Introduction
The coronavirus disease 2019 (COVID-19) pandemic has dramatically impacted families globally, exacerbating existing stressors and racial and socioeconomic inequities across a wide range of psychological and health domains (Purtle, Reference Purtle2020; Tai, Shah, Doubeni, Sia, & Wieland, Reference Tai, Shah, Doubeni, Sia and Wieland2021). Common pandemic stressors include health and economic concerns, social isolation, and restrictions on movement (Ammar et al., Reference Ammar, Brach, Trabelsi, Chtourou, Boukhris, Masmoudi and Ahmed2020; Hall, Laddu, Phillips, Lavie, & Arena, Reference Hall, Laddu, Phillips, Lavie and Arena2021). Epidemiological studies have reported an increased prevalence of pandemic-related psychiatric morbidity and psychological distress in the general population (Lei et al., Reference Lei, Huang, Zhang, Yang, Yang and Xu2020; Smith et al., Reference Smith, Jacob, Yakkundi, McDermott, Armstrong, Barnett and Tully2020) with effects projected to continue beyond the current pandemic (Cullen, Gulati, & Kelly, Reference Cullen, Gulati and Kelly2020). Studies have also shown increases in the prevalence of psychological distress among women who were pregnant during the COVID-19 pandemic (Berthelot et al., Reference Berthelot, Lemieux, Garon-Bissonnette, Drouin-Maziade, Martel and Maziade2020; King, Feddoes, Kirshenbaum, Humphreys, & Gotlib, Reference King, Feddoes, Kirshenbaum, Humphreys and Gotlib2020; Lebel, MacKinnon, Bagshawe, Tomfohr-Madsen, & Giesbrecht, Reference Lebel, MacKinnon, Bagshawe, Tomfohr-Madsen and Giesbrecht2020). These trends are particularly concerning given the large body of literature linking prenatal stress and distress with adverse intrauterine development and birth outcomes, such as preterm birth (PTB, < 37 weeks gestation) and low infant birthweight (LBW, <2500 g) (Harville, Xiong, & Buekens, Reference Harville, Xiong and Buekens2010; Lima et al., Reference Lima, El Dib, Rodrigues, Ferraz, Molina, Neto and Rudge2018; Stein et al., Reference Stein, Pearson, Goodman, Rapa, Rahman, McCallum and Pariante2014). Although evidence suggests that exposure to stress during pregnancy leads to negative birth outcomes, in part via heightened maternal psychological distress (e.g. depressive symptoms) (Glover, Reference Glover and Antonelli2015), there has been limited opportunity to examine the impact of the pandemic as a stressor on prenatal mental health as most studies have been descriptive in nature.
Globally, there have been inconsistent findings about the effect of the pandemic on rates of PTB and low birthweight (Ashish et al., Reference Ashish, Gurung, Kinney, Sunny, Moinuddin, Basnet and Shrestha2020; Been et al., Reference Been, Ochoa, Bertens, Schoenmakers, Steegers and Reiss2020; Hedermann et al., Reference Hedermann, Hedley, Bækvad-Hansen, Hjalgrim, Rostgaard, Poorisrisak and Christiansen2021; Kirchengast & Hartmann, Reference Kirchengast and Hartmann2021; Matheson et al., Reference Matheson, McGannon, Malhotra, Palmer, Stewart, Wallace and Rolnik2021; Pasternak et al., Reference Pasternak, Neovius, Söderling, Ahlberg, Norman, Ludvigsson and Stephansson2021; Philip et al., Reference Philip, Purtill, Reidy, Daly, Imcha, McGrath and Dunne2020). Similarly, in the United States, some studies have reported overall reductions in PTB (Berghella, Boelig, Roman, Burd, & Anderson, Reference Berghella, Boelig, Roman, Burd and Anderson2020; Harvey et al., Reference Harvey, McNeer, McDonald, Shapiro-Mendoza, Dupont, Barfield and Patrick2021) or reductions specific to women of White race or from more advantaged neighborhoods (Lemon, Edwards, & Simhan, Reference Lemon, Edwards and Simhan2021) relative to rates before the COVID-19 pandemic onset. Other studies have reported no differences (Greene, Kilpatrick, Wong, Ozimek, & Naqvi, Reference Greene, Kilpatrick, Wong, Ozimek and Naqvi2020; Handley et al., Reference Handley, Mullin, Elovitz, Gerson, Montoya-Williams, Lorch and Burris2021; Wood et al., Reference Wood, Sinnott, Goldfarb, Clapp, McElrath and Little2021) or increased rates of very preterm birth specifically among Hispanic or Latinx women (Main et al., Reference Main, Chang, Carpenter, Wise, Stevenson, Shaw and Gould2021). Results are also equivocal with regard to birthweight, with variable evidence for greater infant birthweight (Kirchengast & Hartmann, Reference Kirchengast and Hartmann2021; Yang et al., Reference Yang, D'souza, Kharrat, Fell, Snelgrove, Murphy and Shah2021), reduced rates of very low birthweight (Philip et al., Reference Philip, Purtill, Reidy, Daly, Imcha, McGrath and Dunne2020), or no change (Chmielewska et al., Reference Chmielewska, Barratt, Townsend, Kalafat, van der Meulen, Gurol-Urganci and Thangaratinam2021; Matheson et al., Reference Matheson, McGannon, Malhotra, Palmer, Stewart, Wallace and Rolnik2021) relative to pandemic exposure.
