Many studies have shown an association between depressive symptoms and dementia or cognitive decline (Reference JormJorm, 2000) but the nature of this relationship remains equivocal. Depressive symptoms have been found to precede cognitive decline (Reference Yaffe, Blackwell and GoreYaffe et al, 1999) or to follow the onset of dementia (Reference Chen, Ganguli and MulsantChen et al, 1999). Some studies did not report any significant association (Reference Dufouil, Fuhrer and DartiguesDufouil et al, 1996; Reference Palsson, Aevarsson and SkoogPalsson et al, 1999). Bassuk et al (Reference Bassuk, Berkman and Wypij1998) found that depression predicted cognitive decline over 3-year and 6-year follow-up periods for people with medium, but not high, cognitive scores at baseline. Geerlings et al (Reference Geerlings, Schoevers and Beekman2000) found that depression was associated with an increased risk of Alzheimer's disease and cognitive decline only in people with higher levels of education. The objectives of our study were:
-
(a) to test whether depressive symptoms predict cognitive decline in an elderly population with normal cognition;
-
(b) to evaluate whether depression predicts persistent low cognitive functioning at follow-up;
-
(c) to examine whether people reporting episodic or persistent depressive episodes show a similar or different association with cognitive decline.
METHOD
Sample and study design
The Étude sur le Vieillissement Artériel (EVA) is a longitudinal study of cognitive and vascular ageing. The EVA study population consisted of 1389 persons born between 1922 and 1932, who were selected from the electoral rolls of the city of Nantes in western France, and were invited by mail to participate in the study. The survey design was approved by the ethics committee of the University Hospital of Bicêtre, and written consent was required for each participant. Baseline data were collected between June 1991 and June 1993.
Interviews and examinations were conducted at the study centre. Data on demographic background, occupation, medical history, drug use and personal habits were obtained using a standardised questionnaire during a face-to-face interview. Mean weekly alcohol intake was estimated from a detailed description of alcoholic beverage consumption during a typical week. Participants were asked whether they had suffered from any of six specified common diseases (myocardial infarction, angina, stroke, hypercholesterolaemia, hypertension, diabetes) or had any other chronic medical condition. All drugs used during the month preceding the examination were noted, and were subsequently coded according to the classification proposed by the French national prescription handbook, the Guide National de Prescription des Médicaments (1991). Psychotropic drugs included anxiolytic, hypnotic, sedative, neuroleptic, antidepressant and normothymic agents.
The group was invited to participate in two follow-up examinations, which were conducted 2 years and 4 years after the baseline evaluation.
Population
Among the initial sample of 1389 persons, 200 (14.4%) had a baseline score on the Mini-Mental State Examination (MMSE) below 26 and were excluded from the analysis. Of the 1189 persons considered eligible for the study, 18 died before the 4-year follow-up assessment, 137 were not followed up at 2 years or 4 years, and 31 had not completed the Center for Epidemiologic Studies depression scale (CES—D) rating at baseline. So, 84% of eligible participants were analysed (n=1003). A further 31 participants had no CES—D assessment at 2-year or 4-year follow-up.
Cognitive function testing
A trained psychologist administered a battery of tests assessing different areas of cognitive and psychomotor functioning. Tests were chosen with special regard for their sensitivity to ageing. For the current analysis we considered a measure of global cognitive abilities, the MMSE (Reference Folstein, Folstein and McHughFolstein et al, 1975), which yields scores ranging from 0 to 30. The MMSE was administered to participants at baseline and at 2-year and 4-year follow-up.
Assessment of depressive symptoms
Depressive symptoms were measured using the CES—D (Reference RadloffRadloff, 1977). It consists of 20 self-report items concerning symptoms and feelings experienced during the preceding week. Each item is scored from 0 to 3 according to the frequency of the symptom. Evaluation of the CES—D in a French population (Reference Fuhrer and RouillonFuhrer & Rouillon, 1989) showed that men and women scoring more than 16 or 22, respectively, should be considered at high risk of clinical depression. Using this cut-off point the authors obtained a sensitivity and specificity which were, respectively, 0.76 and 0.71 in detecting a major depressive episode according to DSM—III criteria (American Psychiatric Association, 1980). In our study the internal consistency of the CES—D was good (standardised Cronbach's α=0.88). Work with the CES—D in community samples has consistently identified four factors that underlie the measure: depressed affect, positive affect, somatic complaints and interpersonal relationships.
