Mineralogical Magazine and Journal of the Mineralogical Society, Volume 35 - Issue 269 - March 1965
- This volume was published under a former title. See this journal's title history.
Other
Bye-Laws and List of Members1
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. ix-xlviii
-
- Article
- Export citation
Meeting Report
Proceedings of the Mineralogical Society (1964)
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. xlix-lxiv
-
- Article
- Export citation
Research Article
Gyrolite and reyerite from ’S Airde Beinn, northern Mull
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 1-4
-
- Article
- Export citation
The thermal decomposition of crocidolite from Koegas, South Africa
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 5-30
-
- Article
- Export citation
The thermal disintegration of crocidolite in air and in vacuum
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 31-37
-
- Article
- Export citation
A study of uvarovite
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 38-45
-
- Article
- Export citation
A scheme of cation distribution in the amphiboles
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 46-54
-
- Article
- Export citation
Alteration products of olivine and pyroxene in basalt lavas from the Isle of Mull1
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 55-68
-
- Article
- Export citation
Rhombohedral molybdenite from the Minas da Panasqueira, Beira Baixa, Portugal
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 69-71
-
- Article
- Export citation
Xanthiosite and aerugite
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 72-83
-
- Article
- Export citation
Mineralogy of certain pyrite concretions from Israel and their alteration products
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 84-87
-
- Article
- Export citation
Aspects of the geochemistry of arsenic and antimony, exemplified by the Skaergaard intrusion
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 88-107
-
- Article
- Export citation
The identification, mode of occurrence, and quantitative determination of crystalline phases in granulated blast-furnace slag
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 108-117
-
- Article
- Export citation
Intergrowths in ilmenite of the beach sands of Kerala
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 118-130
-
- Article
- Export citation
Oriented transformation of manganite during heat treatment
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 131-139
-
- Article
- Export citation
A chlorite–corundum roclc from Mount Painter, South Australia
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 140-145
-
- Article
- Export citation
Calciostrontianite in the basal Purbeck beds of Durlston Head, Dorset
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 146-150
-
- Article
- Export citation
An occurrence of phlogopite and its transformation to vermiculite by weathering
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 151-164
-
- Article
- Export citation
An electron-microscope study of peristerite plagioclases
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 165-176
-
- Article
- Export citation
Enhanced production of magnesium silicates from strained magnesia1
-
- Published online by Cambridge University Press:
- 14 March 2018, pp. 177-188
-
- Article
- Export citation

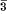
 of Mn
of Mn . A discussion is given of the part played by these lamellae when optical schiller is exhibited; and the effect of heat treatment on the lamellar structure and optical schiller is described.
. A discussion is given of the part played by these lamellae when optical schiller is exhibited; and the effect of heat treatment on the lamellar structure and optical schiller is described.