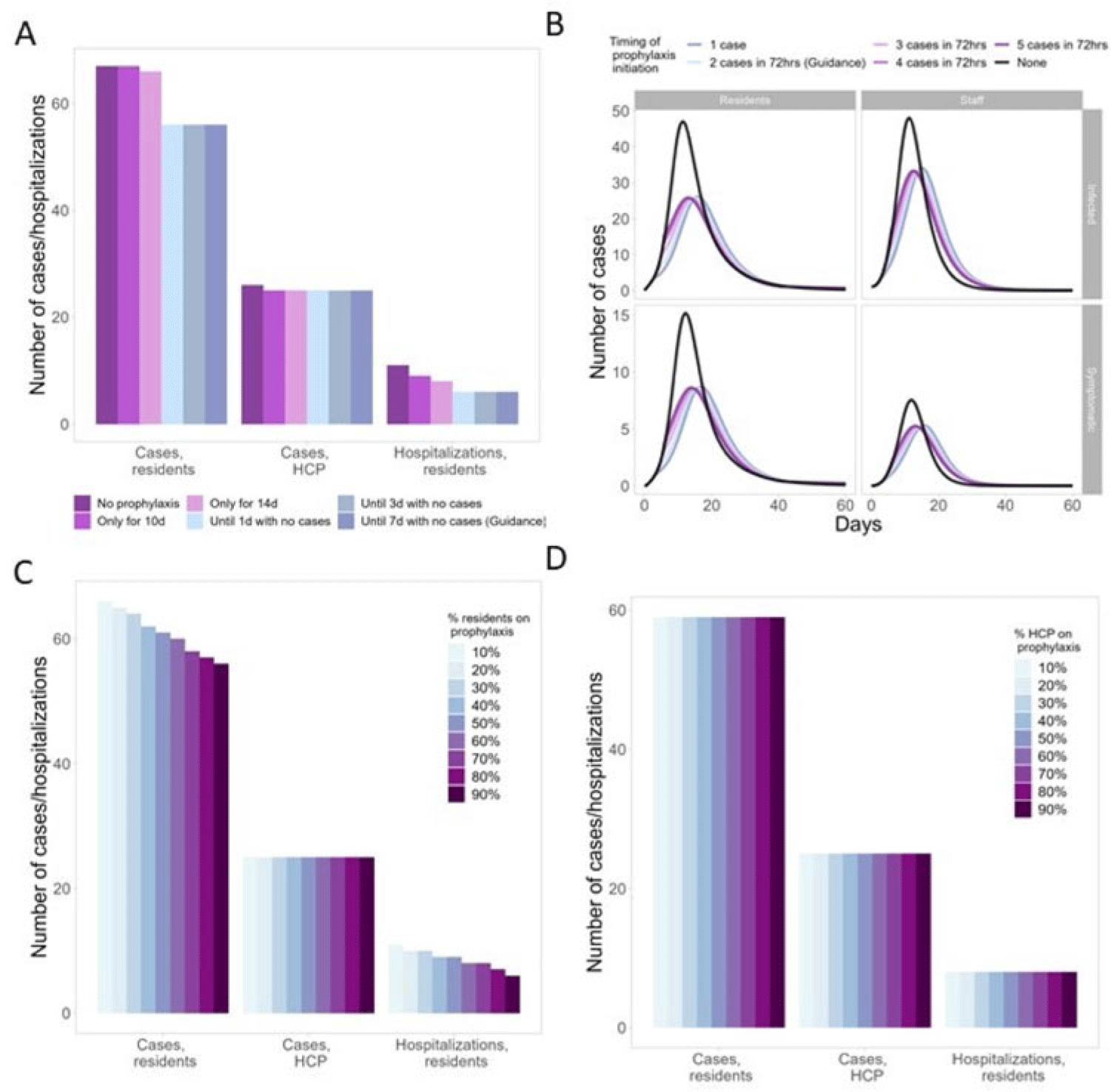
Modeling the impacts of influenza antiviral prophylaxis strategies in nursing homes
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s18-s19
-
- Article
-
- You have access
- Open access
- Export citation
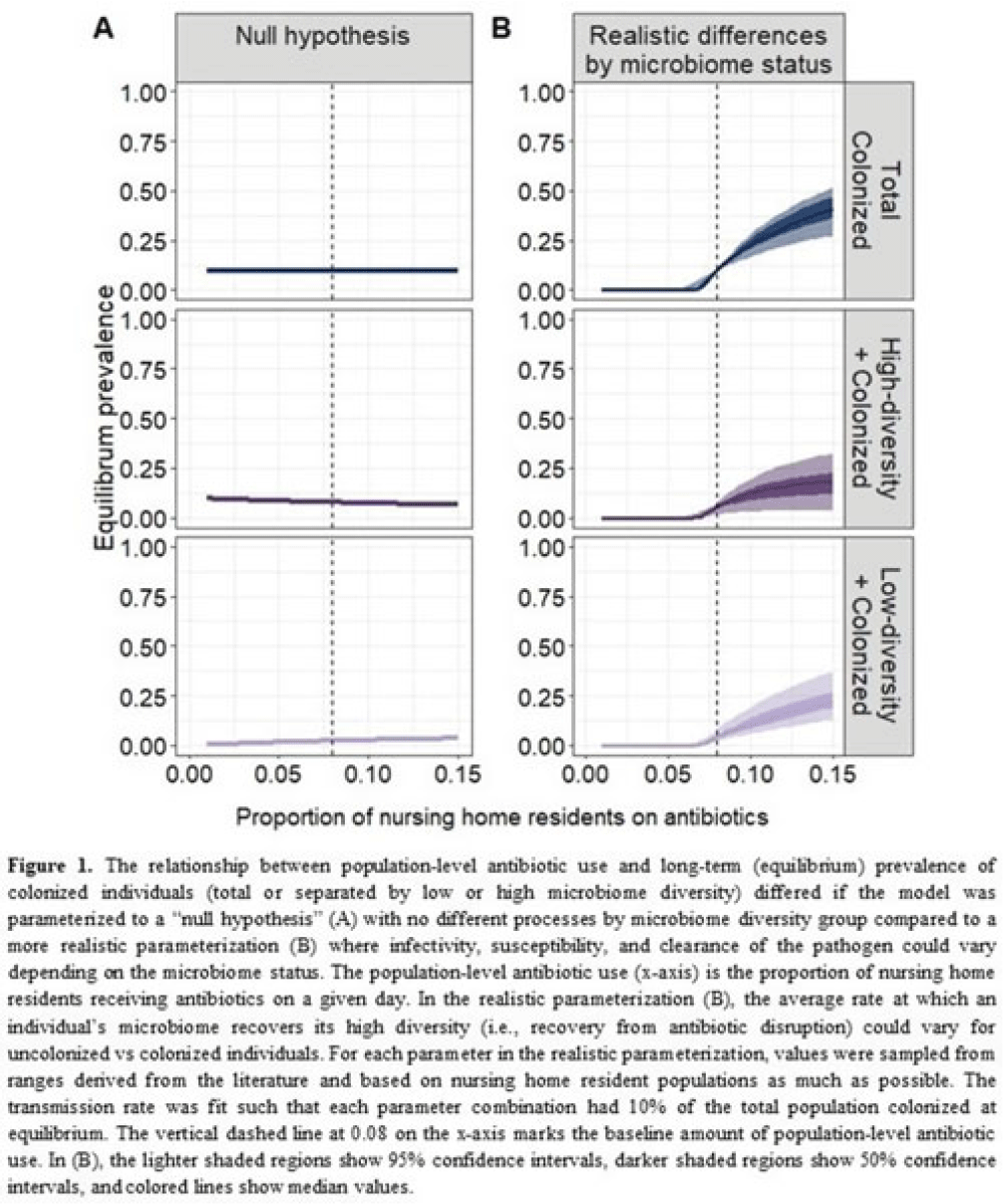
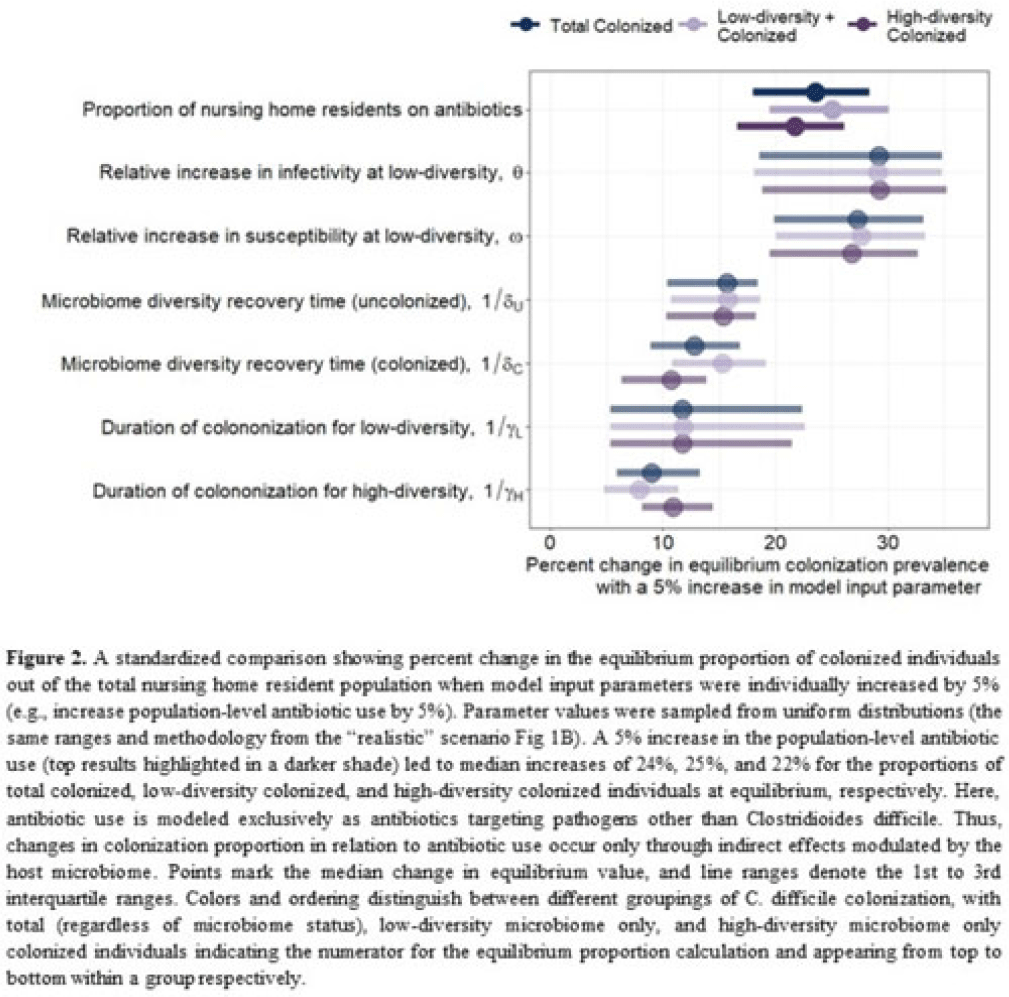
Uncovering gut microbiota-mediated indirect effects of antibiotic use on Clostridioides difficile transmission
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s104-s105
-
- Article
-
- You have access
- Open access
- Export citation
