1. Introduction
The quantitative study of biochemical reaction networks represents an interdisciplinary research area of (bio)chemistry, physics and mathematics. Enzymes catalyse chemical reactions under physiologically relevant conditions. Enzyme activity directly depends on temperature, pH, ion strength and redox potential of a cell or compartment showing characteristic optima (Arcus & Mulholland, Reference Arcus and Mulholland2020; Bisswanger, Reference Bisswanger2017). In addition, enzyme activity in cellular systems is affected and regulated by diverse biochemical effectors, for example, comprising other proteins and metabolites (Atkinson, Reference Atkinson1969; Chen et al., Reference Chen, Inzé and Vanhaeren2021). As a result, cellular enzyme activity represents a variable of biochemical networks which is shaped by a large parameter space challenging experimental, but also theoretical, analysis. Enzyme kinetic models mathematically describe enzymatic reaction rates as a function of one or more parameters and variables. In general, biochemical kinetics is based on the mass action law assuming the reaction rate to be proportional to the probability of reactant collision (Waage & Gulberg, Reference Waage and Gulberg1864; Reference Waage and Gulberg1986). This probability is proportional (a) to the concentration of reactants, and (b) to the number of molecules of each reactant that participate in a reaction, that is, to the power of molecularity. The rate v of a reaction following the mass action law with molecularities mi and mj of substrates Si and products Pj , respectively, is described by the rate equation (equation (1)):
 $$\begin{align}v={v}_f-{v}_b={k}_f\prod \limits_{i=1}^{l_i}{S}_i^{m_i}-{k}_b\prod \limits_{j=1}^{l_j}{P}_j^{m_j}.\end{align}$$
$$\begin{align}v={v}_f-{v}_b={k}_f\prod \limits_{i=1}^{l_i}{S}_i^{m_i}-{k}_b\prod \limits_{j=1}^{l_j}{P}_j^{m_j}.\end{align}$$
Here, kf and kb represent the rate constants, that is, proportionality factors, for the forward (kf ) and backward (kb ) reaction. Introducing the reversible formation of an enzyme-substrate complex (E + S → ES), a release of product P from ES (ES → E + P), and the simplifying assumption that formation of ES is much faster than its decomposition into E and P, finally yields the Henri–Michaelis–Menten kinetics (Henri, Reference Henri1902; Henri & Hermann, Reference Henri and Hermann1903; Michaelis & Menten, Reference Michaelis and Menten1913). Due to its capability to accurately describe and quantify mechanisms of enzyme catalysis and regulation, the Michaelis–Menten equation is crucial for biochemical understanding (Cornish-Bowden, Reference Cornish-Bowden2015). It was derived based on experimental observations of sucrose hydrolysis, catalysed by invertase enzymes (Brown, Reference Brown1902; Michaelis & Menten, Reference Michaelis and Menten1913). Within this reaction, the glycosidic bond of sucrose is hydrolysed, and glucose and fructose are released (equation (2)):
 $$\begin{align}\overset{r_{\mathrm{in}}}{\to}\mathrm{Suc}\;\overset{r_{\mathrm{in}\mathrm{v}}}{\to}\;\mathrm{Glc}+\mathrm{Frc}\;\overset{\begin{array}{c}{r}_{\mathrm{out},\mathrm{Glc}}\\ {}{r}_{\mathrm{out},\mathrm{Frc}}\end{array}}{\longrightarrow }.\end{align}$$
$$\begin{align}\overset{r_{\mathrm{in}}}{\to}\mathrm{Suc}\;\overset{r_{\mathrm{in}\mathrm{v}}}{\to}\;\mathrm{Glc}+\mathrm{Frc}\;\overset{\begin{array}{c}{r}_{\mathrm{out},\mathrm{Glc}}\\ {}{r}_{\mathrm{out},\mathrm{Frc}}\end{array}}{\longrightarrow }.\end{align}$$
In this kinetic model, r in represents the rate of sucrose biosynthesis, r inv the rate of invertase-driven hydrolysis and r out,Glc and r out,Frc hexose consuming processes, for example, phosphorylation by hexokinase enzymes. The corresponding ODE model of this reaction system describes sucrose, fructose and glucose dynamics by the sum of in- and effluxes (equations (3)–(5)):
The right side of the ODEs, that is, the sum of reactions, is summarised by metabolic functions f and their integration yields the time course of metabolite concentrations. Dynamics of substrate and product concentrations can then be described by differential equations (DEs). If concentration dynamics are considered (only) over time, ordinary differential equations (ODEs) are applied while partial DEs account for more than one independent variable, for example, time and space.