While partly attributable to geographic differences in the timing and extent of pandemic mitigation measures, these mixed results may also reflect differences in the quality and rigor of study designs. Most birth outcome studies have drawn on electronic records to compare rates of PTB and infant birthweight categories before or during the pandemic. However, record-based studies have limited data on important covariates (e.g. maternal race/ethnicity, socioeconomic status) that are associated with experiences of stress and birth outcomes. Because the pandemic disproportionally affected people of color and individuals in low resourced environments (Maroko, Nash, & Pavilonis, Reference Maroko, Nash and Pavilonis2020), these studies cannot clarify whether the pandemic itself is a driving factor of health outcomes. Methods such as propensity-score matching, a quasi-experimental approach, enable the risks for birth outcomes conferred by the pandemic to be examined separately from those related to sociodemographic factors.
Individual differences in daily behavior and social interactions could also modify the impact of the pandemic on prenatal distress and subsequent birth outcomes. For some individuals, social isolation, loss of daily routines, and enforced working from home led to increased time in sedentary behaviors (Stockwell et al., Reference Stockwell, Trott, Tully, Shin, Barnett, Butler and Smith2021). Time spent in sedentary behavior is a known risk factor for poor health outcomes, independent of physical activity levels (Clark et al., Reference Clark, Sugiyama, Healy, Salmon, Dunstan and Owen2009; Pate, O'Neill, & Lobelo, Reference Pate, O'Neill and Lobelo2008), and is also associated bi-directionally with mental health problems and perceived stress (Chekroud et al., Reference Chekroud, Gueorguieva, Zheutlin, Paulus, Krumholz, Krystal and Chekroud2018). Although pregnant women typically spend more than 50% of their waking hours in sedentary behaviors (Fazzi, Saunders, Linton, Norman, & Reynolds, Reference Fazzi, Saunders, Linton, Norman and Reynolds2017), evidence for birth outcome risks is unclear (Both, Overvest, Wildhagen, Golding, & Wildschut, Reference Both, Overvest, Wildhagen, Golding and Wildschut2010; Reid, McNeill, Alderdice, Tully, & Holmes, Reference Reid, McNeill, Alderdice, Tully and Holmes2014; Ruifrok et al., Reference Ruifrok, Althuizen, Oostdam, Van Mechelen, Mol, De Groot and Van Poppel2014). Sedentary behavior may exacerbate the impact of the pandemic on prenatal psychological distress and subsequent birth outcomes.
Studies examining pandemic-related health in pregnant women have focused largely on negative impacts. However, to inform strengths-based preventative care, studies need to identify protective factors that can be readily implemented to improve prenatal health and support positive birth outcomes. One factor broadly linked to stress resilience is perceived social support (Panagioti, Gooding, Taylor, & Tarrier, Reference Panagioti, Gooding, Taylor and Tarrier2014; Sim, Bowes, & Gardner, Reference Sim, Bowes and Gardner2019), particularly emotional support that has been shown to influence stress physiology during pregnancy and may buffer the effects of stress on health outcomes (Coburn, Gonzales, Luecken, & Crnic, Reference Coburn, Gonzales, Luecken and Crnic2016; Nierop, Wirtz, Bratsikas, Zimmermann, & Ehlert, Reference Nierop, Wirtz, Bratsikas, Zimmermann and Ehlert2008; Tung et al., Reference Tung, Krafty, Delcourt, Melhem, Jennings, Keenan and Hipwell2021). Although some evidence suggests that higher levels of prenatal support during the pandemic are associated with less psychological distress (Lebel et al., Reference Lebel, MacKinnon, Bagshawe, Tomfohr-Madsen and Giesbrecht2020), no studies to our knowledge have directly investigated emotional support as a psychosocial buffer of pandemic effects.
Studies have reported mixed findings with respect to the impact of the COVID-19 pandemic on pregnant women and birth outcomes; differences that may be explained in part by sampling, geographical differences and social determinants of health. In the current study, we used propensity-score matching to examine the effects of the pandemic on psychological distress (i.e. perceived stress, depressive symptoms) during pregnancy and on birth outcomes (see conceptual model in Fig. 1). We hypothesized that after controlling for potential sociodemographic confounds, pandemic exposure would be associated with shorter infant gestational age and lower birthweight for gestational age. We also hypothesized that perceived stress and depressive symptoms during pregnancy would mediate the association between pandemic exposure and adverse birth outcomes. Finally, we expected that sedentary behavior would exacerbate, and emotional support would buffer, the negative effects of the pandemic on both prenatal distress and birth outcomes.

Fig. 1. Conceptual model.
Methods
Participants
The Environmental influences on Child Health Outcomes (ECHO) Program is an NIH-funded nationwide consortium of multiple cohort studies across the United States designed to investigate the effects of early life exposures on child health and development (Paneth & Monk, Reference Paneth and Monk2018; Romano, Buckley, Elliott, Johnson, Paneth, & program collaborators for Environmental influences on Child Health Outcomes, Reference Romano, Buckley, Elliott, Johnson and Paneth2022). The ECHO program combines existing prenatal and pediatric data collected via cohort-specific protocols with a standardized ECHO-wide protocol that was established in 2019 (Gillman & Blaisdell, Reference Gillman and Blaisdell2018; Knapp et al., Reference Knapp, Kress, Parker, Page, McArthur, Gachigi and Jacobsonin press; LeWinn, Caretta, Davis, Anderson, Oken & program collaborators for Environmental influences on Child Health Outcomes, Reference LeWinn, Caretta, Davis, Anderson and Oken2022) (https://echochildren.org/echo-program-protocol/). The ECHO study was approved by the local and/or central ECHO Institutional Review Board, and written informed consent was obtained for participation in specific cohorts and the ECHO-wide data collection protocol.
