145 results
36 Naming in Monolingual and Bilingual Children with Epilepsy
-
- Journal:
- Journal of the International Neuropsychological Society / Volume 29 / Issue s1 / November 2023
- Published online by Cambridge University Press:
- 21 December 2023, pp. 35-36
-
- Article
-
- You have access
- Export citation
Changes in sleep and the prevalence of probable insomnia in undergraduate university students over the course of the COVID-19 pandemic: findings from the U-Flourish cohort study
-
- Journal:
- BJPsych Open / Volume 9 / Issue 6 / November 2023
- Published online by Cambridge University Press:
- 07 November 2023, e210
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Cognitive-behavioural versus cognitive analytic guided self-help for mild-to-moderate anxiety: a pragmatic, randomised patient preference trial – CORRIGENDUM
-
- Journal:
- The British Journal of Psychiatry / Volume 223 / Issue 5 / November 2023
- Published online by Cambridge University Press:
- 18 September 2023, p. 531
- Print publication:
- November 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Cognitive–behavioural versus cognitive–analytic guided self-help for mild-to-moderate anxiety: a pragmatic, randomised patient preference trial
-
- Journal:
- The British Journal of Psychiatry / Volume 223 / Issue 3 / September 2023
- Published online by Cambridge University Press:
- 03 July 2023, pp. 438-445
- Print publication:
- September 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Association between job role and coronavirus disease 2019 (COVID-19) among healthcare personnel, Iowa, 2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 01 December 2022, e188
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
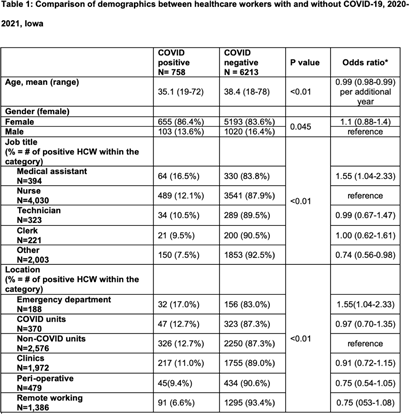
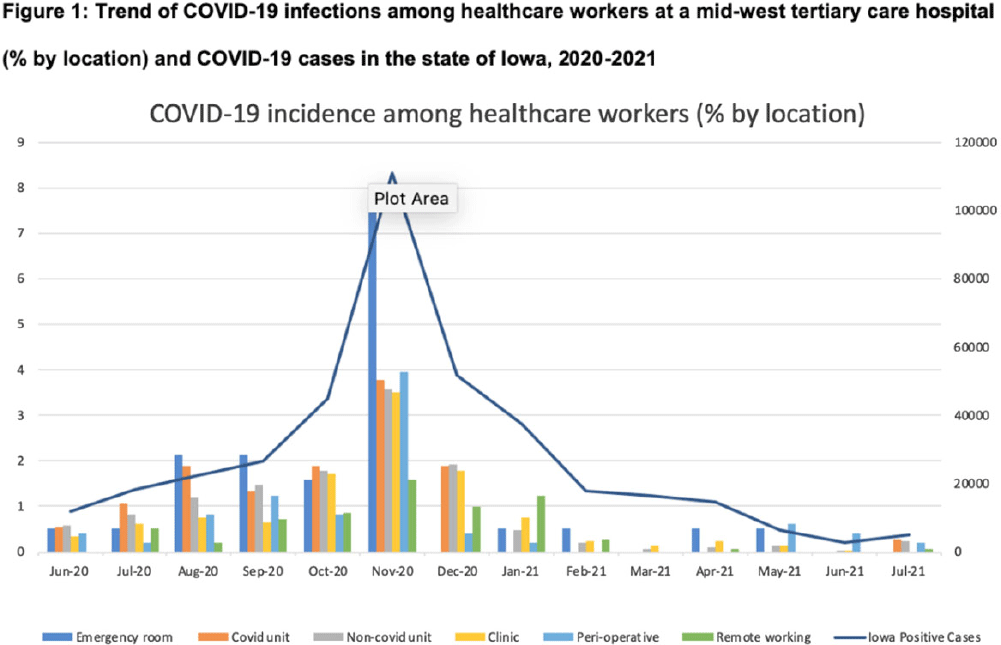
