49 results
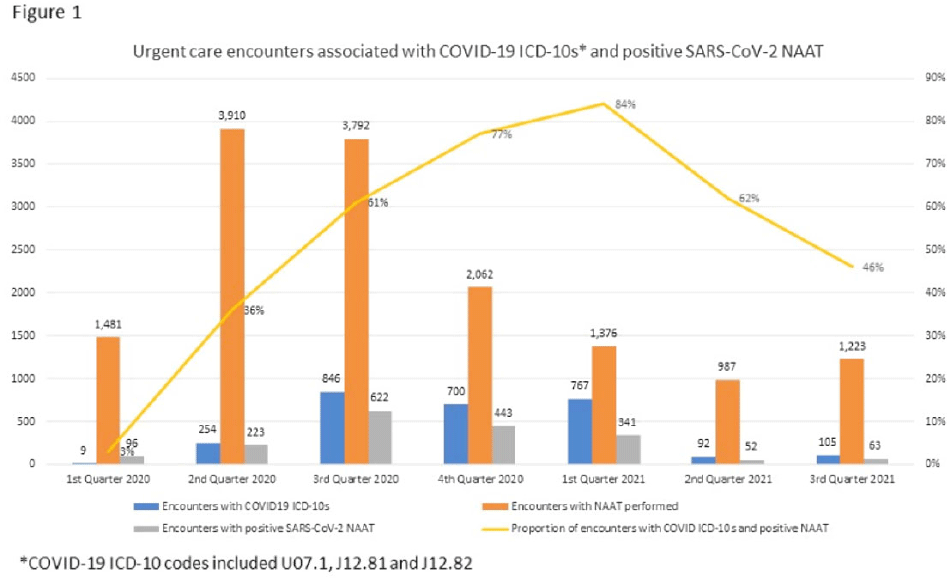
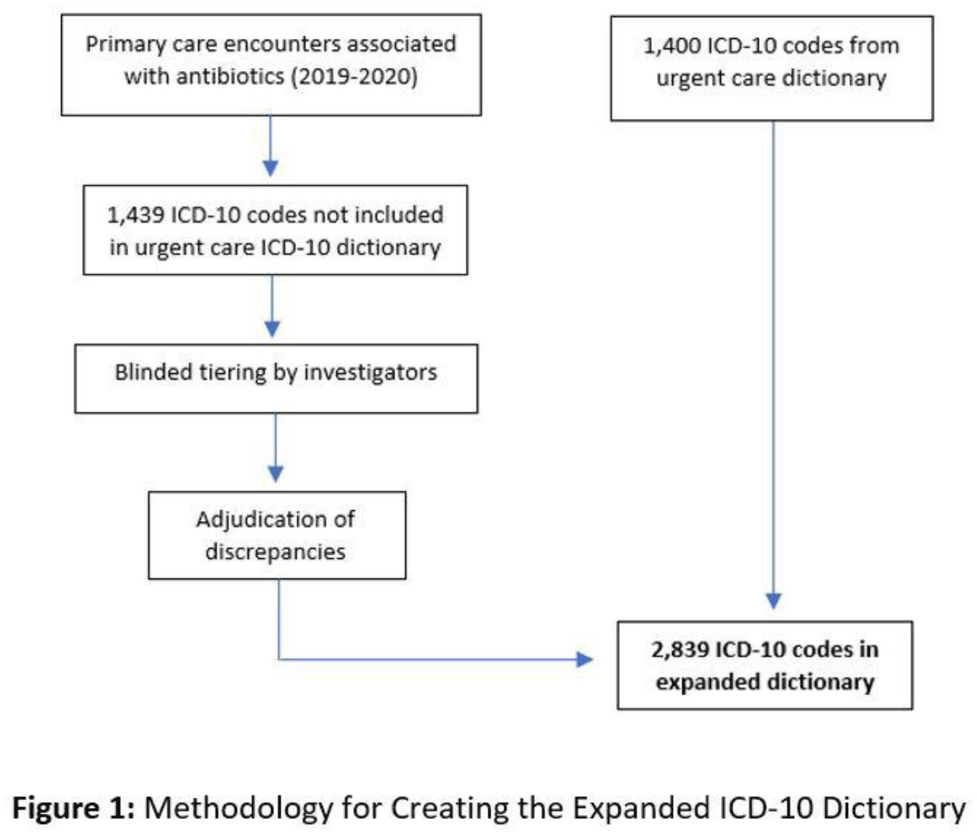
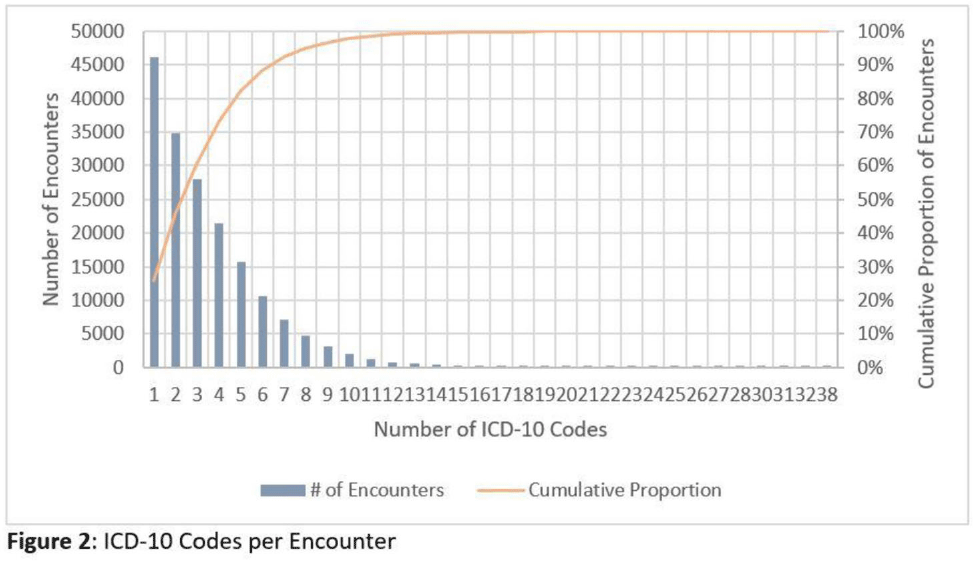
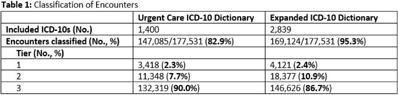
Impact of COVID-19 on urgent care diagnoses and the new AXR metric
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue 1 / 2024
- Published online by Cambridge University Press:
- 18 April 2024, e49
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Impact of an antibiotic stewardship initiative on urgent-care respiratory prescribing across patient race, ethnicity, and language
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 4 / April 2024
- Published online by Cambridge University Press:
- 11 December 2023, pp. 530-533
- Print publication:
- April 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Beyond antibiotic prescribing rates: first-line antibiotic selection, prescription duration, and associated factors for respiratory encounters in urgent care
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 05 September 2023, e146