In the following paragraph, invertase-catalysed sucrose hydrolysis is quantitatively explained and analysed down to a subcellular level applying an enzyme kinetic model based on Michaelis–Menten enzyme kinetics and experimental data of previous studies (further details about mathematical analysis are provided in the Supplementary Material). Invertases play a central role in diverse processes of plant metabolism, development and response to environmental stress (Koch, Reference Koch2004; Ruan, Reference Ruan2014; Vu et al., Reference Vu, Martins Rodrigues, Jung, Meissner, Klemens, Holtgrawe, Furtauer, Nagele, Nieberl, Pommerrenig and Neuhaus2020; Weiszmann et al., Reference Weiszmann, Fürtauer, Weckwerth and Nägele2018; Xiang et al., Reference Xiang, Le Roy, Bolouri-Moghaddam, Vanhaecke, Lammens, Rolland and Van den Ende2011). Plant invertases occur in different isoforms with different compartmental localisation and biochemical properties (Sturm, Reference Sturm1996; Tymowska-Lalanne & Kreis, Reference Tymowska-Lalanne and Kreis1998). Both plant vacuolar and extracellular invertases possess an acidic pH optimum between 4.5 and 5.0 while cytosolic invertase has a neutral pH optimum between 7.0 and 7.8 (Sturm, Reference Sturm1999). Acidic and neutral invertases hydrolyse sucrose with a K M in a low-millimolar range (Sturm, Reference Sturm1999; Unger et al., Reference Unger, Hofsteenge and Sturm1992). Invertases are product inhibited, with glucose acting as a non-competitive inhibitor and fructose as a competitive inhibitor (Sturm, Reference Sturm1999). While biochemistry and kinetics of plant invertase reactions have been analysed in numerous studies, the physiological role of different subcellular isoforms and their regulatory impact on plant stress and acclimation reactions remain elusive. Further, due to its participation in cyclic sucrose breakdown and re-synthesis, the experimental study of invertase reactions remains challenging, particularly under changing environmental conditions. Due to such cycling structures, it remains difficult to estimate metabolite amounts, their dynamics and effects on other segments of metabolic networks (Reznik & Segrè, Reference Reznik and Segrè2010). Previous work has suggested a dominant role of invertase-driven sucrose cycling in regulation and stabilisation of primary metabolism and photosynthesis (Geigenberger & Stitt, Reference Geigenberger and Stitt1991; Weiszmann et al., Reference Weiszmann, Fürtauer, Weckwerth and Nägele2018). Here, metabolic functions of sucrose and hexoses are quantified to analyse compartment-specific invertase reactions in context of subcellular metabolite transport during plant cold exposure to evaluate its impact on metabolic acclimation.
2. Results and discussion
Due to the regulatory plasticity of metabolism, metabolite concentrations may vary significantly under similar environmental conditions and without stress exposure. For example, sucrose and hexoses may accumulate significantly, and even double in amount, during the light period of a diurnal cycle (Brauner et al., Reference Brauner, Hörmiller, Nägele and Heyer2014; Seydel et al., Reference Seydel, Biener, Brodsky, Eberlein and Nägele2022; Sulpice et al., Reference Sulpice, Flis, Ivakov, Apelt, Krohn, Encke, Abel, Feil, Lunn and Stitt2014). Such strong dynamics of reaction product and substrate concentrations aggravate the quantitative analysis of metabolic regulation due to their non-linear impact on enzymatic rates. It follows that instead of analysing one (single) snapshot, a broad range of physiologically relevant metabolite concentrations and/or enzyme parameters needs to be analysed in order to cope with metabolic plasticity. Here, an example of such an analysis is provided applying a kinetic parameter set of invertase reactions (Table 1), which has previously been determined in Arabidopsis thaliana under ambient (22°C) and low (4°C) temperature (Kitashova et al., Reference Kitashova, Schneider, Furtauer, Schroder, Scheibenbogen, Furtauer and Nagele2021).
Table 1 Carbon uptake rates and kinetic parameters of invertase-catalysed sucrose hydrolysis in Arabidopsis thaliana, accession Col-0, at 22 and 4°C.