The CES—D was sent to the participants by mail approximately 3 weeks before the examination and completed questionnaires were brought back at the time of the examination. The instrument was administered to the participants at the baseline assessment and at 2-year and 4-year follow-up.
Data analysis
We excluded from the data analysis people with a baseline MMSE score lower than 26 for two reasons: to exclude those with subthreshold levels of dementia, and to include only those at risk of cognitive decline.
The differences in MMSE scores between baseline and 4-year follow-up examinations were analysed as quantitative variables, and also as binary variables. For the latter analyses, cognitive decline was defined as a decrease of 3 points or more in the MMSE scores. We defined ‘low cognitive functioning at follow-up’ as MMSE scores of 25 or less at 2-year and 4-year follow-up. Participants were defined as having high baseline levels of depressive symptoms when their baseline CES—D score was above the gender-specific cut-off point. In order to evaluate whether the risk of cognitive decline is higher when the participant reports several depressive episodes at follow-up evaluation, we defined three study groups. Participants reporting high levels of depressive symptoms at one or at more than one evaluation were assigned to an ‘episodic’ or a ‘persistent’ depressive group, respectively, and were compared with participants who did not have high depressive symptom scores at any of the three assessments.
We tested whether a high depressive symptom score was associated with cognitive decline using multivariate analyses (covariance analysis and logistic regression models). All multivariate analyses were adjusted for gender, age, education (years of schooling), alcohol (mean weekly alcohol intake evaluated in millilitres of pure alcohol) and tobacco use (current smokers, former smokers, or never smokers), psychotropic drug use and the presence of chronic medical diseases, because these variables are known to be significantly associated with cognitive performance and psychopathological characteristics.
In order to examine whether the observed relationship between CES—D score and cognitive decline was due to depressed mood rather than to the cumulative number of symptoms, we evaluated whether the CES—D sub-score ‘depressive affect’ was predictive of cognitive decline, using linear and logistic regression models. We calculated the ‘depressive affect’ subscore, by summing the scores of items measuring the cognitive symptoms of depression (‘bothered’, ‘blues’, ‘depressed’, ‘fearful’, ‘lonely’, ‘cried’ and ‘sad’) (Reference Radloff and TeriRadloff & Teri, 1986). The sub-score ranged from 0 to 21.
Statistical testing was done at the conventional two-tailed α-level of 0.05. Data were analysed using the SAS package, Version 6 (Reference InstituteSAS Institute, 1989).
RESULTS
Population characteristics
The population characteristics are shown in Table 1. Participants with high levels of depressive symptoms more often used tobacco and psychotropic drugs.
Table 1 Demographic characteristics of the study population
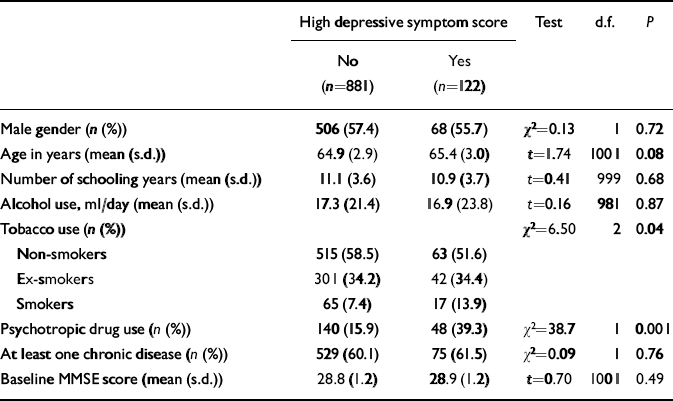
| High depressive symptom score | Test | d.f. | P | ||
|---|---|---|---|---|---|
| No (n=881) | Yes (n=122) | ||||
| Male gender (n (%)) | 506 (57.4) | 68 (55.7) | χ2=0.13 | 1 | 0.72 |
| Age in years (mean (s.d.)) | 64.9 (2.9) | 65.4 (3.0) | t=1.74 | 1001 | 0.08 |
| Number of schooling years (mean (s.d.)) | 11.1 (3.6) | 10.9 (3.7) | t=0.41 | 999 | 0.68 |
| Alcohol use, ml/day (mean (s.d.)) | 17.3 (21.4) | 16.9 (23.8) | t=0.16 | 981 | 0.87 |
| Tobacco use (n (%)) | χ2=6.50 | 2 | 0.04 | ||
| Non-smokers | 515 (58.5) | 63 (51.6) | |||
| Ex-smokers | 301 (34.2) | 42 (34.4) | |||
| Smokers | 65 (7.4) | 17 (13.9) | |||
| Psychotropic drug use (n (%)) | 140 (15.9) | 48 (39.3) | χ2=38.7 | 1 | 0.001 |
| At least one chronic disease (n (%)) | 529 (60.1) | 75 (61.5) | χ2=0.09 | 1 | 0.76 |
| Baseline MMSE score (mean (s.d.)) | 28.8 (1.2) | 28.9 (1.2) | t=0.70 | 1001 | 0.49 |
Comparison between participants according to follow-up
A slightly higher percentage of participants who were not followed up had a high depressive symptom score at baseline; they also had lower MMSE scores than those followed up. Participants not followed up had fewer years of schooling and used more psychotropic drugs. No difference was observed for gender, age or alcohol use.
