164 results
The devil’s in the defaults: An interrupted time-series analysis of the impact of default duration elimination on exposure to fluoroquinolone therapy
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 13 February 2024, pp. 1-7
-
- Article
- Export citation
Identification of carbapenem-resistant organism (CRO) contamination of in-room sinks in intensive care units in a new hospital bed tower
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 3 / March 2024
- Published online by Cambridge University Press:
- 19 January 2024, pp. 302-309
- Print publication:
- March 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
The impact of minimally invasive surgical approaches on surgical-site infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 5 / May 2024
- Published online by Cambridge University Press:
- 03 January 2024, pp. 557-561
- Print publication:
- May 2024
-
- Article
- Export citation
A cluster of three extrapulmonary Mycobacterium abscessus infections linked to well-maintained water-based heater-cooler devices
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 5 / May 2024
- Published online by Cambridge University Press:
- 21 December 2023, pp. 644-650
- Print publication:
- May 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
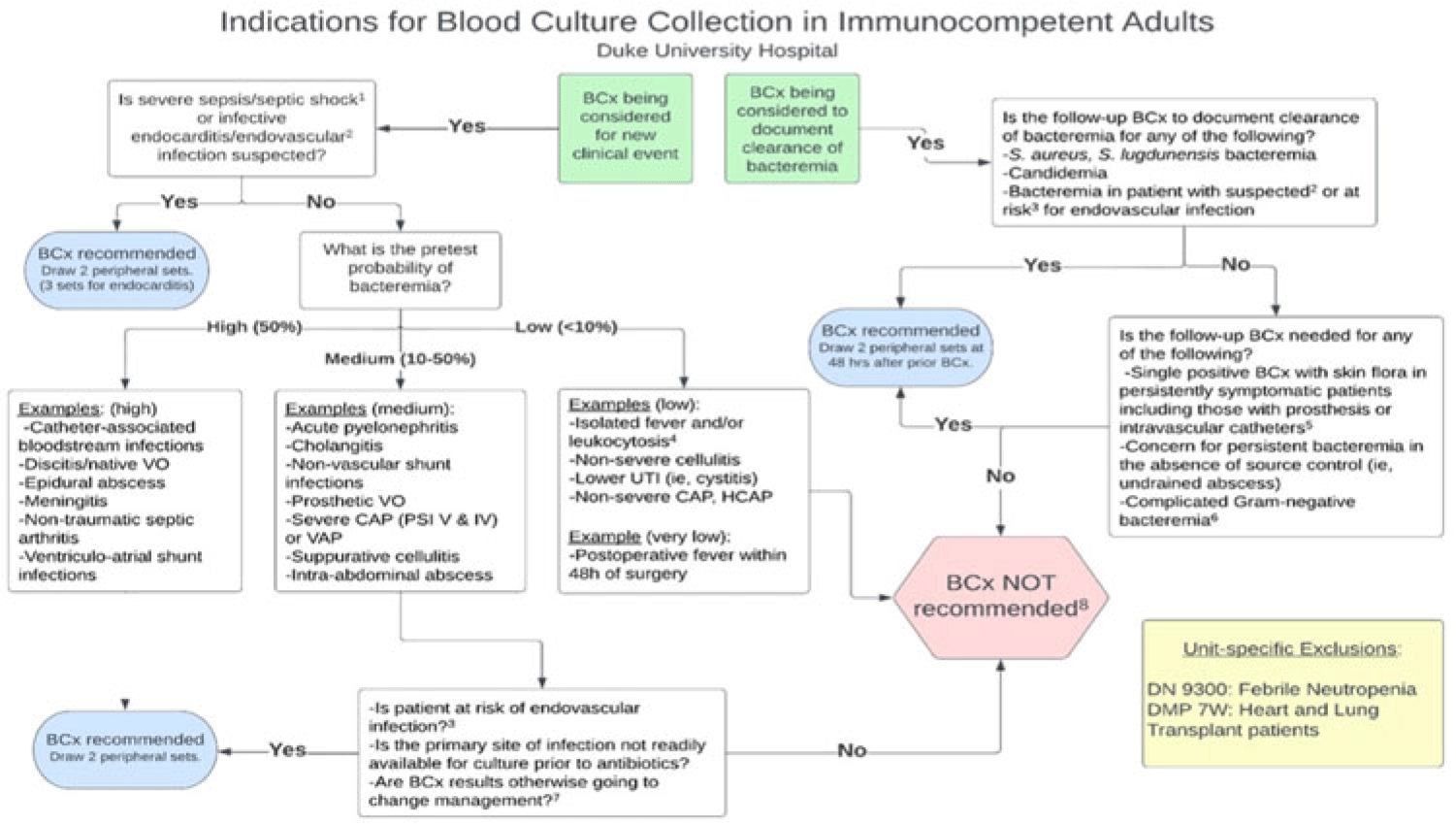
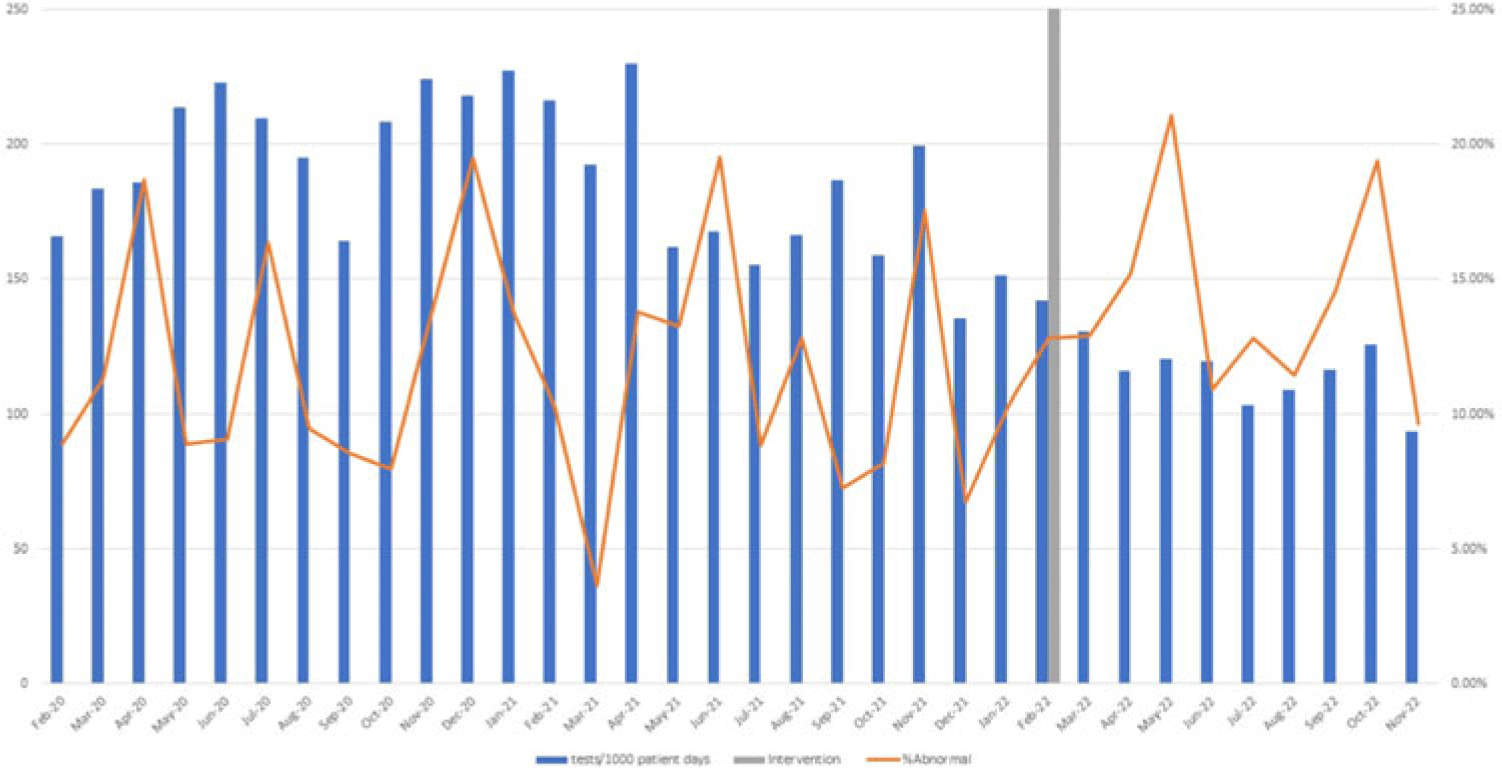
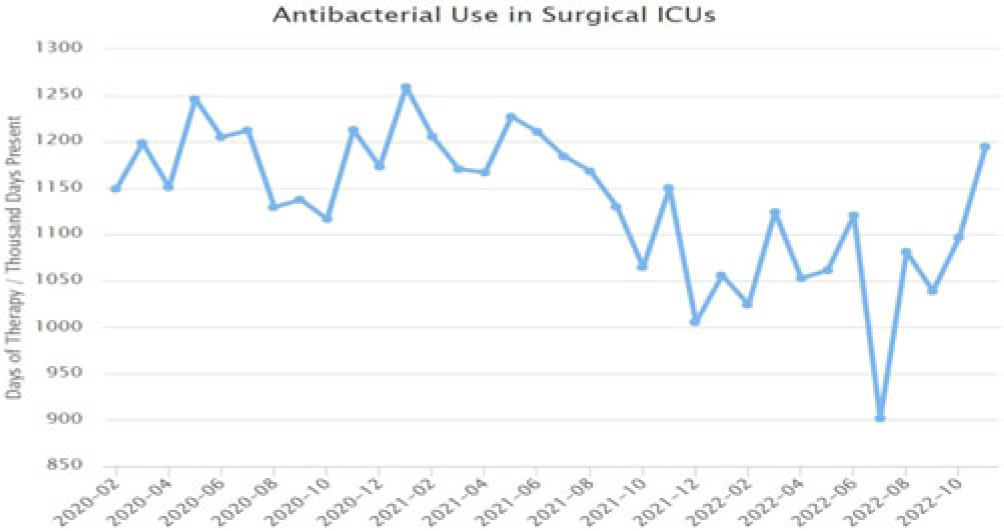
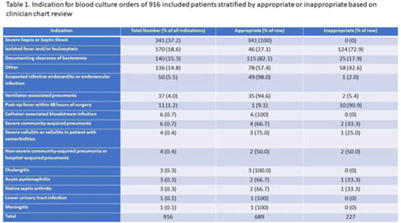
Implementation of a diagnostic stewardship intervention to improve blood-culture utilization in 2 surgical ICUs: Time for a blood-culture change
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 4 / April 2024
- Published online by Cambridge University Press:
- 11 December 2023, pp. 452-458
- Print publication:
- April 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Racial disparities in Clostridioides difficile testing in three southeastern US hospitals
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 4 / April 2024
- Published online by Cambridge University Press:
- 20 November 2023, pp. 429-433
- Print publication:
- April 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Introduction to A Compendium of Strategies to Prevent Healthcare-Associated Infections In Acute-Care Hospitals: 2022 Updates
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 10 / October 2023
- Published online by Cambridge University Press:
- 19 October 2023, pp. 1533-1539
- Print publication:
- October 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
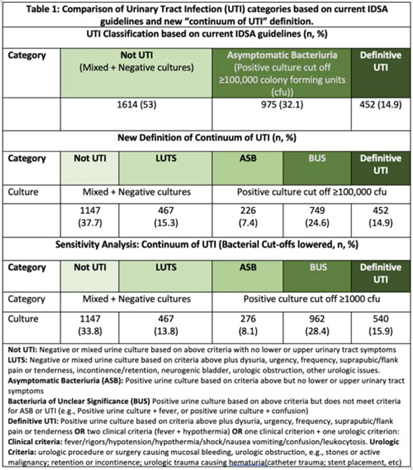
Proposing the “continuum of UTI” for a nuanced approach to antimicrobial stewardship
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s108
-
- Article
-
- You have access
- Open access
- Export citation
Implementation of diagnostic stewardship in two surgical ICUs: Time for a blood-culture change
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s9-s10
-
- Article
-
- You have access
- Open access
- Export citation
Measuring the efficacy of routine disinfection methods on frequently used physical therapy equipment
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s64-s65
-
- Article
-
- You have access
- Open access
- Export citation
Executive Summary: A Compendium of Strategies to Prevent Healthcare-Associated Infections in Acute-Care Hospitals: 2022 Updates
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 10 / October 2023
- Published online by Cambridge University Press:
- 22 August 2023, pp. 1540-1554
- Print publication:
- October 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
A 7-year analysis of attributable costs of healthcare-associated infections in a network of community hospitals in the southeastern United States
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 1 / January 2024
- Published online by Cambridge University Press:
- 17 August 2023, pp. 103-105
- Print publication:
- January 2024
-
- Article
- Export citation
Disinfection efficacy of Oxivir TB wipe residue on severe acute respiratory coronavirus virus 2 (SARS-CoV-2)
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 11 / November 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. 1891-1893
- Print publication:
- November 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Strategies to prevent surgical site infections in acute-care hospitals: 2022 Update