Reaction rates of invertase enzymes, r
inv, were calculated across different combinations of physiologically relevant sucrose and hexose concentrations to determine the metabolic function of sucrose, that is, f(Suc) = r
in − r
inv. Simulation results of different sucrose concentrations were plotted against glucose and fructose concentrations (Figure 1). Thus, each shown plane in the figure corresponds to solutions of f(Suc), J and H for one sucrose concentration (a detailed definition of concentrations is provided in the figure legend). Although sucrose concentrations used for 4°C simulations were up to 8-fold higher than under 22°C, resulting absolute values and dynamics of f(Suc) were significantly lower than under 22°C (Figure 1a,b). Reduced absolute values were due to a decreased input rate r
in,4°C (based on experimental findings). As expected, under conditions of low product concentration, f(Suc) became minimal under both temperatures due to increased rates of sucrose cleavage (Figure 1a,b). However, reduced dynamics of f(Suc) was due to increased hexose concentrations (inhibitors) and a reduced V
max of invertase (see Table 1). As a result, also the dynamic range of J and H decreased across all simulated scenarios by several orders of magnitude (10−1 → 10−4/10−5; Figure 1c–f). A main low temperature effect became visible in entries of Jacobian matrices which was a reduced degree of overlap between j
12
![]() $\big(\frac{\partial \left(f\left(\mathrm{Suc}\right)\right)}{\partial \left(\mathrm{Glc}\right)}\big)$
and j
13
$\big(\frac{\partial \left(f\left(\mathrm{Suc}\right)\right)}{\partial \left(\mathrm{Glc}\right)}\big)$
and j
13
![]() $\big(\frac{\partial \left(f\left(\mathrm{Suc}\right)\right)}{\partial \left(\mathrm{Frc}\right)}\big)$
(Figure 1c,d). Both terms describe changes of f(Suc) induced by (slight) changes of glucose and fructose concentrations, respectively. At 22°C, high glucose concentrations (~ 2.5−3 μmol gFW−1) minimise j
13 and, with this, also the regulatory effect of fructose dynamics on f(Suc) (see Figure 1c). At 4°C, high glucose concentrations (~ 14−15 μmol gFW−1) also lead to minimal values of j
13, which were, however, still significantly higher than j
12 (see Figure 1d; ANOVA, p < .001). This discrepancy became also visible in the curvature of f(Suc), that is, in the Hessian matrix (Figure 1e,f).
$\big(\frac{\partial \left(f\left(\mathrm{Suc}\right)\right)}{\partial \left(\mathrm{Frc}\right)}\big)$
(Figure 1c,d). Both terms describe changes of f(Suc) induced by (slight) changes of glucose and fructose concentrations, respectively. At 22°C, high glucose concentrations (~ 2.5−3 μmol gFW−1) minimise j
13 and, with this, also the regulatory effect of fructose dynamics on f(Suc) (see Figure 1c). At 4°C, high glucose concentrations (~ 14−15 μmol gFW−1) also lead to minimal values of j
13, which were, however, still significantly higher than j
12 (see Figure 1d; ANOVA, p < .001). This discrepancy became also visible in the curvature of f(Suc), that is, in the Hessian matrix (Figure 1e,f).
These observations suggest that, under ambient conditions and (increased) glucose concentrations, it is j
12 ≈ j
13, that is,
![]() $\frac{\partial \left(f\left(\mathrm{Suc}\right)\right)}{\partial \left(\mathrm{Glc}\right)}\approx \frac{\partial \left(f\left(\mathrm{Suc}\right)\right)}{\partial \left(\mathrm{Frc}\right)}$
, and
$\frac{\partial \left(f\left(\mathrm{Suc}\right)\right)}{\partial \left(\mathrm{Glc}\right)}\approx \frac{\partial \left(f\left(\mathrm{Suc}\right)\right)}{\partial \left(\mathrm{Frc}\right)}$
, and
![]() ${h}_{f\left(\mathrm{Suc}\right),12}\approx {h}_{f\left(\mathrm{Suc}\right),13}$
, that is,
${h}_{f\left(\mathrm{Suc}\right),12}\approx {h}_{f\left(\mathrm{Suc}\right),13}$
, that is,
![]() $\frac{\partial^2\left({r}_{\mathrm{in}}-{r}_{\mathrm{in}\mathrm{v}}\right)}{\partial \left(\mathrm{Suc}\right)\partial \left(\mathrm{Glc}\right)}\approx \frac{\partial^2\left({r}_{\mathrm{in}}-{r}_{\mathrm{in}\mathrm{v}}\right)}{\partial \left(\mathrm{Suc}\right)\partial \left(\mathrm{Frc}\right)}$
. At low temperature, this similarity is not given even under (relatively) high glucose concentrations which might suggest a cold-induced switch of the regulatory role which fructose plays in plant metabolism (Klotke et al., Reference Klotke, Kopka, Gatzke and Heyer2004).