The current study focused on individuals enrolled in prenatal ECHO cohorts who had a singleton gestation pregnancy and who delivered during or before the COVID-19 pandemic. Between 12 March 2020 and 30 May 2021, 501 pandemic-exposed pregnant women delivered a live infant and had complete sociodemographic data on maternal age, race and ethnicity, and child sex assigned at birth. Given that the decision or ability to participate in research before and during a pandemic may vary for different individuals as a function of sociodemographic characteristics, and evidence that child sex differences can emerge under conditions of stress (Walsh et al., Reference Walsh, McCormack, Webster, Pinto, Lee, Feng and Champagne2019), participants were propensity-score-matched in a 1:1 (pandemic: pre-pandemic) design on the above sociodemographic characteristics with 501 women who delivered before the pandemic onset between 1 January 2009 and 10 March 2020.
Measures
Birth outcomes
Gestational age at birth (GA) in completed weeks and infant birthweight (in grams) were obtained from maternal medical record abstraction (15% GA; 5% birthweight), childbirth/neonatal medical record abstraction (28% GA; 38% birthweight), childbirth information (18% GA; 32% birthweight) or other maternal report (39% GA; 25% birthweight). Sex-specific birthweight adjusted for gestational age z scores (BWGA-z scores) were assigned based on prior work (Aris, Kleinman, Belfort, Kaimal, & Oken, Reference Aris, Kleinman, Belfort, Kaimal and Oken2019).
Sociodemographic variables
Sociodemographic variables were obtained from maternal medical record abstraction, childbirth/neonatal medical record abstraction, or via maternal report during pregnancy, depending on the ECHO cohort. Maternal age at delivery was calculated from maternal and child dates of birth. Maternal self-reported race was categorized as American Indian or Alaska Native, Asian, Black, White, multiple race, and other race. Self-reported ethnicity was categorized as Hispanic or non-Hispanic. Child sex assigned at birth was coded as female or male.
Psychological distress
Severity of prenatal stress was assessed via self-report using the Perceived Stress Scale [PSS, (Cohen, Kamarck, & Mermelstein, Reference Cohen, Kamarck and Mermelstein1983). Three versions of the PSS (consisting of 4, 10, or 14 items) were administered across the ECHO cohorts; each item was rated on a 5-point Likert scale. Raw scores were normed to a common, standardized T score metric (Mean = 50, s.d. = 10) (McDonald, Reference McDonald1999). Maternal depressive symptoms during pregnancy were measured by self-report on at least one of the following: (1) the Patient Reported Outcomes Measurement Information System (PROMIS) Depression 8a (Cella et al., Reference Cella, Riley, Stone, Rothrock, Reeve, Yount and Choi2010; Pilkonis et al., Reference Pilkonis, Choi, Reise, Stover, Riley, Cella and Group2011); (2) the Edinburgh Postnatal Depression Scale (Cox, Holden, & Sagovsky, Reference Cox, Holden and Sagovsky1987); (3) the Adult Self-Report Achenbach System Depression Problems Syndrome Scale (Rescorla & Achenbach, Reference Rescorla, Achenbach and Maruish2004); (4) the Brief Symptom Inventory (Derogatis & Melisaratos, Reference Derogatis and Melisaratos1983); (5) the Center for Epidemiological Studies Depression Scale (Radloff & Locke, Reference Radloff, Locke, Weissman, Meyer and Ross1986); (6) the Patient Health Questionnaire-9 (Kroenke, Spitzer, & Williams, Reference Kroenke, Spitzer and Williams2001); (7) the Beck Depression Inventory (Beck & Steer, Reference Beck and Steer1984); and (8) the Kessler 6 Mental Health Scale (Kessler et al., Reference Kessler, Barker, Colpe, Epstein, Gfroerer, Hiripi and Walters2003). Depression measures were harmonized to the PROMIS T score metric using validated crosswalk tables (Blackwell et al., Reference Blackwell, Tang, Elliott, Thomes, Louwagie, Gershon and Cella2021; Reference Cella, Schalet, Kallen, Lai, Cook, Rutsohn and ChoiCella et al.,; Choi et al., Reference Choi, Podrabsky, McKinney, Schalet, Cook and Cella2022; Kaat, Newcomb, Ryan, & Mustanski, Reference Kaat, Newcomb, Ryan and Mustanski2017). After harmonization, depression scores were expected to have a mean of 50 and standard deviation (s.d.) of 10 on the PROMIS T score (normed for the general population).
Sedentary behavior
Sedentary behavior was measured via self-report on the five-item inactivity/sedentary behavior subscale of the Pregnancy Physical Activity Questionnaire (PPAQ), (Chasan-Taber et al., Reference Chasan-Taber, Schmidt, Roberts, Hosmer, Markenson and Freedson2004). This PPAQ subscale is a validated and widely used measure for pregnant women (Chasan-Taber et al., Reference Chasan-Taber, Silveira, Pekow, Braun, Manson, Solomon and Markenson2015; Nascimento, Surita, Godoy, Kasawara, & Morais, Reference Nascimento, Surita, Godoy, Kasawara and Morais2015).
