The Institute of Medicine estimates that 54 % of non-Hispanic Blacks have very low levels of vitamin D (25-hydroxycholecalifierol (25(OH)D3) <16 ng/ml), v. 27 % of Mexican Americans and 11 % of non-Hispanic Whites(1, Reference Looker, Dawson-Hughes and Calvo2). A growing literature(1–Reference Hossein-Nezhad and Holick3) suggests that vitamin D may be associated with reduced mortality (overall(Reference Zittermann, Iodice and Pilz4) and hospital-associated(Reference Lange, Litonjua and Gibbons5)), incidence of cancer(Reference Toner, Davis and Milner6–Reference Robsahm, Schwartz and Tretli13), CVD(Reference Tomson, Emberson and Hill14, Reference Wang, Song and Manson15), diabetes(Reference Song, Wang and Pittas16), fractures(Reference Bischoff-Ferrari, Willett and Wong17, Reference Bischoff-Ferrari, Dawson-Hughes and Staehelin18) and ageing-related functional limitations(Reference Sohl, van Schoor and de Jongh19), although some studies find an unclear relationship(Reference Kupferschmidt20, Reference Murdoch, Slow and Chambers21) or a harmful association with disease(22). Randomized controlled trials are underway to examine the potential role of vitamin D in the prevention of multiple diseases, including cancer, CVD and diabetes(Reference Kupferschmidt20, Reference Lappe and Heaney23).
There are known racial and seasonal variations in vitamin D. Black individuals synthesize less vitamin D per unit of sun exposure than white individuals, because darker complexion acts as a natural sunscreen(Reference Clemens, Adams and Hendeson24, Reference Ginde, Liu and Camargo25). The majority of vitamin D is estimated to derive from sunshine for most people(Reference Holick26), with the liver converting cholecalciferol into 25(OH)D3, the form of vitamin D typically measured in blood serum. 25(OH)D3 has a reported half-life of 15 d(Reference Jones27), and therefore decreases from summer (when solar radiation is highest) to winter(Reference Hyppönen and Power28). Observational studies suggest higher cancer mortality rates for patients diagnosed in winter(Reference Robsahm, Tretli and Dahlback29, Reference Lagunova, Porojnicu and Dahlback30), at northern latitudes(Reference Hanchette and Schwartz31, Reference Schwartz and Hanchette32) and in areas with less UV radiation from sunshine(Reference Grant33, Reference Grant and Garland34), although genetics may contribute(Reference Grant35). Additionally, previous research suggests that racial differences in the synthesis of vitamin D from sunshine are largest in January(Reference Taksler, Cutler and Giovannucci36). However, studies have not adequately considered the association between the UV index and racial differences in vitamin D deficiency or insufficiency.
We assessed the extent of vitamin D insufficiency by race/ethnicity and geography (as categorized by the UV index) in January, when low UV radiation limited the body’s ability to synthesize vitamin D from sunshine.
Experimental methods
Data and participants
We obtained data on serum vitamin D and race/ethnicity from the National Health and Nutrition Examination Survey (NHANES)(37) for 14 319 individuals aged ≥18 years from 2000 to 2006. Individuals had known serum 25-hydroxyvitamin D (25(OH)D), race (non-Hispanic White, Hispanic, non-Hispanic Black), age, gender, marital status, household income, smoking status and BMI. We also obtained restricted data on each participant’s census tract of residence, month and year of NHANES examination and testing, and serum 25(OH)D levels for 2000 (vitamin D data were publicly available for later years).
For UV irradiance, we used satellite data from the National Aeronautics and Space Administration in increments of 1° latitude and 1·25° longitude, throughout the USA, collected from 1978 to 2005(38, Reference Boscoe and Schymura39). These data covered the UV-B and UV-A spectrum (280–400 nm). We obtained the average noontime UV index (‘UV index’) for each calendar month, for the centroid of each NHANES respondent’s census tract(40, Reference Herman and Celarier41). The UV index measures UV radiation reaching the Earth’s surface; in the continental USA, it ranges from 0–5 in January to 4–12 in July(40). The index is a function of latitude, time, solar irradiation, elevation, ozone and cloud cover(40, Reference Herman and Celarier41). Although we used the long-term average UV index, the correlation of the long-term average and the 2000–2005 average was ≥99·0 % for each calendar month.
Control variables
Using NHANES data, we obtained each participant’s age, gender, marital status, annual household income, hair colour, skin reaction to half an hour of sun exposure without sunscreen or protective clothing, how often an individual typically stayed in the shade on a sunny day when outside for >1 h, number of sunburns in the past year, how often an individual wore a hat that covered the face, ears and neck, how often an individual wore a long-sleeved shirt, how often an individual wore sunscreen, the sun protection factor (SPF) most often used, average daily vitamin D supplementation during the past 30 d, BMI, smoking status, and month and year of examination. Table 1 provides categorizations of each variable.