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 5 / May 2023
- Published online by Cambridge University Press:
- 04 May 2023, pp. 695-720
- Print publication:
- May 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Disparities in central line-associated bloodstream infection and catheter-associated urinary tract infection rates: An exploratory analysis
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 11 / November 2023
- Published online by Cambridge University Press:
- 14 April 2023, pp. 1857-1860
- Print publication:
- November 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Using the COM-B model to identify barriers to and facilitators of evidence-based nurse urine-culture practices
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 31 March 2023, e62
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Infection prevention and antibiotic stewardship program needs and practices in 2021: A survey of the Society for Healthcare Epidemiology of America Research Network
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 14 March 2023, pp. 948-950
- Print publication:
- June 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Central-line–associated bloodstream infections secondary to anaerobes: Time for a definition change?
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 10 / October 2023
- Published online by Cambridge University Press:
- 03 February 2023, pp. 1697-1698
- Print publication:
- October 2023
-
- Article
- Export citation
Optimizing reflex urine cultures: Using a population-specific approach to diagnostic stewardship
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 2 / February 2023
- Published online by Cambridge University Press:
- 10 January 2023, pp. 206-209
- Print publication:
- February 2023
-
- Article
- Export citation
A comparison of methods for microbiologic environmental sampling
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 9 / September 2023
- Published online by Cambridge University Press:
- 01 December 2022, pp. 1502-1504
- Print publication:
- September 2023
-
- Article
- Export citation