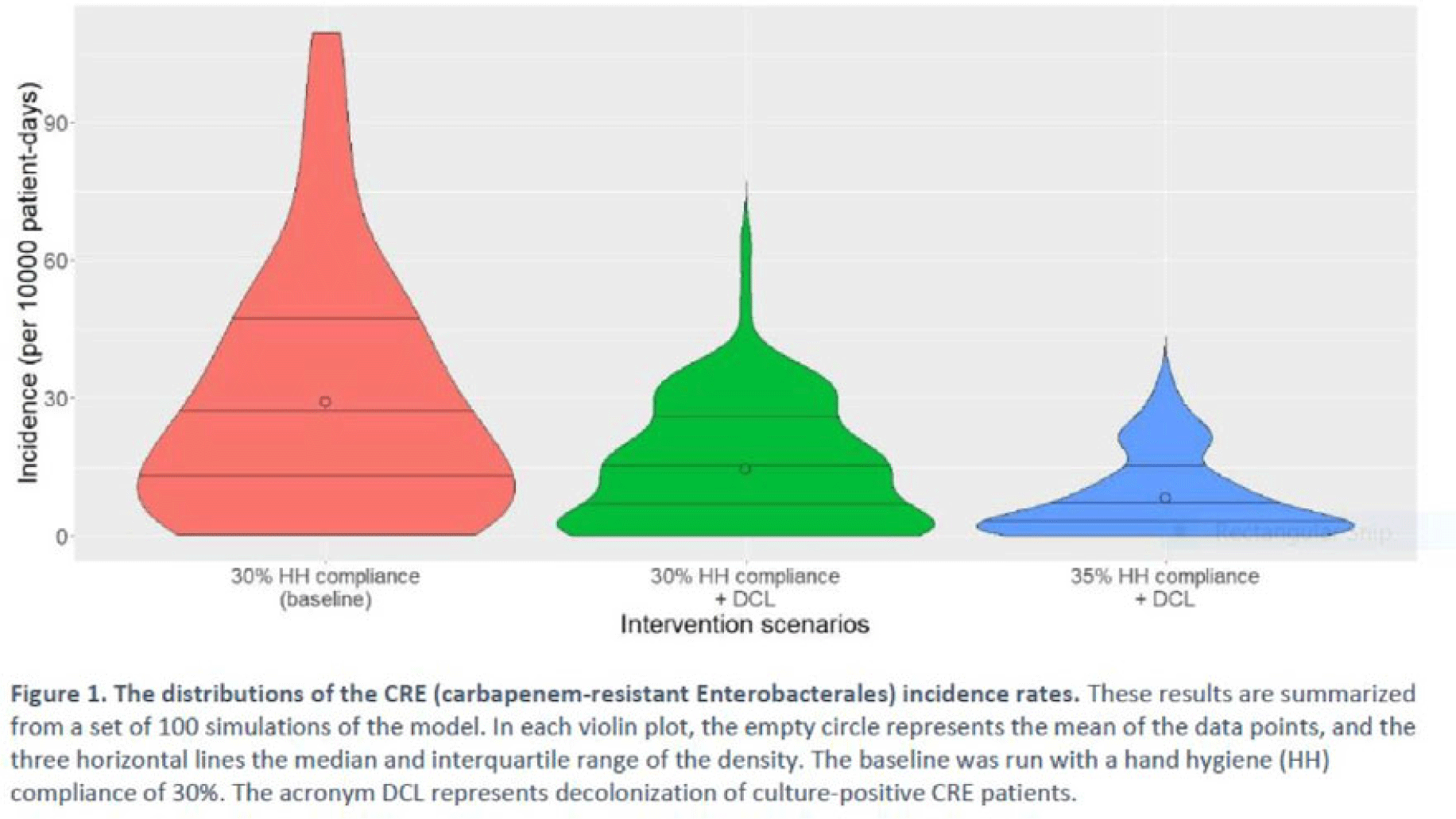
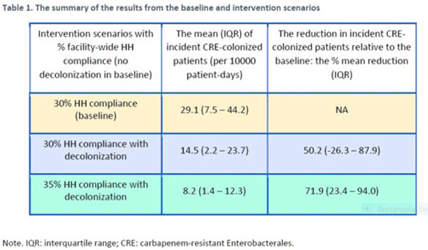
Decolonization of hospital patients may aid efforts to reduce transmission of carbapenem-resistant Enterobacterales
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s59-s60
-
- Article
-
- You have access
- Open access
- Export citation
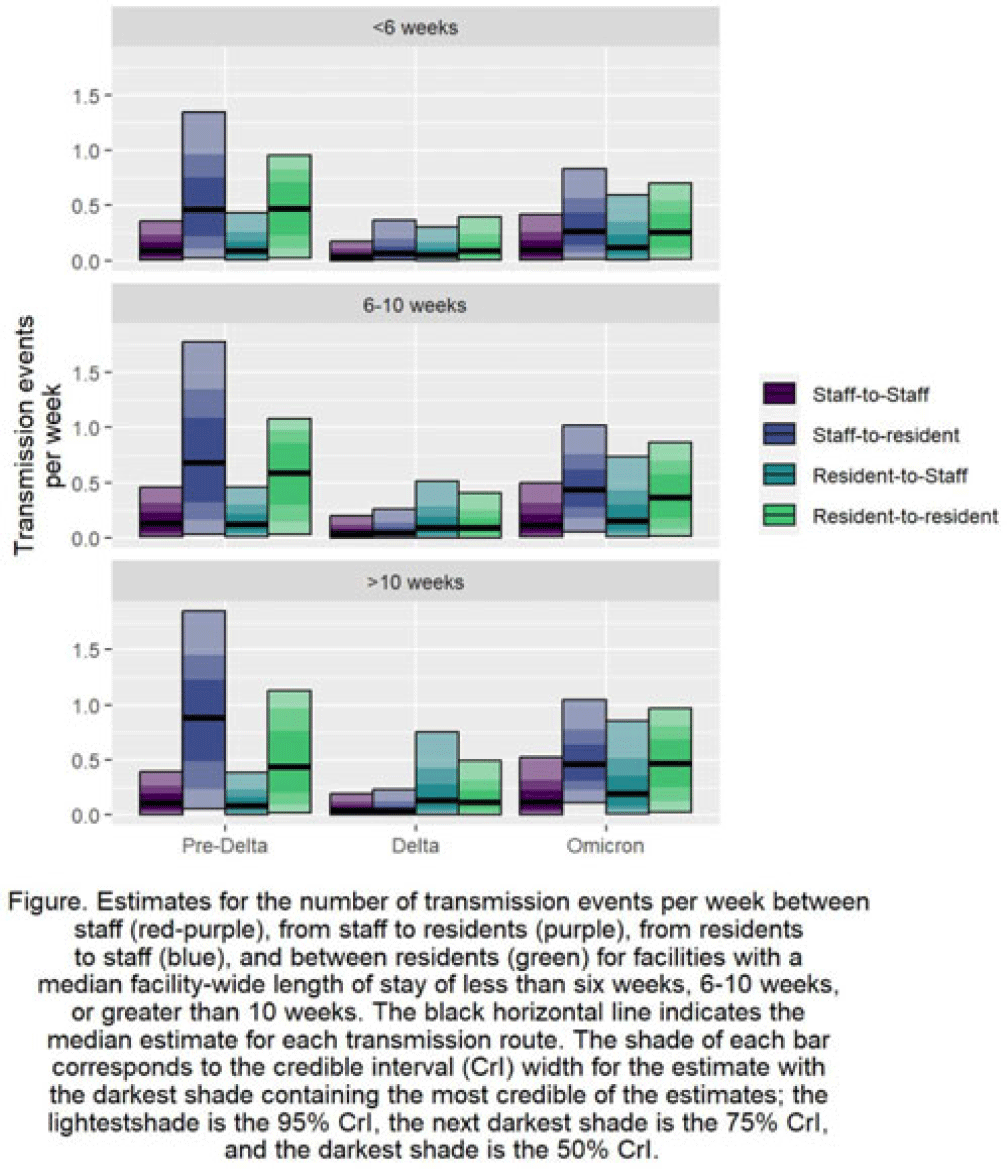
How COVID-19 spread varied by resident length of stay and resident–staff transmission pathways over time in US nursing homes
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s111
-
- Article
-
- You have access
- Open access
- Export citation
