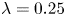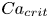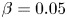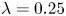496 results
A variationist study of /tʊt/ in Michif French
-
- Journal:
- Journal of French Language Studies , First View
- Published online by Cambridge University Press:
- 30 April 2024, pp. 1-23
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Late Silurian event stratigraphy and facies of South Wales and the Welsh Borderland, United Kingdom
-
- Journal:
- Geological Magazine / Volume 160 / Issue 11 / November 2023
- Published online by Cambridge University Press:
- 29 April 2024, pp. 2010-2055
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Dynamics and active mixing of a droplet in a Stokes trap
-
- Journal:
- Journal of Fluid Mechanics / Volume 985 / 25 April 2024
- Published online by Cambridge University Press:
- 22 April 2024, A15
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Chapter 13 - Activity and Positive Engagement within a PICU
-
-
- Book:
- Psychiatric Intensive Care
- Published online:
- 15 March 2024
- Print publication:
- 28 March 2024, pp 159-177
-
- Chapter
- Export citation
Rethinking Transnational Activism through Regional Perspectives: Reflections, Literatures and Cases
-
- Journal:
- Transactions of the Royal Historical Society , First View
- Published online by Cambridge University Press:
- 08 January 2024, pp. 1-27
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Glycerol-Intercalated Mg-Al Hydrotalcite as a Potential Solid Base Catalyst for Transesterification
-
- Journal:
- Clays and Clay Minerals / Volume 58 / Issue 4 / August 2010
- Published online by Cambridge University Press:
- 01 January 2024, pp. 475-485
-
- Article
- Export citation
Developing a framework to improve global estimates of conservation area coverage
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
VERTICO V: The environmentally driven evolution of the inner cold gas discs of Virgo cluster galaxies
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 40 / 2023
- Published online by Cambridge University Press:
- 27 April 2023, e017
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Lumateperone 42 mg in an Open-Label Switch Study in Patients with Stable Schizophrenia: Results by Previous Antipsychotic
-
- Journal:
- CNS Spectrums / Volume 28 / Issue 2 / April 2023
- Published online by Cambridge University Press:
- 14 April 2023, pp. 235-236
-
- Article
-
- You have access
- Export citation
Lumateperone in Pooled Late-Phase Schizophrenia Trials: Number Needed to Treat, Number Needed to Harm, and Likelihood to Be Helped or Harmed
-
- Journal:
- CNS Spectrums / Volume 28 / Issue 2 / April 2023
- Published online by Cambridge University Press:
- 14 April 2023, p. 243
-
- Article
-
- You have access
- Export citation
The Memphis Pandemic Health Informatics System (MEMPHI-SYS)—Creating a Metropolitan COVID-19 Data Registry Linked Directly to Community Testing to Enhance Population Health Surveillance
-
- Journal:
- Disaster Medicine and Public Health Preparedness / Volume 17 / 2023
- Published online by Cambridge University Press:
- 12 December 2022, e326
-
- Article
- Export citation
Flow of a concentrated emulsion with surfactant through a periodic porous medium
-
- Journal:
- Journal of Fluid Mechanics / Volume 953 / 25 December 2022
- Published online by Cambridge University Press:
- 07 December 2022, A21
-
- Article
- Export citation
Particle capture by expanding droplets: effects of inner diffusion
-
- Journal:
- Journal of Fluid Mechanics / Volume 948 / 10 October 2022
- Published online by Cambridge University Press:
- 13 September 2022, A36
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Conducting a pediatric randomized clinical trial during a pandemic: A shift to virtual procedures
- Part of
-
- Journal:
- Journal of Clinical and Translational Science / Volume 6 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 25 August 2022, e115
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Objective measures of reward sensitivity and motivation in people with high v. low anhedonia
-
- Journal:
- Psychological Medicine / Volume 53 / Issue 10 / July 2023
- Published online by Cambridge University Press:
- 12 May 2022, pp. 4324-4332
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
16 - Managing Risks to Improve the Resilience of Arable Farming in the East of England
-
-
- Book:
- Resilient and Sustainable Farming Systems in Europe
- Published online:
- 21 April 2022
- Print publication:
- 05 May 2022, pp 263-278
-
- Chapter
-
- You have access
- Open access
- HTML
- Export citation
2 - The Importance of Improving and Enlarging the Scope of Risk Management to Enhance Resilience in European Agriculture
-
-
- Book:
- Resilient and Sustainable Farming Systems in Europe
- Published online:
- 21 April 2022
- Print publication:
- 05 May 2022, pp 18-37
-
- Chapter
-
- You have access
- Open access
- HTML
- Export citation
The Use of Portable Oxygen Concentrators in Low-Resource Settings: A Systematic Review
- Part of
-
- Journal:
- Prehospital and Disaster Medicine / Volume 37 / Issue 2 / April 2022
- Published online by Cambridge University Press:
- 02 March 2022, pp. 247-254
- Print publication:
- April 2022
-
- Article
- Export citation
The long-term sustainability of a respiratory culture nudge
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 31 January 2022, e15
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
The Impact of the Tips from Former Smokers® Campaign on Reducing Cigarette Smoking Relapse
-
- Journal:
- Journal of Smoking Cessation / Volume 2022 / 2022
- Published online by Cambridge University Press:
- 01 January 2024, e3
- Print publication:
- 2022
-
- Article
-
- You have access
- Open access
- HTML
- Export citation