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Comparison of antibiotic prescribing between physicians and advanced practice clinicians
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 1 / January 2024
- Published online by Cambridge University Press:
- 09 August 2023, pp. 117-119
- Print publication:
- January 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Characteristics of antifungal utilization for hospitalized children in the United States
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 02 December 2022, e190
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Urgent-care antibiotic prescribing: An exploratory analysis to evaluate health inequities
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 14 November 2022, e184
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Pediatric infectious disease physician perceptions of antimicrobial stewardship programs
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 7 / July 2023
- Published online by Cambridge University Press:
- 08 August 2022, pp. 1155-1158
- Print publication:
- July 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
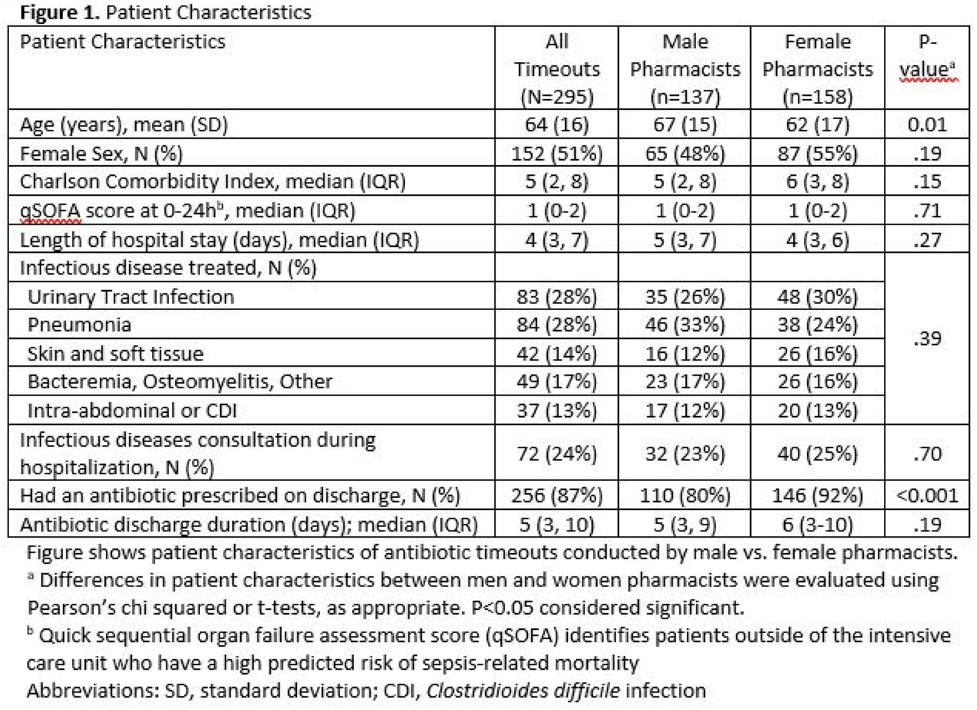
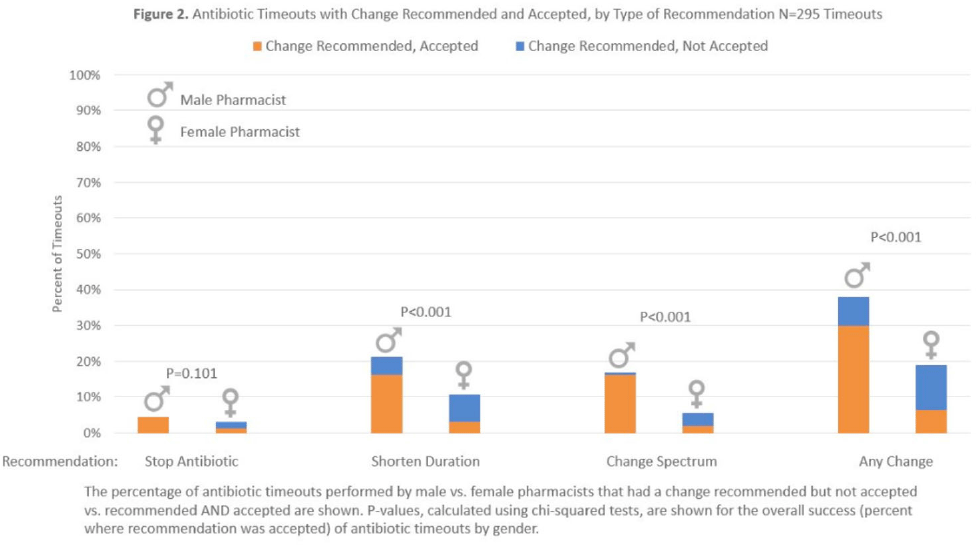
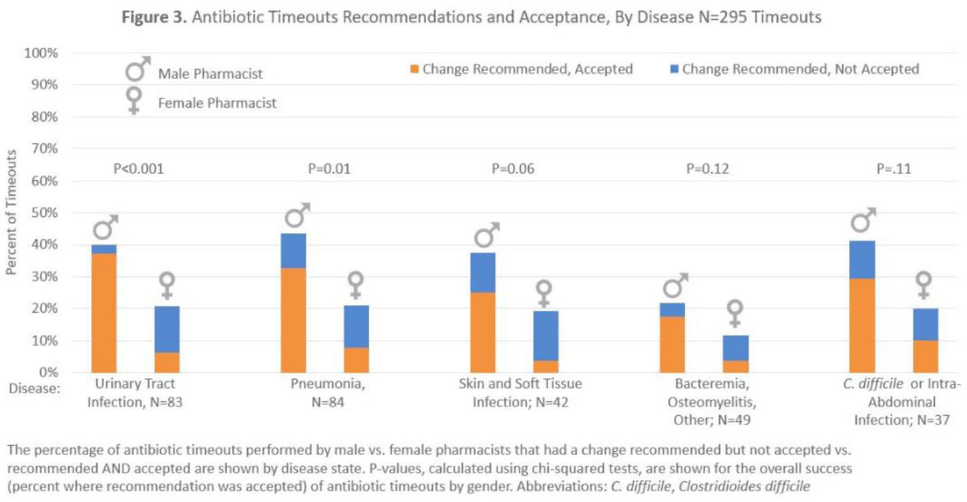
Pharmacist gender and physician acceptance of antibiotic stewardship recommendations: An analysis of the reducing overuse of antibiotics at discharge home intervention
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 4 / April 2023
- Published online by Cambridge University Press:
- 07 June 2022, pp. 570-577
- Print publication:
- April 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
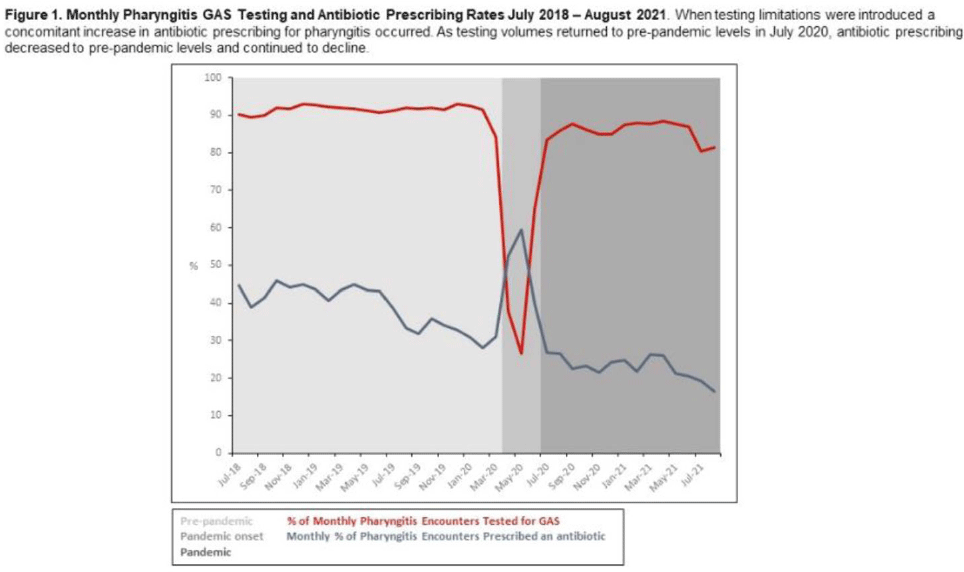
Rapid streptococcal pharyngitis testing and antibiotic prescribing before and during the COVID-19 pandemic
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s20-s21
-
- Article
-
- You have access
- Open access
- Export citation
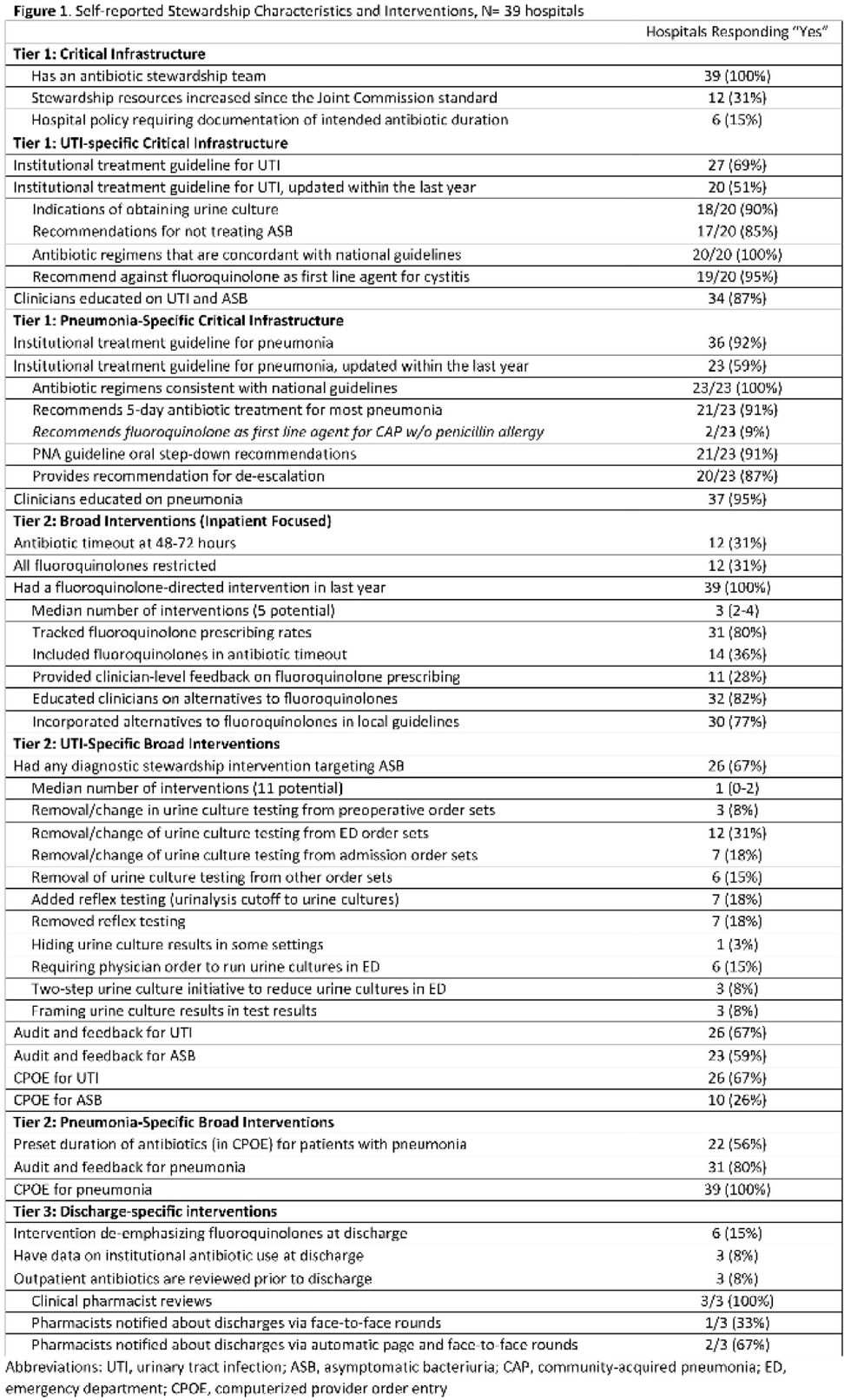
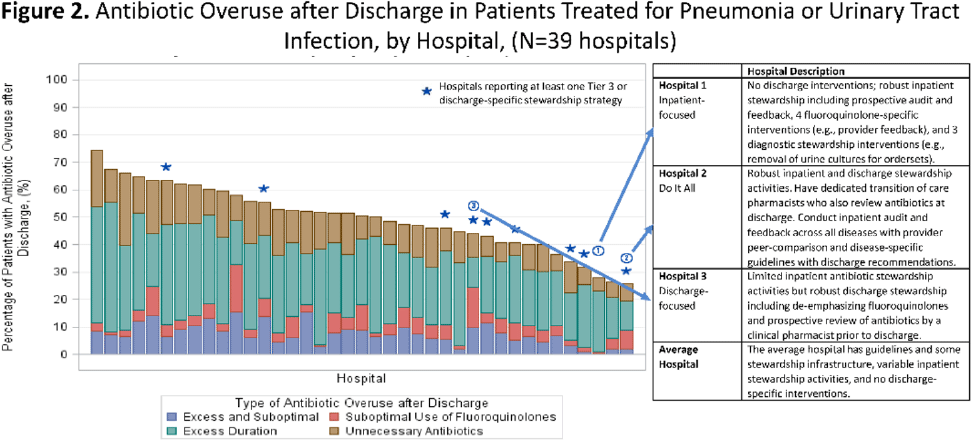