$\frac{\partial^2\left({r}_{\mathrm{in}}-{r}_{\mathrm{in}\mathrm{v}}\right)}{\partial \left(\mathrm{Suc}\right)\partial \left(\mathrm{Glc}\right)}\approx \frac{\partial^2\left({r}_{\mathrm{in}}-{r}_{\mathrm{in}\mathrm{v}}\right)}{\partial \left(\mathrm{Suc}\right)\partial \left(\mathrm{Frc}\right)}$
. At low temperature, this similarity is not given even under (relatively) high glucose concentrations which might suggest a cold-induced switch of the regulatory role which fructose plays in plant metabolism (Klotke et al., Reference Klotke, Kopka, Gatzke and Heyer2004).

Fig. 1. Dynamics of f(Suc) under ambient and low temperature. (a) f(Suc) at 22°C under variable concentrations of fructose (x-axis), glucose (y-axis) and sucrose (planes). (b) f(Suc) at 4°C under variable concentrations of fructose (x-axis), glucose (y-axis) and sucrose (planes). Unit of f(Suc): (μmol Suc h−1 gFW−1). (c) Jacobian matrix entries of f(Suc) at 22°C under variable concentrations of glucose (x-axis), fructose (y-axis) and sucrose (planes; see equation S5 in the supplements; j 11: blue; j 12: green; j 13: grey). (d) Jacobian matrix entries of f(Suc) at 4°C under variable concentrations of glucose (x-axis), fructose (y-axis) and sucrose (planes). See equation S5 in the supplements; J 11: blue; J 12: green; J 13: grey. (e) Hessian matrix entries of f(Suc) at 22°C under variable concentrations of glucose (x-axis), fructose (y-axis) and sucrose (planes), see equation S11 in the supplements; h f(Suc,11): blue; h f(Suc,12): green; h f(Suc,13): grey. (f) Hessian matrix entries of f(Suc) at 4°C under variable concentrations of glucose (x-axis), fructose (y-axis) and sucrose (planes), see equation S11 in the supplements; h f(Suc,11): blue; h f(Suc,12): green; h f(Suc,13): grey. Each plane corresponds to a sucrose concentration which was varied between 1−3 μmol gFW−1 and 4−8 μmol gFW−1 for simulations at 22 and 4°C, respectively.
3. Vacuolar metabolite transport increases elasticity of the cellular sucrose function
To study a regulatory effect of hexose accumulation under low temperature in more detail, the model was extended to subcellular distribution of sugars and invertase isoforms (Figure 2). Effective cytosolic and vacuolar sugar concentrations in Arabidopsis leaf mesophyll cells were estimated as previously described assuming the cytosol to comprise 5% and the vacuole 80% of the total cell volume (Nägele & Heyer, Reference Nägele and Heyer2013). Further, subcellular sugar distribution at 22 and 4°C, respectively, was derived from a previous study (Fürtauer et al., Reference Fürtauer, Weckwerth and Nägele2016). Details about the relative distribution of sugars are provided below (see legend of Figure 2).

Fig. 2. Schematic overview of subcellular invertase reactions. Green colour indicates cytosolic metabolites and enzymes and blue colour indicates vacuolar metabolites and enzymes. For simulation of subcellular sucrose cleavage, effective metabolite concentrations were calculated based on assumptions and findings of previous studies (Kitashova et al., Reference Kitashova, Schneider, Furtauer, Schroder, Scheibenbogen, Furtauer and Nagele2021; Nägele & Heyer, Reference Nägele and Heyer2013). Based on previous findings (Fürtauer et al., Reference Fürtauer, Weckwerth and Nägele2016), subcellular sugar distribution was assumed as follows: cytosolic sucrose, 22°C: 50%; vacuolar sucrose, 22°C: 25%; cytosolic hexoses, 22°C: 30%; vacuolar hexoses, 22°C: 55%; cytosolic sucrose, 4°C: 40%; vacuolar sucrose, 4°C: 33%; cytosolic hexoses, 4°C: 30%; vacuolar hexoses, 4°C: 50%.