Coronavirus disease 2019 (COVID-19) among nonphysician healthcare personnel by work location at a tertiary-care center, Iowa, 2020–2021
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 8 / August 2023
- Published online by Cambridge University Press:
- 02 June 2022, pp. 1351-1354
- Print publication:
- August 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
COVID-19 incidence among nonphysician healthcare workers at a tertiary care center–Iowa, 2020–2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s6-s7
-
- Article
-
- You have access
- Open access
- Export citation
Blood-culture ordering practices in patients with a central line at an academic medical center–Iowa, 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s30
-
- Article
-
- You have access
- Open access
- Export citation
Age-related brain deviations and aggression
-
- Journal:
- Psychological Medicine / Volume 53 / Issue 9 / July 2023
- Published online by Cambridge University Press:
- 22 April 2022, pp. 4012-4021
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
When Agronomy Flirts with Markets, Gender, and Nutrition: A Political Ecology of the New Green Revolution for Africa and Women’s Food Security in Burkina Faso
-
- Journal:
- African Studies Review / Volume 65 / Issue 1 / March 2022
- Published online by Cambridge University Press:
- 20 April 2022, pp. 41-65
-
- Article
- Export citation
Blood culture practices in patients with a central line at an academic medical center—Iowa, 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 13 April 2022, e64
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
IIT, half masked and half disfigured
-
- Journal:
- Behavioral and Brain Sciences / Volume 45 / 2022
- Published online by Cambridge University Press:
- 23 March 2022, e60
-
- Article
- Export citation