Emotional support
The self-report PROMIS-Emotional Support 4a measure (Cella et al., Reference Cella, Riley, Stone, Rothrock, Reeve, Yount and Choi2010) assesses the availability of confidante relationships and feeling cared for and valued as a person. PROMIS-Emotional Support 4a applies item response theory to generate T scores with scores greater than 50 indicating levels of emotional support higher than in the general population.
Covariates
Highest level of maternal education reported during pregnancy was reduced to three categories: less than high school, high school completion, or some college and above. Participants reported receipt of any (yes/no) prenatal public assistance (e.g. State Children's Health Insurance Program, Supplemental Nutrition Assistance Program, Head Start, housing assistance, Medicaid, Supplemental Security Income, Temporary Assistance for Needy Families). Prenatal employment status was coded as working or not working for wages. Pre-pregnancy body mass index (BMI) was self-reported or calculated from measured pre-pregnancy weight and height.
Analytic approach
The study employed a quasi-experimental longitudinal design with women who delivered during or before pandemic onset. Propensity-score matching was used to maximize comparability of the two groups and account for systematic differences in sociodemographic characteristics (i.e. maternal age, maternal race and ethnicity, child sex) based on a 1:1 (pandemic: pre-pandemic) design using the nearest neighbor matching method. A propensity score in the form of probability of belonging to the pandemic or pre-pandemic group conditional on the matching sociodemographic variables was estimated for each case. The pair of cases in the two groups was matched if they had very similar propensity scores (Austin, Reference Austin2011b). Remaining cases with discrepant propensity scores were removed from the data. Consistent with prior propensity score modeling studies (Austin, Reference Austin2011a; Rosenbaum & Rubin, Reference Rosenbaum and Rubin1983), we estimated ‘treatment’ effects (in this case, pandemic exposure) by directly comparing outcomes across matched groups.
We first estimated two multiple regression models with pandemic exposure as the independent variable and infant GA and birthweight for GA z score (BWGA-z score) as the dependent variables (DV). Models included the following covariates: maternal education level, receipt of public assistance, employment status and pre-pregnancy BMI. We then estimated four structural equation models (SEMs) comprising mediator/DVs as follows: (1) perceived stress/GA; (2) perceived stress/BWGA-z score; (3) depressive symptoms/GA; and (4) depressive symptoms/BWGA-z score using semTools in the R software package (Jorgensen, Pornprasertmanit, Schoemann, & Rosseel, Reference Jorgensen, Pornprasertmanit, Schoemann and Rosseel2022). Models were conducted in a stepwise fashion to test for the incremental prediction of the predictor and moderator variables. Step 1 tested direct and indirect effects between pandemic exposure, psychological distress (perceived stress or depressive symptoms), and birth outcomes (GA or BWGA-z score). In Step 2, the main (direct) effects of sedentary behavior (Step 2a) or emotional support (Step 2b) were added to examine the incremental association effect of these factors on prenatal psychological distress and birth outcomes, above and beyond the effect of the pandemic. Finally, in Step 3, the interactions between sedentary behavior × pandemic (Step 3a) or emotional support × pandemic (Step 3b) were added to examine moderation of the direct effects of pandemic exposure on prenatal psychological distress and birth outcomes. In addition, we used the Index of Moderated Mediation (Hayes, Reference Hayes2015) to test whether sedentary behavior and emotional support moderated the indirect associations of pandemic exposure on birth outcomes via prenatal psychological distress. The SEM included the following covariates' direct effects on the mediators: education level, receipt of public assistance and employment status, and the same covariates with the addition of pre-pregnancy BMI on birth outcomes. Rates of covariate missingness varied between 4.2% and 31.4% (mean = 13.5%, see Table 1). To minimize parameter biases associated with listwise deletion, missing data on covariates, mediator and moderator variables were imputed using the mice package in R (Van Buuren & Groothuis-Oudshoorn, Reference Van Buuren and Groothuis-Oudshoorn2011). Multiple imputation (MI) can result in unbiased results with up to 90% missingness with a properly specified MI model that includes all variables related to missingness when data are missing at random (Madley-Dowd, Hughes, Tilling, & Heron, Reference Madley-Dowd, Hughes, Tilling and Heron2019). This resulted in 10 imputed datasets associated with each of the two mediators. The two sets of ten datasets were used to estimate their respective models in lavaan (Rosseel, Reference Rosseel2012) in R and we reported pooled results.
Table 1. Participant characteristics

BMI, body mass index; BWGA, birthweight for gestational age; GA, gestational age; max, maximum; min, minimum; PPAQ, Pregnancy Physical Activity Questionnaire; PROMIS, Patient Reported Outcomes Measurement Information System; s.d., standard deviation.
Note. Cell sizes smaller than 5 are suppressed for privacy in accordance with ECHO's publication and data use policy. Variables with* were covariates used in propensity score matching. Complete data of these variables were available
Groups were compared using t tests for continuous variables. For categorical variables, p values for χ2 tests were computed across categories excluding the missing category between the pre-pandemic and pandemic groups
Results
Descriptive Statistics
Sample characteristics are shown in Table 1. In the overall sample (N = 1002, drawn from 16 ECHO cohorts, see online Supplementary Table S1), participants were on average 30.7 years old (s.d. = 5.03). Most women self-identified as White (75.7%) with 5.8% as Black, 2.4% as Asian, 2.9% as American Indian or Alaska Native, 8.6% more than one race and 4.6% another race, and most participants reported non-Hispanic ethnicity (73.9%). Infants (47.9% female) had an average GA of 38.8 weeks (s.d. = 1.83); 6.3% were born preterm (<37 weeks) and mean birthweight was 3380 g (s.d. = 525). GA was unrelated to BWGA-z score (r = −0.01, ns) indicating their independence for later model estimation. Mean harmonized perceived stress and depression T scores for the overall sample were 47.7 (s.d. = 9.83) and 46.9 (s.d. = 8.61) respectively, close to the population norm. Approximately half of the participants in the exposed group (n = 261, 52%) became pregnant after the start of the pandemic, whereas 97 participants (19.4%) were in the third trimester.
