Article contents
Sapozhnikovite, Na8(Al6Si6O24)(HS)2, a new sodalite-group mineral from the Lovozero alkaline massif, Kola Peninsula
Published online by Cambridge University Press: 10 December 2021
Abstract
The new sodalite-group mineral sapozhnikovite, ideally Na8(Al6Si6O24)(HS)2, was discovered in a hydrothermally altered urtite-like rock at Karnasurt Mountain, Lovozero alkaline massif, Kola Peninsula, Russia. The associated minerals are nepheline, aegirine, potassic feldspar, albite, kyanoxalite, natrolite, fluorapatite, fluorcaphite, lomonosovite (partially or completely altered to murmanite) and loparite-(Ce). Sapozhnikovite forms isolated colourless to pale greyish anhedral equant grains up to 5 mm across. The streak is white and the lustre is vitreous. Strong orange fluorescence under longwave UV radiation (λ = 330 nm) and weak yellow-orange fluorescence under shortwave UV radiation (λ = 245 nm) is observed. Sapozhnikovite is brittle, with a Mohs hardness of 5½. Cleavage is imperfect on (110). Density measured by flotation in heavy liquids is equal to 2.25(1) g⋅cm–3. The calculated density is 2.255 g⋅cm–3. Sapozhnikovite is characterised by infrared, Raman, electron spin resonance, NIR-Vis-UV absorption, and photoluminescence spectroscopy. The chemical composition is (wt.%, electron microprobe, H2O determined by gas chromatography of ignition products): Na2O 25.05, Al2O3 32.44, SiO2 37.58, HS 4.33, Cl 2.22, H2O 0.30, –O≡(Cl,HS) –1.55, total 100.37. The empirical formula is Na7.73Al6.08Si5.97O24(HS)1.25Cl0.60⋅0.16H2O. The crystal structure was determined using single-crystal X-ray diffraction data and refined to R1 = 1.62%. Sapozhnikovite is cubic, P$\bar{4}$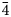 3n, with a = 8.9146(1) Å, V = 708.45(2) Å3 and Z = 1. The new mineral is isostructural with sodalite. The strongest lines of the powder X-ray diffraction pattern [d, Å (I, %) (hkl)] are: 6.30 (37) (110), 3.638 (100) (211), 2.821 (14) (310), 2.572 (18) (222), 2.382 (16) (321) and 2.101 (29) (411). The mineral is named in honour of the Russian mineralogist and crystallographer Dr. Anatoly Nikolaevich Sapozhnikov (b. 1946).
3n, with a = 8.9146(1) Å, V = 708.45(2) Å3 and Z = 1. The new mineral is isostructural with sodalite. The strongest lines of the powder X-ray diffraction pattern [d, Å (I, %) (hkl)] are: 6.30 (37) (110), 3.638 (100) (211), 2.821 (14) (310), 2.572 (18) (222), 2.382 (16) (321) and 2.101 (29) (411). The mineral is named in honour of the Russian mineralogist and crystallographer Dr. Anatoly Nikolaevich Sapozhnikov (b. 1946).
Keywords
- Type
- Article
- Information
- Copyright
- Copyright © Institute of Problems of Chemical Physics, Russian Academy of Sciences, 2021. Published by Cambridge University Press on behalf of the Mineralogical Society of Great Britain and Ireland
Footnotes
Associate Editor: Charles A Geiger
References
- 16
- Cited by