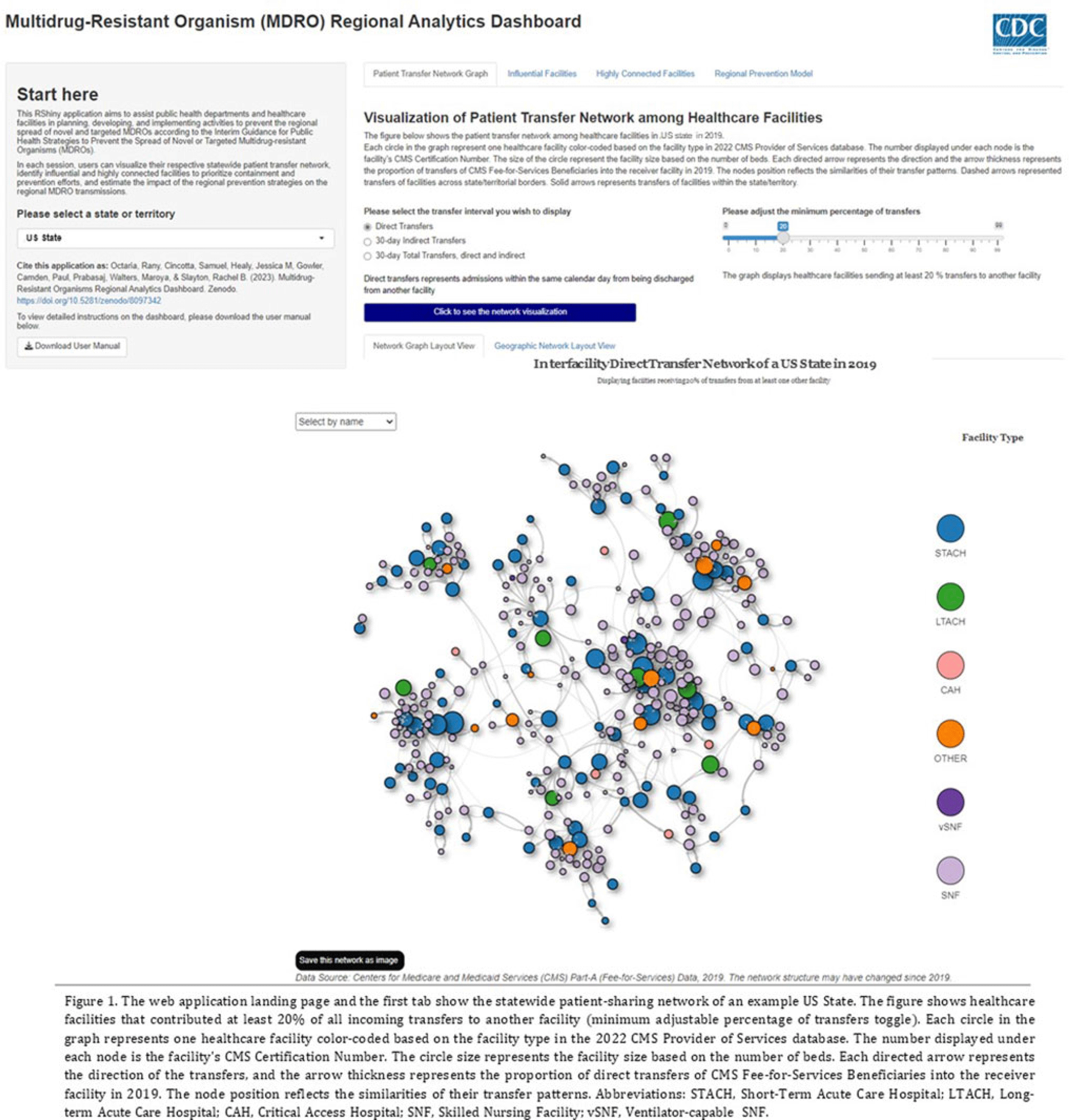
An interactive patient transfer network and model visualization tool for multidrug-resistant organism prevention strategies
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s120-s122
-
- Article
-
- You have access
- Open access
- Export citation
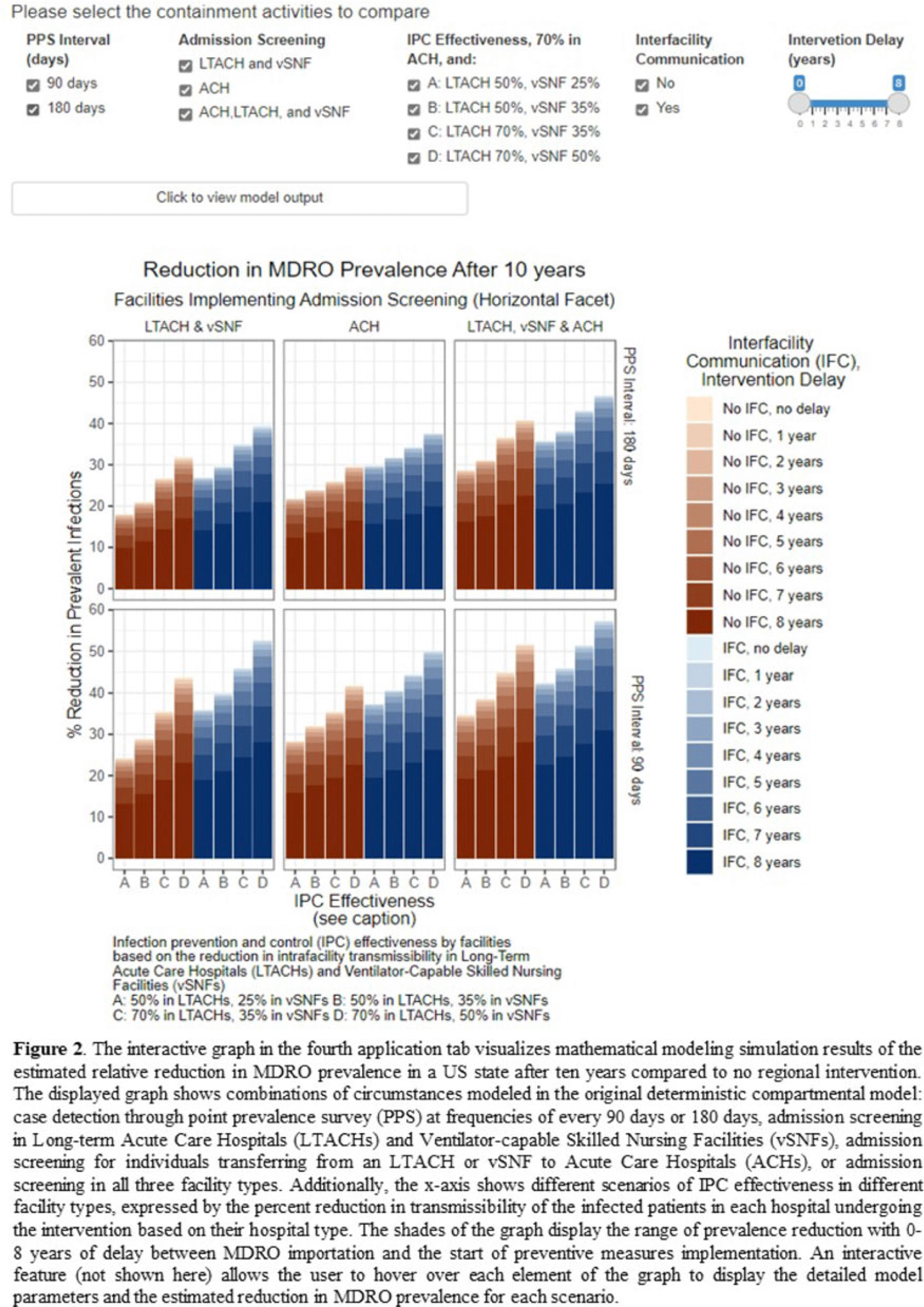
Predicting the regional impact of interventions to prevent and contain multidrug-resistant organisms
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s13-s14
-
- Article
-
- You have access
- Open access
- Export citation