By design, the pandemic and pre-pandemic groups did not differ on maternal age, race, ethnicity and child sex. There was also no group difference on education or income level. However, relative to women in the pre-pandemic group, pandemic-exposed women were more likely to receive public assistance, less likely to be employed and had higher pre-pregnancy BMI (ps < 0.01). These variables were covaried in the predictive and mediation models to account for these group differences.
Effects of pandemic exposure on birth outcomes
Results of the multiple regression models after controlling for covariates showed a small effect of prenatal pandemic exposure on shorter GA at birth [β = −0.56 weeks, 95% CI (−0.89 to −0.24)]. In contrast, pandemic exposure was unrelated to adjusted birthweight [β = 0.01, 95% CI (−0.17 to 0.20)]. Maternal education, receipt of public assistance, and employment status were not significantly associated with birth outcomes.
Mediation models
Pandemic, perceived stress and GA at birth
As shown in Table 2, after adjusting for covariates in Step 1, women who were pregnant during the pandemic reported higher levels of stress compared to those who were pregnant pre-pandemic [B = 2.53, standard error (s.e.) = 0.99, 95% CI (0.59–4.47)]. Furthermore, after adjusting for covariates and perceived stress during pregnancy, pandemic exposure had a small direct effect on GA at birth (B = −0.55, s.e. = 0.18): between 0.20 and 0.90 weeks shorter than pre-pandemic births. However, prenatal stress did not mediate the association between pandemic exposure and infant GA. Higher levels of sedentary behavior were associated with higher levels of perceived stress beyond the significant effects of pandemic status and public assistance (Table 2 GA; Step 2a), but no main effect of sedentary behavior on GA at birth was observed. In Step 2b, higher levels of emotional support were associated with less perceived stress but did not directly predict GA at birth. Neither sedentary behavior nor emotional support moderated the direct and indirect effects of the pandemic on perceived stress and infant GA at birth (Steps 3a and 3b, results not shown).
Table 2. Structural equation models examining perceived stress as mediating the effect of pandemic on birth outcomes

B, unstandardized beta; BMI, body mass index; CI, confidence interval; max, maximum; min, minimum; NA, not applicable; s.e., standard error.
Note: Significant effects are bolded for emphasis.
Pandemic, perceived stress and BWGA
In adjusted models, pandemic exposure showed no direct effect on offspring BWGA-z score (Table 2 BWGA; Step 1). Additionally, there was no main effect of sedentary behavior on adjusted birthweight after accounting for sociodemographic and health covariates, including the significant effects of pre-pregnancy BMI (Table 2 BWGA; Step 2a). Similarly, emotional support did not directly predict BWGA-z score (Step 2b). Neither sedentary behavior nor emotional support moderated direct or indirect effects of the pandemic (Steps 3a and 3b, results not shown).
Pandemic, depressive symptoms and GA at birth
Models examining depressive symptoms as mediating the effect of the pandemic on birth outcomes are shown in Table 3. In Step 1, pandemic-exposed women reported higher levels of prenatal depressive symptoms [B = 3.12, s.e. = 1.07, 95% CI (1.02–5.22)] after adjusting for covariates. In addition, infants delivered during the pandemic had somewhat shorter GA at birth compared to infants delivered pre-pandemic [B = −0.71, s.e. = 0.25, 95% CI (−1.20 to −0.22)]. However, prenatal depressive symptoms did not predict variability in GA, nor did they mediate the association between pandemic exposure and GA at birth. More sedentary behavior was associated with higher levels of prenatal depressive symptoms over and above the significant effects of pandemic status (Table 3 GA; Step 2a). However, no main effect of sedentary behavior on infant GA was observed beyond the significant effect of pandemic status and adjustment for covariates. In Step 2b, emotional support was uniquely associated with lower levels of prenatal depressive symptoms but was unrelated to GA at birth. After adjusting for emotional support, pandemic exposure remained significantly associated with higher prenatal depressive symptoms, although it no longer predicted shorter GA at birth. Neither emotional support nor sedentary behavior moderated the direct and indirect effects of the pandemic on prenatal depressive symptoms and infant GA (Steps 3a and 3b, results not shown).