Antibiotic stewardship strategies and antibiotic overuse after hospital discharge: Analysis of the ROAD Home Framework
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s16-s17
-
- Article
-
- You have access
- Open access
- Export citation
The effect of gender bias on acceptance of antibiotic stewardship recommendations by clinical pharmacists
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s67-s68
-
- Article
-
- You have access
- Open access
- Export citation
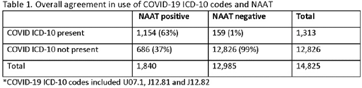
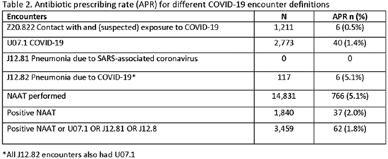
Impact of different COVID-19 encounter definitions on antibiotic prescribing rates in urgent care
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s5
-
- Article
-
- You have access
- Open access
- Export citation
Metrics in outpatient stewardship: Is more always better?
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s70-s71
-
- Article
-
- You have access
- Open access
- Export citation
Rapid streptococcal pharyngitis testing and antibiotic prescribing before and during the coronavirus disease 2019 (COVID-19) pandemic
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 10 May 2022, e80
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Exploring unintended consequences of adult antimicrobial stewardship programs: An Emerging Infections Network survey
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 5 / May 2023
- Published online by Cambridge University Press:
- 20 April 2022, pp. 791-793
- Print publication:
- May 2023
-
- Article
- Export citation
Changes in outpatient antibiotic prescribing for acute respiratory illnesses, 2011 to 2018
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 17 December 2021, e66
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Outpatient antimicrobial stewardship programs in pediatric institutions in 2020: Status, needs, barriers
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 10 / October 2022
- Published online by Cambridge University Press:
- 22 October 2021, pp. 1396-1402
- Print publication:
- October 2022
-
- Article
- Export citation
A Veterans’ Healthcare Administration (VHA) antibiotic stewardship intervention to improve outpatient antibiotic use for acute respiratory infections: A cost-effectiveness analysis
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 10 / October 2022
- Published online by Cambridge University Press:
- 29 September 2021, pp. 1389-1395
- Print publication:
- October 2022
-
- Article
- Export citation
Pediatric antimicrobial stewardship practices at discharge: A national survey
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 11 / November 2022
- Published online by Cambridge University Press:
- 16 July 2021, pp. 1686-1688
- Print publication:
- November 2022
-
- Article
- Export citation
Association between antibiotic prescribing and visit duration among patients with respiratory tract infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 9 / September 2022
- Published online by Cambridge University Press:
- 11 June 2021, pp. 1238-1241
- Print publication:
- September 2022
-
- Article
- Export citation