Assuming a volume of 1 ml H2O to equal (approximately) 1 g fresh weight of Arabidopsis leaf material (Nägele & Heyer, Reference Nägele and Heyer2013), effective sugar concentrations were derived from the sugar amounts used before (see Figure 1; Kitashova et al., Reference Kitashova, Schneider, Furtauer, Schroder, Scheibenbogen, Furtauer and Nagele2021). To simulate compartment-specific sucrose cleavage, neutral (cytosolic) and acidic (vacuolar) invertase activities were considered separately (Kitashova et al., Reference Kitashova, Schneider, Furtauer, Schroder, Scheibenbogen, Furtauer and Nagele2021). Subcellular simulations of f(Suc) across a physiologically feasible range of metabolite concentrations at 22°C revealed higher variability of vacuolar metabolic functions than in the cytosol (Figure 3a,d). Entries of Jacobian and Hessian matrices, which accounted for changes in substrate and/or product concentrations, differed by orders of magnitude between cytosolic and vacuolar reactions (Figure 3b,c,e,f).

Fig. 3. Estimated cytosolic and vacuolar dynamics of f(Suc) under ambient and low temperature at variable concentrations of fructose, glucose and sucrose. Planes represents simulations for different sucrose concentrations (sucrose concentration ranges: cytosol, 22°C: 8–24 mM; vacuole, 22°C: 0.25–0.75 mM; cytosol, 4°C: 25.6–51.2 mM; vacuole, 4°C: 1.32–2.64 mM). (a–f) estimations at 22°C, (g–l) estimations at 4°C. (a) cytosolic f(Suc) at 22°C, (b) Jacobian entries j 11 (blue), j 12 (green), j 13 (grey) of cytosolic f(Suc) at 22°C, (c) Hessian entries h f(Suc,11) (blue), h f(Suc,12) (green), h f(Suc,13) (grey) of cytosolic f(Suc) at 22°C, (d) vacuolar f(Suc) at 22°C, (e) Jacobian entries j 11 (blue), j 12 (green), j 13 (grey) of vacuolar f(Suc) at 22°C, (f) Hessian entries h f(Suc,11) (blue), h f(Suc,12) (green), h f(Suc,13) (grey) of vacuolar f(Suc) at 22°C, (g) cytosolic f(Suc) at 4°C, (h) Jacobian entries j 11 (blue), j 12 (green), j 13 (grey) of cytosolic f(Suc) at 4°C, (i) Hessian entries h f(Suc,11) (blue), h f(Suc,12) (green), h f(Suc,13) (grey) of cytosolic f(Suc) at 4°C, (j) vacuolar f(Suc) at 4°C, (k) Jacobian entries j 11 (blue), j 12 (green), j 13 (grey) of vacuolar f(Suc) at 4°C, (l) Hessian entries h f(Suc,11) (blue), h f(Suc,12) (green), h f(Suc,13) (grey) of vacuolar f(Suc) at 4°C. Colour bars in the left panel (a,d,g,j) indicate values of f(Suc).
These differences were due to 16-fold dilution of metabolites comparing the vacuolar and cytosolic volume (5 vs. 80% of the total cell volume). Hence, lowered effective metabolite concentrations in the vacuole resulted in higher elasticity of f(Suc) due to lowered invertase inhibition by glucose and fructose. At 4°C, the discrepancy between concentration effects on f(Suc) in cytosol and vacuole became stronger due to significant cold-induced sugar accumulation (Figure 3g,j). Under these conditions, f(Suc) in the cytosol was almost invariant across the simulated sucrose concentration range of 25.6–51.2 mM (Figure 3g) while dynamics were still observable for the vacuolar f(Suc) (Figure 3j). This was numerically reflected in Jacobian and Hessian matrix entries of the subcellular metabolic function of f(Suc) which revealed dynamics of vacuolar fructose concentration to have the strongest regulatory effect within the simulated scenario (Figure 3h,i,k,l).
