Spectrin was first identified in red blood cells, where it functions as a scaffolding that is flexible and extensible, allowing the cells to deform in order to pass through tiny capillaries and then rebound to their original shape. It has since been determined that this cytoskeletal protein lines the intracellular surface of the plasma membrane of other eukaryotic cells including the hair cells of the inner ear. What were originally viewed as “hairs” are now known to be stereocilia. These specialized organelles are attached to the cuticular plate of what are still called hair cells. Nanoscale movements of these cilia in the inner ear are a critical step in converting physical movements into neural impulses that are interpreted by the brain as hearing and balance. The cochlea of the inner ear mediates hearing whereas the vestibular apparatus in the inner ear functions for balance.
The cuticular plate of hair cells in the cochlea is thought to be critical in hearing in mammals by securing stereocilia in place and providing them with the rigidity and support necessary for converting the mechanical energy of sound vibrations into electrical signals. The rootlets of stereocilia are electron-dense structures penetrating into the cuticular plate and forming an anchoring complex. This anchoring complex is a key structural component for stereocilia to withstand constant mechanical stresses and therefore plays a critical role in hearing. However, until now, the specific molecules that anchor the stereocilia and provide the necessary elasticity and flexibility have been elusive. In an elegant study published by Yan Liu, Jieyu Qi, Xin Chen, Mingliang Tang, Shuijin He, Renjie Chai, Guisheng Zhong and others, spectrin was identified as a critical element of the anchoring complex.
To determine the extent to which spectrin structures underpin the function of stereocilia, Liu et al. used super-resolution fluorescene imaging, which can provide molecular labeling specificity and live-cell imaging with nanoscale resolution. Experiments using immunostaining investigated the structure of both αII- and βII-spectrin in the cuticular plates of hair cells in mice. Using stimulated emission depletion imaging (one form of super-resolution microscopy) and transmission electron microscopy they obtained evidence that suggested spectrin forms cylinders that wrap around rootlets, functioning as a mechanical sensor during deflection of stereocilia. The intrinsic extensibility and elasticity of spectrin would provide this functional capability. These and other results suggested that αII- and βII-spectrin form flexible tetramers outlining the edges of the stereocilia rootlets of hair cells in the cuticular plate.
Since hair cells in the vestibule are not subject to the same mechanical stresses as those in the cochlea, Liu et al. compared the distribution of spectrin in the cuticular plate of hair cells in both the cochlea and the vestibule. Interestingly, spectrin was distributed in a meshwork arrangement in the vestibular hair cells, but no rings were observed.
Knockout mice were created that lacked βII-spectrin and, as expected, this molecule was completely deleted from the cuticular plate of these animals. αII-spectrin could be detected elsewhere in these animals but was nearly deleted from the cuticular plates, suggesting that βII-spectrin may be required for the specific location of αII-spectrin in the cuticlar plate of hair cells. Additional results suggested that βII-spectrin is important for arranging three other proteins known to be normally associated with the base of stereocilia: F-actin (Figure 1), taperin, and FAM65B.
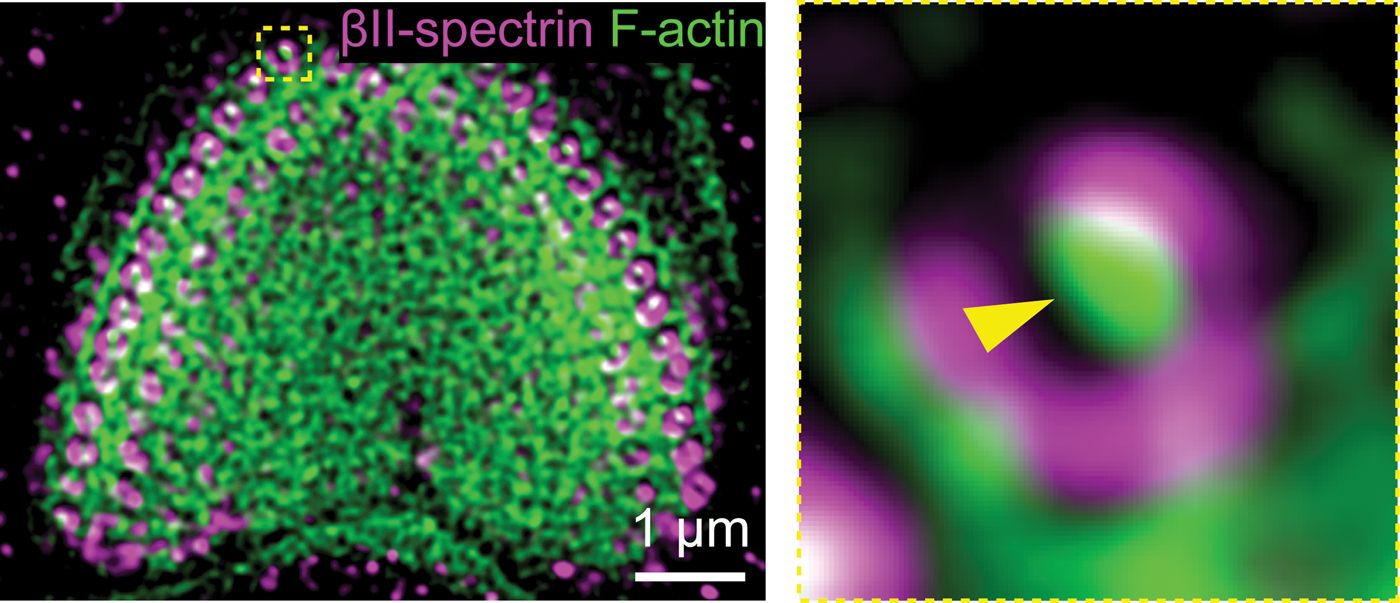
Figure 1: Representative two-color stimulated emission depletion images of βII-spectrin (magenta) and F-actin (cyan) in the cuticular plate of a hair cell. The yellow box at the top of the image on the left indicates the area of the image on the right.
Hearing acuity can be determined in animals by the auditory brainstem response (ABR) where electrical signals are detected by electrodes placed near the ear. Mice displayed a severe hearing loss when βII-spectrin was knocked out. Similarly in aging mice and mice that were subjected to a loud noise known to damage hearing, the ABR was diminished. Imaging studies demonstrated a marked decrease in both forms of spectrin in the cuticular plate of the hair cells. Also in these studies functional tests for balance found no significant loss of vestibular function. These results demonstrate a crucial role of spectrin in hair cell function for hearing, but not for balance. Further studies showed that spectrin also plays a critical role in the development of hearing in young mice. The study of Liu et al. used multiple approaches to provide a structural basis for understanding the function of spectrin in the auditory system.