Regional Impact of a CRE Intervention Targeting High Risk Postacute Care Facilities (Chicago PROTECT)
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s48-s49
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Optimizing Sentinel Surveillance to Target Containment of Emerging Multidrug-Resistant Organisms in Regional Networks
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s336-s337
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Decreased Hospitalizations and Costs From Infection in Sixteen Nursing Homes in the SHIELD OC Regional Decolonization Initiative
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s7-s8
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Validation of Administrative Codes for Identification of Staphylococcus aureus Infections Among Electronic Health Data
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s507-s509
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Surgical site infection risk following cesarean deliveries covered by Medicaid or private insurance
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 6 / June 2019
- Published online by Cambridge University Press:
- 09 April 2019, pp. 639-648
- Print publication:
- June 2019
-
- Article
- Export citation
The projected burden of complex surgical site infections following hip and knee arthroplasties in adults in the United States, 2020 through 2030
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 39 / Issue 10 / October 2018
- Published online by Cambridge University Press:
- 30 August 2018, pp. 1189-1195
- Print publication:
- October 2018
-
- Article
- Export citation
Attributable Mortality of Healthcare-Associated Infections Due to Multidrug-Resistant Gram-Negative Bacteria and Methicillin-Resistant Staphylococcus Aureus
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 38 / Issue 7 / July 2017
- Published online by Cambridge University Press:
- 01 June 2017, pp. 848-856
- Print publication:
- July 2017
-
- Article
- Export citation
The Cost–Benefit of Federal Investment in Preventing Clostridium difficile Infections through the Use of a Multifaceted Infection Control and Antimicrobial Stewardship Program
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 36 / Issue 6 / June 2015
- Published online by Cambridge University Press:
- 18 March 2015, pp. 681-687
- Print publication:
- June 2015
-
- Article
- Export citation