Improving the generalizability of infant psychological research: The ManyBabies model
-
- Journal:
- Behavioral and Brain Sciences / Volume 45 / 2022
- Published online by Cambridge University Press:
- 10 February 2022, e35
-
- Article
- Export citation
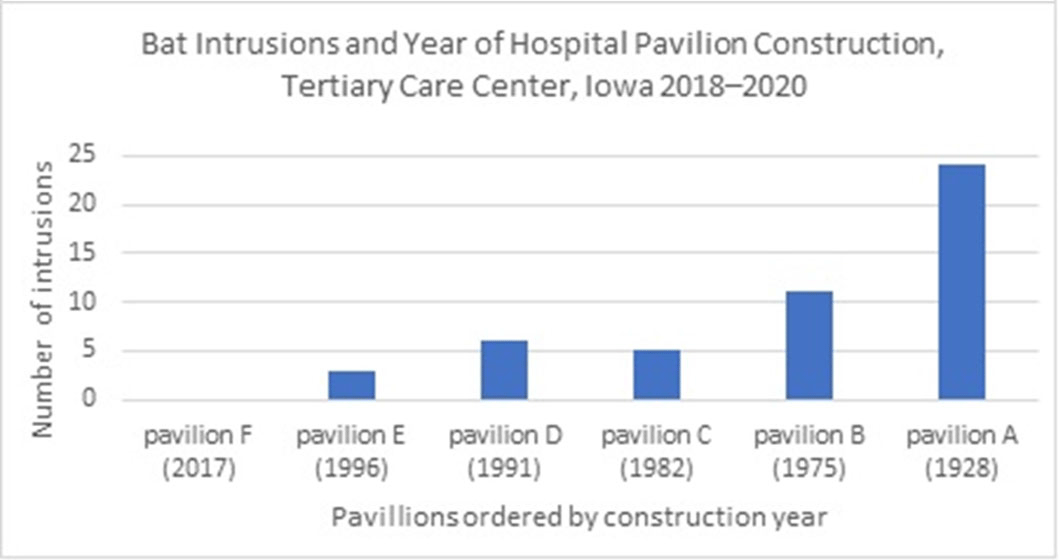
Bat intrusions at a tertiary care center in Iowa, 2018–2020
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 12 / December 2022
- Published online by Cambridge University Press:
- 16 August 2021, pp. 1948-1950
- Print publication:
- December 2022
-
- Article
- Export citation
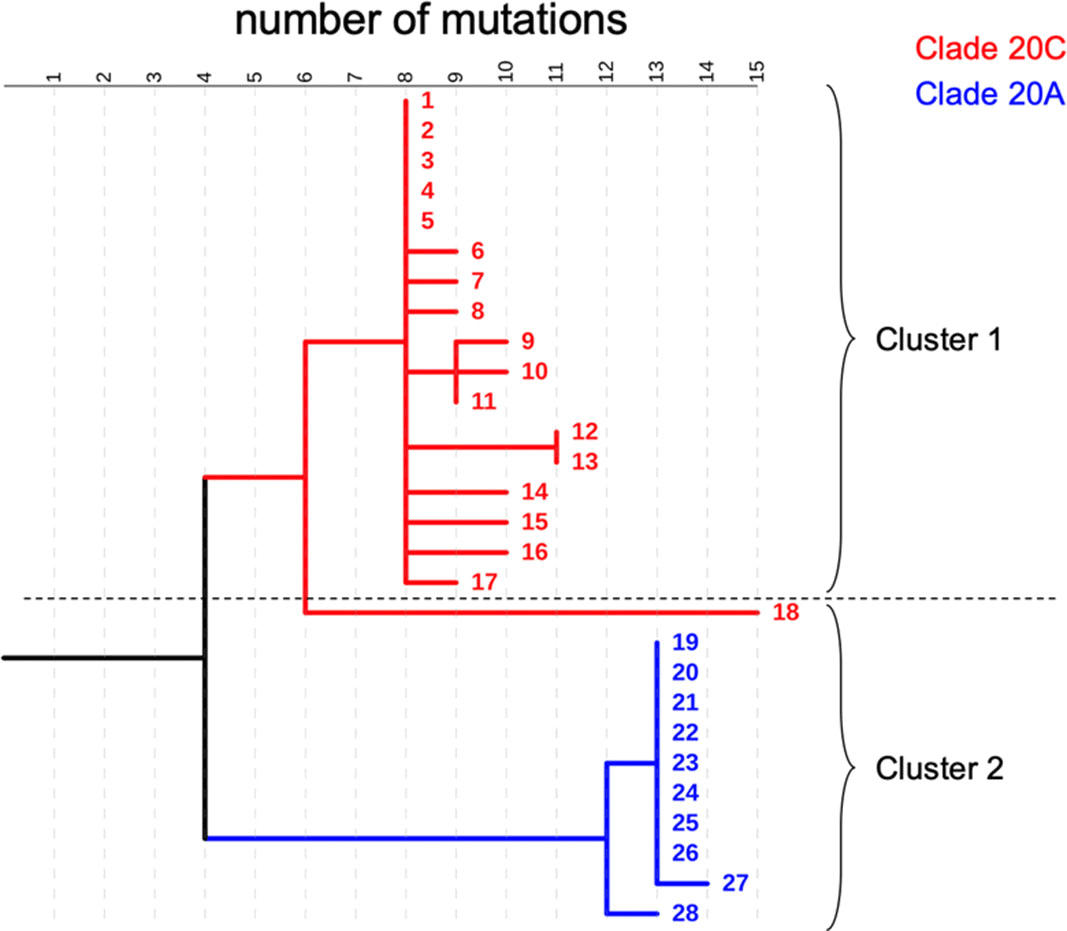
Molecular Epidemiology of Large COVID-19 Clusters at an Academic Medical Center, March–October 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s10-s11
-
- Article
-
- You have access
- Open access
- Export citation
Bat Intrusions at a Tertiary Care Center, Iowa 2018–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s16
-
- Article
-
- You have access
- Open access
- Export citation
COVID-19 Conversion after Exposure in a Semiprivate Room at a Tertiary Care Center in Iowa, July–December 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s20-s21
-
- Article
-
- You have access
- Open access
- Export citation
Coronavirus Disease 2019 (COVID-19) Admission Screening at a Tertiary-Care Center, Iowa 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s1
-
- Article
-
- You have access
- Open access
- Export citation
Suspected COVID-19 Reinfections at a Tertiary Care Center, Iowa 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s19
-
- Article
-
- You have access
- Open access
- Export citation
Impact of COVID-19 on Volume of Infection Prevention and Control Calls at a Tertiary-Care Center in Iowa, 2018–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s53-s54
-
- Article
-
- You have access
- Open access
- Export citation