Depressive symptoms at baseline and the risk of cognitive decline
Participants with high levels of depressive symptoms showed a greater decrease in MMSE score between the baseline and 4-year follow-up assessments (Table 2). High levels of depressive symptoms were also associated with a higher risk of a 3-point decrease in MMSE score and with a higher risk of low cognitive functioning at follow-up.
Table 2 Depressive symptoms at baseline and risk of cognitive decline at 4-year follow-up
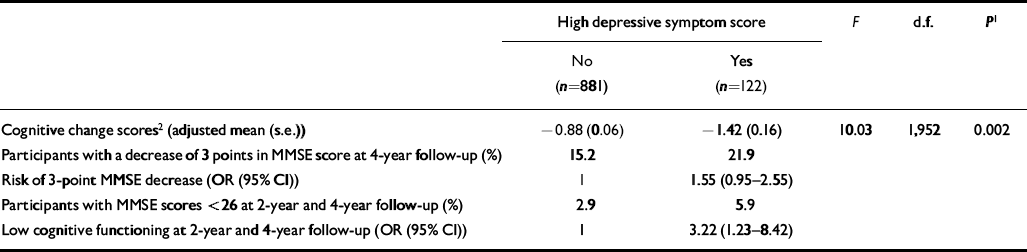
| High depressive symptom score | F | d.f. | P 1 | ||
|---|---|---|---|---|---|
| No (n=881) | Yes (n=122) | ||||
| Cognitive change scores2 (adjusted mean (s.e.)) | -0.88 (0.06) | -1.42 (0.16) | 10.03 | 1,952 | 0.002 |
| Participants with a decrease of 3 points in MMSE score at 4-year follow-up (%) | 15.2 | 21.9 | |||
| Risk of 3-point MMSE decrease (OR (95% CI)) | 1 | 1.55 (0.95-2.55) | |||
| Participants with MMSE scores <26 at 2-year and 4-year follow-up (%) | 2.9 | 5.9 | |||
| Low cognitive functioning at 2-year and 4-year follow-up (OR (95% CI)) | 1 | 3.22 (1.23-8.42) | |||
Persistent depressive symptoms and cognitive decline
Persistent — but not episodic — depressive symptoms were associated with greater cognitive decline and with low cognitive functioning at follow-up (Table 3). In order to test whether persistent but not episodic depressive symptoms predicted cognitive decline in those with depressive symptoms at baseline, we compared participants depressed at baseline but not at 2-year and 4-year follow-up (‘episodic’, n=39) and participants depressed at baseline and at 2-year and/or 4-year follow-up (‘persistent’, n=77) with participants not depressed at baseline. The ‘persistent’ group had twice the risk of cognitive decline (odds ratio 2.0, 95% CI 1.1-3.5) of participants who were not depressed; episodic depressive symptoms were not associated with cognitive decline (OR 0.8, 95% CI 0.3-2.2).