Table 3. Structural equation models examining depressive symptoms as mediating the effect of the pandemic on birth outcomes
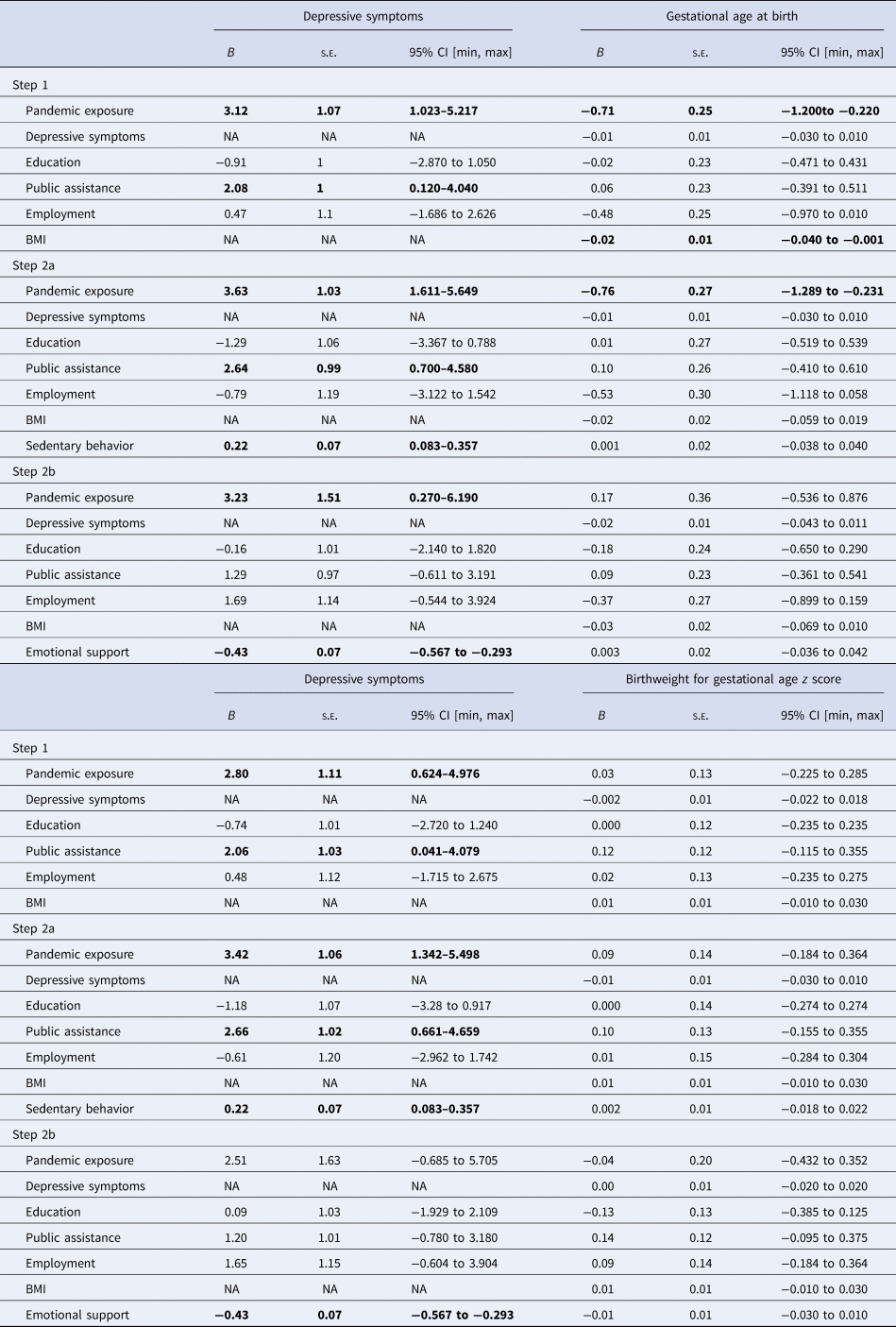
B, unstandardized beta; BMI, body mass index; CI, confidence interval; max, maximum; min, minimum; NA, not applicable; s.e., standard error.
Note: Significant effects are bolded for emphasis.
Pandemic, depressive symptoms and BWGA
In adjusted models, neither pandemic exposure, nor prenatal depressive symptoms, predicted offspring BWGA-z score (Table 3 BWGA; Step 1). In Step 2a, sedentary behavior was associated with higher depressive symptoms beyond the significant effects of pandemic status, whereas in Step 2b, emotional support was associated with fewer depressive symptoms. BWGA-z score was unrelated to sedentary behavior or emotional support in adjusted models. Sedentary behavior and emotional support did not moderate any direct or indirect effects of the pandemic (Steps 3a and 3b).
Discussion
There is an urgent need for rigorously designed studies to examine the impact of the pandemic on women's prenatal health and subsequent birth outcomes, as well as studies that can identify modifiable daily life factors that could exacerbate or attenuate pandemic effects. The ECHO study provides a valuable opportunity to fill these gaps via common data elements collected before and during the pandemic from cohorts located across the United States. The current study used propensity-score matching to increase causal inferences made about the effect of the pandemic on birth outcomes and determine whether heightened psychological distress associated with the pandemic explained these effects.
The results showed that women pregnant during the pandemic reported higher levels of stress and depressive symptoms compared with a propensity-score matched group of women who delivered prior to the pandemic. This increase may reflect the disruptions to daily life and health, social, and financial concerns experienced by many during the pandemic (Fitzpatrick, Drawve, & Harris, Reference Fitzpatrick, Drawve and Harris2020; Tai et al., Reference Tai, Shah, Doubeni, Sia and Wieland2021), and is consistent with prior descriptive studies showing increased prevalence of psychiatric disorders and psychological distress. However, by leveraging a quasi-experimental design, the current study could increase the sociodemographic comparability of the pandemic and pre-pandemic groups to provide a more rigorous test of exposure on prenatal distress. This approach, combined with inclusion of additional covariates, allowed us to delineate the effects of the pandemic from the effects of various sociodemographic confounders.