Together with the findings of the whole cell model (Figure 1), these observations suggest that transport of sucrose into the vacuole maximise effects of metabolic regulation on f(Suc) and provide further evidence for a dominant role of fructose in regulation of sucrose cleavage under low temperature. Previous studies have shown that sugar accumulation, in general, plays a central role in plant cold response and acclimation (Guy et al., Reference Guy, Kaplan, Kopka, Selbig and Hincha2008; Hannah et al., Reference Hannah, Wiese, Freund, Fiehn, Heyer and Hincha2006; Seydel, Kitashova, et al., Reference Seydel, Kitashova, Fürtauer and Nägele2022). Fructose and its phosphorylation product, fructose 6-phosphate (F6P), have been found to significantly contribute to stabilisation of a plant metabolic homeostasis during cold exposure (Bogdanović et al., Reference Bogdanović, Mojović, Milosavić, Mitrović, Vučinić and Spasojević2008). F6P is a direct product of the Calvin Benson Cycle and serves as substrate for many other metabolic pathways, for example, starch biosynthesis, sucrose biosynthesis and glycolysis (Ruan, Reference Ruan2014). Thus, findings of the present study suggest that tight regulation of f(Suc) in the cytosol and vacuole by fructose directly connects sucrose dynamics with the stabilisation of many other cellular pathways. In future studies, a combination of the presented kinetic approach with subcellular sugar analysis of mutants being affected in sucrose cleavage and subcellular sugar transport might reveal further detailed insights into the regulatory network of plant sucrose metabolism.
4. Conclusions
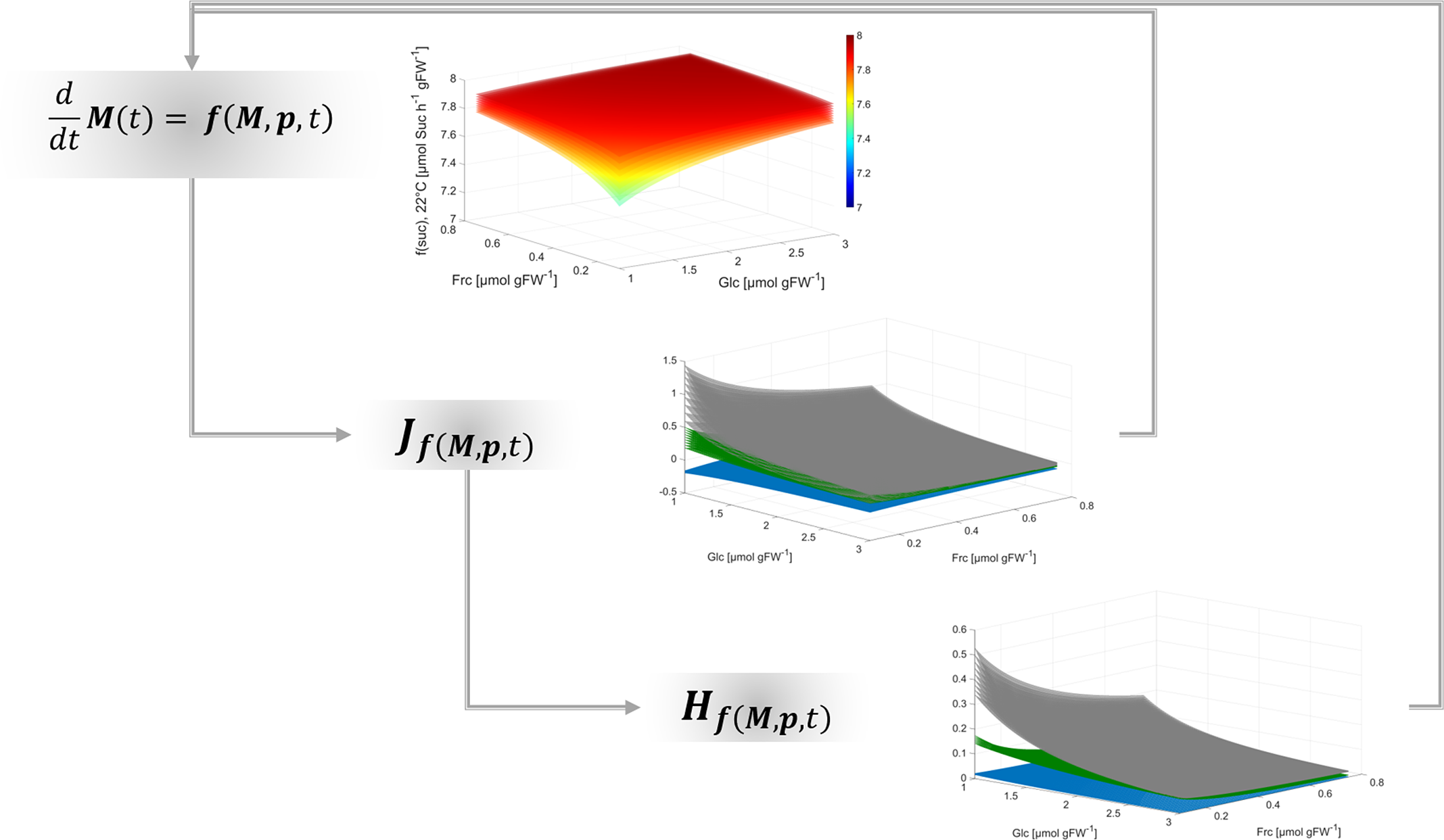
Together with the Jacobian matrix, Hessian matrices are commonly applied to study n-dimensional functions and surfaces, their extrema and their curvature (see e.g., (Basterrechea & Dacorogna, Reference Basterrechea and Dacorogna2014; Ivochkina & Filimonenkova, Reference Ivochkina and Filimonenkova2019). In context of the presented theory for analysis of biochemical metabolic functions, this suggests that metabolism can be summarised by a multi-dimensional function which supports the analysis of complex metabolic regulation, for example, of metabolic cycling. Although calculation of metabolic functions, Jacobian and Hessian matrices is straight forward, it essentially supports quantitative analysis of multi-dimensional dynamics, shape and curvature of a metabolic landscape (Figure 4).

Fig. 4. Workflow for deriving regulatory principles of metabolism.
