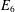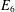OBJECTIVES/SPECIFIC AIMS: Objective: apply checkpoint inhibitors that are specific to the exhaustive markers expressed on tumor CD8+ T-cells ex vivo in order to improve cytokine release and cytotoxic function in comparison to two control groups: (1.) T-cells that receive no antibodies; (2.) T-cells that receive standard inhibition with PD-1 and CTLA-4 antibodies only. Long-term objective: provide personalized medicine in the treatment of HCC by using checkpoint inhibitors that are specific to the receptors expressed by an individual tumor. METHODS/STUDY POPULATION: The study population includes patients undergoing liver transplantation or surgical resection for HCC. Two grams of tumor, two grams of healthy liver tissue at least one centimeter from the tumor margin, and 50 milliliters of blood will be obtained. Solid tissue will be mechanically and enzymatically disrupted and CD8+ T-cells will be isolated from all sites. Using flow cytometry, the expression of surface receptors PD-1, CTLA-4, LAG-3, TIM-3, BTLA, CD244, and CD160 will be categorized in each tissue to identify which receptors are upregulated in the tumor microenvironment. Up to three antibodies specific to the upregulated receptor(s) on the tumor T-cells will be applied per specimen. The experimental arm will receive these antibodies and co-stimulation with CD3/CD28 and will be compared to two controls. One control will receive only CD3/CD28, and the other will receive CD3/CD28 in addition to the standard combination of PD-1 and CTLA-4 inhibitors. From each condition, flow cytometry will be used to assess the mean production of interleukin-2, tumor necrosis factor-α, interferon-γ, granzyme B, and perforin expression as an assessment of T-cell function. RESULTS/ANTICIPATED RESULTS: Preliminary data from the peripheral blood of healthy controls confirms that the developed flow cytometry panels effectively identify the surface receptors and cytokine production of CD8+ T-cells. Two patients have successfully been enrolled in this study. It is predicted that T-cells extracted from the tumor will express more inhibitory receptors than normal liver or peripheral blood and will have increased function after they are targeted with checkpoint inhibitors that are specific to the inhibitory surface receptors they express. DISCUSSION/SIGNIFICANCE OF IMPACT: HCC is the second leading cause of cancer-related death worldwide and therapeutic options are limited for patients who are not surgical candidates. T-cells are a critical component of the anti-tumor response to HCC. However, T-cells can develop an exhausted phenotype characterized by up-regulated inhibitory receptors (PD-1, CTLA-4, LAG-3, TIM-3, CD-244, CD-160, BTLA) and decreased function, allowing for immune escape. Clinical trials using combined checkpoint inhibition with PD-L1 and CTLA-4 antibodies have been considered a breakthrough for patients with advanced HCC, as up to 25% show an objective tumor response. The explanation for the varied susceptibility to checkpoint inhibition remains unknown and is hypothesized to be secondary to inconsistencies in the expression of surface inhibitory receptors. Although inhibitory receptor expression has been shown to be upregulated under conditions of hepatitis and/or HCC, there has been no single study to effectively investigate the expression of all known inhibitors in order to better explore the interplay between them. It will be of great academic interest and clinical purpose to evaluate individual receptor expression and engage the correlating antibodies given the possibility of synergism between receptors and the need for a more profound anti-tumor T-cell response in HCC.
 $E_{6}$
$E_{6}$