Contrary to our hypothesis, the study did not reveal a substantial negative effect of pandemic exposure on birth outcomes. Although the results showed a shorter GA in the pandemic relative to the pre-pandemic group, this effect translated to an overall mean difference of about half a week, which may be important for preterm births, but may have little clinical significance for early term and term births. This result is commensurate with several other U.S.-based studies that have shown no, or only a small association, between pandemic exposure and categorical definitions of preterm birth (Greene et al., Reference Greene, Kilpatrick, Wong, Ozimek and Naqvi2020; Handley et al., Reference Handley, Mullin, Elovitz, Gerson, Montoya-Williams, Lorch and Burris2021; Wood et al., Reference Wood, Sinnott, Goldfarb, Clapp, McElrath and Little2021). In addition, the current study revealed no main effects of the pandemic on GA-adjusted infant birthweight, similar to some prior descriptive studies focused on (unadjusted) birthweight (Chmielewska et al., Reference Chmielewska, Barratt, Townsend, Kalafat, van der Meulen, Gurol-Urganci and Thangaratinam2021) but at odds with others conducted outside the United States (Yang et al., Reference Yang, D'souza, Kharrat, Fell, Snelgrove, Murphy and Shah2021). Taken together, our results suggest that pandemic mitigation measures (e.g. focus on hygiene, physical distancing, reduced physical demands of work and travel) while not reducing psychological distress, may have been generally effective in protecting some women' (Goldenberg, Culhane, Iams, & Romero, Reference Goldenberg, Culhane, Iams and Romero2008).
Despite the elevated rates of psychological distress among women pregnant during the pandemic, neither perceived stress nor depressive symptoms predicted birth outcomes beyond the effect of the pandemic. Thus, our hypothesis that psychological distress would mediate the association between prenatal pandemic exposure and negative birth outcomes was not supported. However, this study may only partially capture the range of stress and depression, or birth outcomes experienced by pregnant women in the United States. Specifically, most participants in the analytic sample identified as White (75.7%), non-Hispanic (73.9%), and college educated (81.6%), whereas other racial and ethnic groups and individuals with fewer resources were under-represented. Thus, our observation of minimal effects of the pandemic on birth outcomes may be most relevant to highly educated White women in the United States; an important consideration given that systemic racism and structural processes underlying economic disparities significantly contribute to known inequities in prenatal stress and birth outcomes (Alhusen, Bower, Epstein, & Sharps, Reference Alhusen, Bower, Epstein and Sharps2016; Braveman et al., Reference Braveman, Heck, Egerter, Marchi, Dominguez, Cubbin and Curtis2015; Mendez, Hogan, & Culhane, Reference Mendez, Hogan and Culhane2013). Given the elevated rates of preterm birth among Black, American Indian, and Hispanic/Latinx infants (March of Dimes Foundation, 2022), there is a clear need for additional studies that focus specifically on the impact of the pandemic on prenatal distress and birth outcomes for these groups. Future work should also consider the contributions of psychological resources, given evidence that resilience, optimism and life satisfaction are associated with offspring birth outcomes (Bhatia, Chao, Higgins, Patel, & Crespi, Reference Bhatia, Chao, Higgins, Patel and Crespi2015; Maxson, Edwards, Valentiner, & Miranda, Reference Maxson, Edwards, Valentiner and Miranda2016) and may explain variability beyond prenatal distress (Ramiro-Cortijo et al., Reference Ramiro-Cortijo, De la Calle, Gila-Díaz, Moreno-Jiménez, Martin-Cabrejas, Arribas and Garrosa2021). Thus, it is possible that the current findings masked subgroups differentially characterized by personal resources.
An important strength of the current study was examination of potential pandemic-related effect modifiers (sedentary behavior and emotional support) with relevance for health policy and practice. The results showed a consistent pattern of main effects, whereby sedentary behavior was associated with higher levels of perceived stress and depressive symptoms, and emotional support was robustly associated with lower levels of each. However, none of the hypothesized moderating effects were observed. Although sedentary behavior did not exacerbate the negative effects of the pandemic on distress or birth outcomes in the current analysis, the additive risk to psychological distress highlights a universal need for targeted interventions that reduce sedentary behavior to improve psychological health during pregnancy (DiPietro et al., Reference DiPietro, Evenson, Bloodgood, Sprow, Troiano, Piercy and Powell2019; Kołomańska, Zarawski, & Mazur-Bialy, Reference Kołomańska, Zarawski and Mazur-Bialy2019), regardless of pandemic conditions. Furthermore, despite a lack of association with birth outcomes in the current study, sedentary behavior likely confers risk for maternal cardiovascular diseases such as hypertension, diabetes, and metabolic syndrome (Narici et al., Reference Narici, Vito, Franchi, Paoli, Moro, Marcolin and Biolo2021) that could impact the health of future pregnancies (Xie, Madkour, & Harville, Reference Xie, Madkour and Harville2015).
Emotional support was robustly associated with lower levels of prenatal stress and depressive symptoms and, in most cases, the negative effect of the pandemic on psychological distress became negligible once emotional support was accounted for. These results support the utility of emotional support as a critical target for healthcare efforts in terms of both screening and intervention (Dunkel Schetter, Reference Dunkel Schetter2011; Marques, Bjørke-Monsen, Teixeira, & Silverman, Reference Marques, Bjørke-Monsen, Teixeira and Silverman2015). Emotional support can take many forms such as having a confidante, friends and family in the community, connections with health workers (Hans, Edwards, & Zhang, Reference Hans, Edwards and Zhang2018; Orr, Reference Orr2004), and/or perinatal support groups (Chae, Chae, Kandula, & Winter, Reference Chae, Chae, Kandula and Winter2017; Chan & Chen, Reference Chan and Chen2019). Further research is needed to understand how pregnant women best access/receive emotional support, and the types that are most impactful on psychological well-being during pregnancy.