Findings of the present study emphasise the necessity to resolve eukaryotic metabolism to a subcellular level in order to reliably estimate dynamics of metabolite concentrations in terms of reaction rates and transport processes. Finally, applying such analysis to dynamic metabolic systems can unravel non-intuitive regulatory patterns. This supports the quantitative interpretation of experimental observations on metabolism within a dynamic environment.
Acknowledgements
I would like to thank all members of Plant Evolutionary Cell Biology, LMU Munich, for many fruitful seminars and discussions. Special thanks go to Lisa Fürtauer, RWTH Aachen, and AG Weckwerth, University of Vienna, as well as Jakob Weiszmann and Matthias Nagler for constructive advice, support and discussion. Finally, I thank the SFB/TR175 consortium for a supportive research environment and fruitful discussions.
Financial support
This work was supported by Deutsche Forschungsgemeinschaft (DFG), grants TR175/D03 and NA 1545/4-1.
Conflict of interest
The author declares no conflicts of interest.
Data availability statement
The main data supporting the findings of this study are contained within the article and cited literature.
Supplementary Materials
To view supplementary material for this article, please visit http://doi.org/10.1017/qpb.2022.5.










Comments
Dear Dr. Hamant,
Dear Dr. Fleck,
I am submitting a theories article to be considered for publication in your journal Quantitative Plant Biology. The article is entitled “Metabolic regulation inferred from Jacobian and Hessian matrices of metabolic functions”. It presents a theoretical approach to develop first-order and second-order partial derivatives of metabolic functions summarised by Jacobian and Hessian matrices. Applying a simple kinetic model of invertase-driven sucrose hydrolysis, an approach was developed for quantitative analysis of kinetic regulation of sucrose metabolism based on partial derivatives of metabolic functions. Based on previously published experimental observations, metabolite dynamics were quantitatively explained in context of underlying metabolic functions and enzyme kinetics. I applied this approach to explain differential regulation of sucrose and hexose concentrations in Arabidopsis thaliana during cold acclimation. Finally, a switch of the metabolic role of fructose metabolism is suggested to be involved in metabolic reprogramming during plant cold acclimation. The manuscript has been made publicly available on the bioRxiv pre-print server (https://doi.org/10.1101/2021.10.05.463227).
In summary, I am convinced that this article is of high interest for the readership of Quantitative Plant Biology. I would be pleased to publish it within your journal and thank you in advance for editing my manuscript.
Sincerely yours,
Thomas Nägele