Table 3 Persistent depressive symptoms and risk of cognitive decline at 4-year follow-up

| No depressive symptoms (n=775) | High depressive symptoms | F | d.f. | P 1 | ||
|---|---|---|---|---|---|---|
| Episodic (n=102) | Persistent (n=95) | |||||
| Cognitive change scores2 (mean (s.e.)) | -0.85 (0.06) | -1.07 (0.17) | -1.48 (0.18) | 5.86 | 2,921 | 0.003 |
| Participants with a decrease of 3 points in MMSE score at 4-year follow-up (%) | 14.7 | 16.3 | 25.3 | |||
| Risk of 3-point MMSE decrease (OR (95% CI))1 | 1 | 1.22 (0.68-2.18) | 2.10 (1.23-3.58) | |||
| Participants with MMSE scores <26 at 2-year and 4-year follow-up (%) | 2.9 | 2.0 | 7.5 | |||
| Low cognitive functioning at 2-year and 4-year follow-up (OR (95% CI))1 | 1 | 0.76 (0.17-3.41) | 3.21 (1.21-8.52) | |||
The finding that depressive symptoms predict cognitive decline might be due to depressive symptoms being the consequence of a slight initial cognitive decline. In order to test this possibility, we considered the group of participants without cognitive decline between baseline and 2-year follow-up (Table 4). A high level of depressive symptoms at 2-year follow-up was associated with a greater decrease in MMSE score between the 2-year and 4-year follow-up assessments in this group.
Table 4 Difference in Mini-Mental State Examination (MMSE) scores according to level of depressive symptoms in participants without previous cognitive decline

| Depressive symptoms | Difference in MMSE score between 4- and 2-year follow-up | |
|---|---|---|
| Mean | (s.d.) | |
| Present (n=16) | -1.5* | (1.3) |
| Absent (n=114) | -1.0 | (1.2) |
Depressive affect
Linear and logistic regression models adjusted for baseline cognitive scores and covariables were used to examine the association between the sub-score ‘depressive affect’ and cognitive decline. In a linear regression model, depressive affect was associated with a greater decrease in MMSE score between baseline and 4-year follow-up (b=-0.06, s.e.=0.02, P=0.0003). Logistic regressions showed that depressive affect was also associated with a higher risk of low cognitive functioning at follow-up (OR 1.07, 95% CI 1.01-1.13) and a higher risk of a 3-point decrease in MMSE score (OR 1.06, 95% CI 1.00-1.12).
DISCUSSION
In this study initially high levels of depressive symptoms predicted cognitive decline in an elderly population of 1003 persons with good cognitive functioning at recruitment. People with initially high depressive symptom levels were more at risk of having persistent low cognitive functioning at follow-up. These associations concerned those with persistent but not episodic depressive symptoms. These results are in agreement with longitudinal studies showing that depressive symptoms are predictive of cognitive decline (Reference Bassuk, Berkman and WypijBassuk et al, 1998; Reference Chen, Ganguli and MulsantChen et al, 1999; Reference Yaffe, Blackwell and GoreYaffe et al, 1999; Reference Geerlings, Schoevers and BeekmanGeerlings et al, 2000).
Mechanisms of association
Different mechanisms may explain why depressive symptoms predict cognitive decline. Depression may be a psychological reaction to perceived cognitive loss. Schmand et al (Reference Schmand, Jonker and Geerlings1997) found that depressive symptoms at baseline did not predict future dementia when memory complaints were accounted for. In our study depressive symptoms preceded cognitive decline, in the absence of previous loss of cognitive performance; it is therefore unlikely that in our group depression is only a psychological reaction to perceived cognitive loss. On the other hand, it is possible that the MMSE does not permit the evaluation of finer cognitive losses. Cognitive deficits usually associated with depression may be responsible for an earlier onset of cognitive decline or dementia. However, in our study depressive symptoms also predicted cognitive decline in participants with the highest MMSE scores, so it is not likely that this phenomenon can explain all the data.
Somatic symptoms such as fatigue or concentration disorders may be early symptoms of dementia; they are also symptoms of depressive disorders and are included as items of depression rating scales. A high depression score may thus reflect early symptoms of dementia. In our study, the observation that the affective dimension of CES—D was predictive of cognitive decline shows that the observed association between depressive score and subsequent cognitive decline was not only due to the somatic component of depression.