Limitations
The findings should be considered in the context of several limitations. First, given some constraints on the availability of data, propensity-score matching of the pandemic and pre-pandemic groups was limited to four sociodemographic variables. Although the groups were comparable on educational level and income level, and all women had a singleton pregnancy, descriptive data indicated that some important differences remained on variables including receipt of public assistance, paid employment, and pre-pregnancy BMI. In addition, limited data on parity prevented inclusion of this variable in analyses. Given associations with birth outcomes, including PTB (Koullali et al., Reference Koullali, Van Zijl, Kazemier, Oudijk, Mol, Pajkrt and Ravelli2020), this is an important covariate for future studies. Unmeasured cohort or period effects (e.g. political climate, population health, mental health awareness) could have affected outcomes. Second, data were gathered from a 15-month period during the pandemic (11 March 2020 to 30 May 2021) during which infection rates and mitigation measures varied. While this extended interval fully captured the entire pregnancy for more than half the women unlike some prior studies, there was likely a range in the type, duration, and severity of stress experienced by women (e.g. disruptions to prenatal health care, risk for infection, social isolation, job loss) as well as differences in local and state-level mitigation policies at varying times across the pregnancy that were not modeled. Future studies are needed to examine more fine-grained pandemic experiences in relation to birth outcomes, and to capture the full range of pregnancy experiences and birth outcomes in diverse groups of women. Finally, sample bias may have been introduced by the focus on GA and birthweight among live births included in the ECHO study given some evidence suggesting a higher incidence of stillbirths during the pandemic (Khalil et al., Reference Khalil, Von Dadelszen, Draycott, Ugwumadu, O'Brien and Magee2020).
Conclusion
Using a quasi-experimental design, our results showed that exposure to the COVID-19 pandemic during pregnancy was associated with heightened psychological distress during pregnancy and marginally shorter GA at birth. In addition, we observed a general, but not a pandemic-specific, effect of sedentary behavior and emotional support on prenatal stress and depressive symptoms, highlighting the importance of these factors for maternal health regardless of pandemic exposure.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0033291723000314
Acknowledgments
The authors wish to thank our ECHO colleagues; the medical, nursing and program staff, and the children and families participating in the ECHO cohorts. We also acknowledge the contribution of the following ECHO program collaborators:
ECHO Components – Coordinating Center: Duke Clinical Research Institute, Durham, North Carolina: Smith PB, Newby KL.
Data Analysis Center: Johns Hopkins University Bloomberg School of Public Health, Baltimore, Maryland: Jacobson LP: Research Triangle Institute, Durham, North Carolina: Parker CB.
Person-Reported Outcomes Core: Northwestern University, Evanston, Illinois: Gershon R, Cella D.
Pediatric Cohorts: Northeastern University, Boston, Massachusetts: Alshawabkeh AN, University of Southern California, Los Angeles: Breton CV, Bastain T, Farzan S, Habre R, Memorial Hospital of Rhode Island, Pawtucket: Deoni S, D'Sa VA, University of Colorado Denver, Denver, CO: Dabelea D, New York State Psychiatric Institute at Columbia University, New York: Duarte CS, Canino GJ, Monk CE, Posner JE, Emory University, Atlanta, Georgia: Dunlop AL, Brennan PA, Corwin EJ, Avera McKennan Hospital and University Health Center, Sioux Falls, South Dakota: Elliott AJ, Kaiser Permanente, Oakland, California: Ferrara A, Croen LA, University of Wisconsin- Madison: Gern J, Columbia University Medical Center, New York, NY: Herbstman J, University of Pittsburgh, Pennsylvania: Hipwell AE, Keenan KE, Dartmouth College, Hanover, New Hampshire: Karagas MR, University of Rochester, New York: O'Connor TG, Buss C, Miller RK, Simhan H, Wadhwa PD, Michigan State University, East Lansing, Michigan: Paneth N, Kerver JM, Ruden DM, University of Illinois, Urbana: Schantz SL, and Icahn School of Medicine at Mount Sinai, New York, New York: Wright RJ, Wright RO.
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Financial support
Research reported in this publication was supported by the Environmental influences on Child Health Outcomes (ECHO) program, Office of the Director, National Institutes of Health, under Award Numbers U2COD023375 (Coordinating Center), U24OD023382 (Data Analysis Center), U24OD023319 (PRO Core) and UH3OD023251, UH3OD023287, UH3OD023313, UH3OD023248, UH3OD023328, UH3OD023318, UH3OD023279, UH3OD023289, UH3OD023282, UH3OD023290, UH3OD023244, UH3OD023275, UH3OD023349, UH3OD023285, UH3OD023272 and UH3OD023337 (Pediatric Cohorts).
Conflict of interest
None.
Ethical standards
The study protocol was approved by the local [or single ECHO] institutional review board. Written informed consent or parent's/guardian's permission was obtained for ECHO-wide Cohort Data Collection Protocol participation and for participation in specific cohorts.









