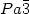HfV2O7 powders were prepared by means of thermal decomposition of a mixture of vanadyl
and hafnyl oxalates in air at 973 K. The room temperature structure and its evolution with
temperature was investigated using both electron microscopy (TEM and HREM) and powder
diffraction (X-ray and neutron) techniques combined with DSC measurements. HfV2O7 exhibits
two temperature-induced phase transitions between room temperature and 480 K. Over the highest
transition (370 K), HfV2O7 crystallizes in a cubic system with a cell parameter of 8.75 Å and $Pa\bar{3}$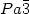 space group. A negative thermal expansion is observed in this high temperature phase and can be
correlated to anisotropic oxygen atom displacements. On the other hand, the room temperature
structure of HfV2O7 is also described in a cubic symmetry, but the existence of superstructure
reflections leads to a cell parameter three times larger than for the high temperature structure
(~ 26.25 Å). Moreover, HREM calculations have shown that the atomic positions, usually used to
describe the superstructures of the ZrV2O7 compounds, cannot be used in the case of HfV2O7 at
room temperature.
space group. A negative thermal expansion is observed in this high temperature phase and can be
correlated to anisotropic oxygen atom displacements. On the other hand, the room temperature
structure of HfV2O7 is also described in a cubic symmetry, but the existence of superstructure
reflections leads to a cell parameter three times larger than for the high temperature structure
(~ 26.25 Å). Moreover, HREM calculations have shown that the atomic positions, usually used to
describe the superstructures of the ZrV2O7 compounds, cannot be used in the case of HfV2O7 at
room temperature.