In our study group of people with initial depressive symptoms, persistent (but not episodic) depression was associated with a higher risk of cognitive decline. This result is in agreement with the observation that people with depressive symptoms often suffer from cognitive impairment, which may be severe (pseudo-dementia) but is generally transitory and resolves on treatment of the depression (Reference Stoudemire, Hill and MorrisStoudemire et al, 1993). The fact that initially high levels of depressive symptoms predict subsequent persistent low cognitive functioning together with the observation that persistent but not episodic depressive episodes are associated with cognitive decline suggest that a chronic mechanism is responsible for the observed association. Depressive symptoms may be a prodrome of cognitive decline — the early manifestation of a neurodegenerative process, causing depression and dementia. Depressive symptoms and cognitive decline are linked to modifications in activity of similar cerebral areas (Reference Baker, Frith and DolanBaker et al, 1997). Some studies have found that both are associated with the presence of signal hyperintensities on magnetic resonance brain scans (Reference O'Brien, Desmond and AmesO'Brien et al, 1996). The loss of noradrenergic neurons associated with dementia might also explain depression as a prodrome of dementia (Reference Förstl, Burns and LuthertFörstl et al, 1992). Alternatively, depression might represent a causal factor in cognitive decline. Some studies indicate that early-onset depression (Reference Palsson, Aevarsson and SkoogPalsson et al, 1999) or depression that has a history of 10 years or more (Reference Speck, Kukull and BrennerSpeck et al, 1995) are stronger risk factors for dementia. Some authors have proposed that chronic depression causes cognitive decline by the release of adrenocorticotrophic hormone and the consequent secretion of glucocorticoids (Reference O'BrienO'Brien, 1997). Prolonged secretion of glucocorticoids may have harmful effects and lead to hippocampal atrophy (Reference SapolskySapolsky, 1996).
Practical and clinical implications
Although it is not possible to define causal mechanisms in an epidemiological study, the fact that depression predicts cognitive decline in elderly people has practical and clinical implications. The detection of depressive symptoms in these people is important because early treatment of depression may improve prognosis. In addition, depressive symptoms, especially when they are persistent, may be the first sign of the decrease in cognitive functioning, carrying not only a higher risk of dementia, but also a higher mortality (Reference Gussekloo, Westendorp and RemarqueGussekloo et al, 1997; Reference Rozzini, Franzoni and FrisoniRozzini et al, 1998). People with cognitive decline may benefit from drugs that might slow the progression of the disease. Nevertheless, diagnosis of depression is more difficult in elderly people, in whom symptoms such as fatigue, loss of libido and sleep disturbances occur more frequently even in the absence of depression. A possible consequence is that chronic depression in this age group may be underdiagnosed, because somatic symptoms are attributed to physical rather than psychiatric causes (Reference Hasin and LinkHasin & Link, 1988). In our study only 4% of the group with high levels of depressive symptoms were receiving specific (anti-depressant) treatment. A high proportion of people with chronic depression use benzodiazepines (Reference Bonner and HowardBonner & Howard, 1995), which have been associated in the elderly with a higher risk of cognitive impairment (Reference Gorenstein, Bernik and PompéiaGorenstein et al, 1995).
Limitations of the study
In our study no history of depressive symptoms was obtained and no diagnosis of depressive disorder was made. It cannot therefore be deduced whether early or late depression is responsible for cognitive decline and which clinical form of depression is associated with cognitive impairment. Moreover, study participants who were not followed up had higher levels of depressive symptoms and lower MMSE scores at baseline than those who were followed up. The association between depressive symptoms and cognitive decline may be underestimated. However, few patients were lost to follow-up (13%) and so major bias is unlikely.
In conclusion, our finding that depressive symptoms predict cognitive decline may have important clinical implications. Depression may be a harbinger of initial cognitive decline.
CLINICAL IMPLICATIONS
-
▪ Depressive symptoms predict cognitive decline in elderly people with initially normal functioning, independently of educational level and previous cognitive functioning.
-
▪ Initially, people with depression who have good cognitive functioning are more likely to show a subsequent persistent reduction in cognitive functioning than people who are not depressed.
-
▪ Cognitive decline is more likely in those reporting persistent rather than episodic depression.
LIMITATIONS
-
▪ Without a history of depressive symptoms or a diagnosis of depressive disorder for the patients in this study it is not possible to establish whether early or late depression is responsible for cognitive decline or which clinical form of depression is associated with cognitive decline.
-
▪ Participants who were not followed up (13%) had higher levels of depressive symptoms and lower Mini-Mental State Examination (MMSE) scores at baseline than those who were followed up; the association between depressive symptoms and cognitive decline may therefore be underestimated.
-
▪ Although our data suggest that depressive symptoms predict cognitive decline in study participants with previously stable good performances, it is possible that the MMSE was inadequate to detect more subtle cognitive decline.









eLetters
No eLetters have been published for this article.