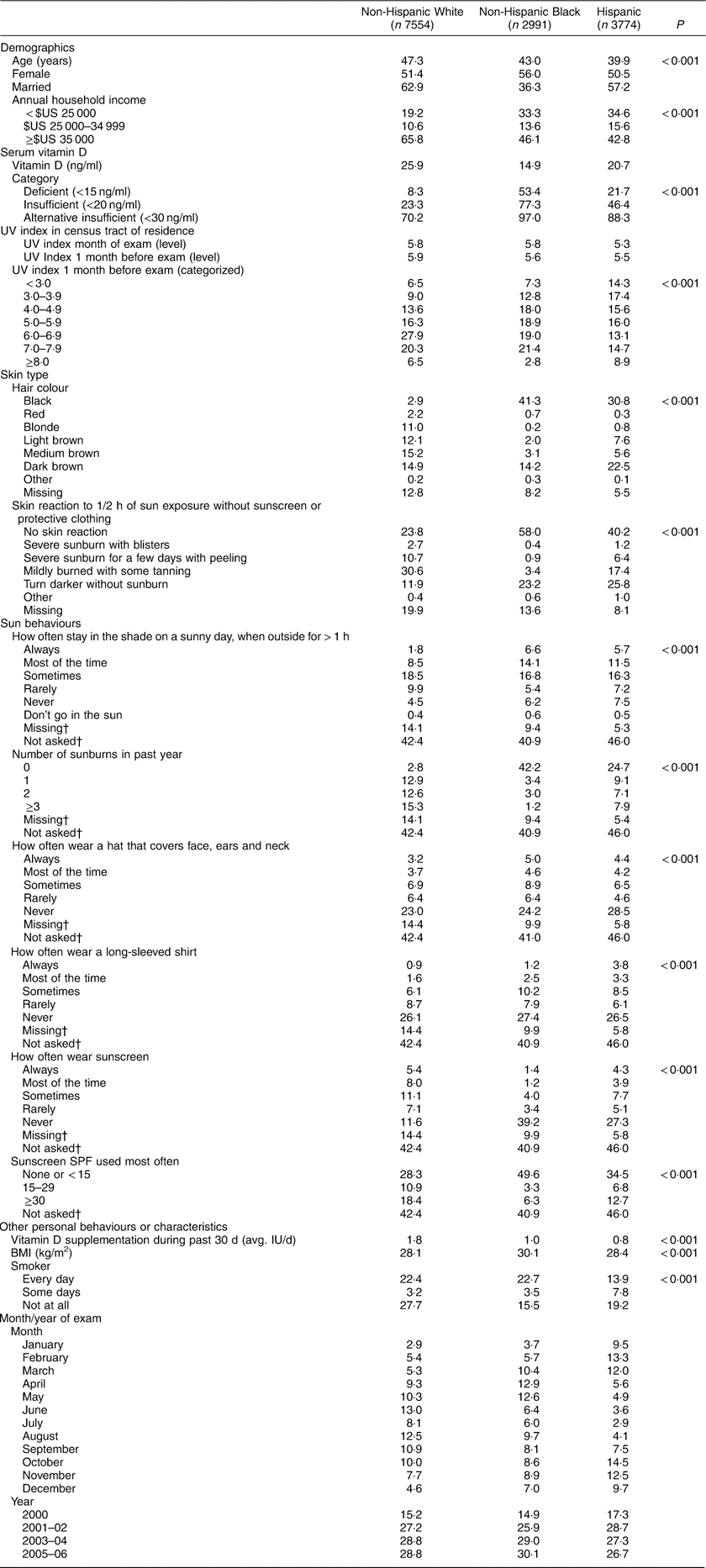
Table 1 Summary statistics of the study population; adults aged ≥18 years (n 14 319), National Health and Nutrition Examination Survey (NHANES), 2000–2006

SPF, sun protection factor.
Data are presented as percentages, unless otherwise specified.
† Question not asked in 2000 and 2001–02. Percentage missing is shown for 2003–04 and 2005–06.
Analyses
To better understand why vitamin D might vary across races and ethnicities, we performed three analyses. First, we estimated the number of minutes outdoors at noon in each calendar month for fair-skinned white, Hispanic and dark-skinned black individuals to synthesize 15 µg (600 IU) of vitamin D, the RDA for adults aged ≤70 years(1). This calculation was defined as the minimum erythemal dose (MED) of UV radiation required for individuals of each race to synthesize 15 µg (600 IU) of vitamin D, divided by the MED supplied per minute of noontime outdoor exposure. For the numerator, we followed dermatology literature to expect that fair-skinned white, Hispanic and dark-skinned black individuals (Fitzpatrick skin types I, IV and VI, respectively, where I is the lightest and VI is the darkest skin tone(Reference Fitzpatrick42)) would require 0·25, 0·56 and 1·25 MED of UV radiation, respectively, to synthesize 15 µg (600 IU) of vitamin D, with 25·5 % of body surface area – the head, neck, arms and hands – uncovered (Appendix 1)(Reference Terushkin, Bender and Psaty43). Again following dermatology literature(Reference Terushkin, Bender and Psaty43), we assumed linearity of this relationship, so that half the MED exposure or uncovered body surface area would yield half the vitamin D absorption. For the denominator, we used the definition of the UV index (1 unit=0·0072 MED/min)(Reference Kiedron, Stierle and Lantz44). We also assumed a relationship between average maximum daily temperature and body parts exposed to sunlight (≥23·9°C (≥75°F): face, neck, hands, arms exposed; 15·6–23·8°C (60–74°F): face, neck, hands, lower arms exposed; –1·1 to 15·5°C (30–59°F): face, neck, hands exposed; –9·4 to –1·2°C (15–29°F): face, neck exposed; ≤–9·5°C (≤14°F): face exposed)(Reference Taksler, Cutler and Giovannucci36, 45, Reference Lund and Broder46). We then estimated the difference in minutes required to synthesize 15 µg (600 IU) of vitamin D, between each minority population (Hispanics, dark-skinned blacks) and fair-skinned whites. To better illustrate our methods, consider Atlanta, Georgia in January. Atlanta had an average UV index of 2·54(38) and maximum temperature of 11·1°C (52°F)(45). We estimated that fair-skinned whites would synthesize 15 µg (600 IU) vitamin D in 31 min (0·25 MED required to synthesize 15 µg of vitamin D ÷ (0·0072 MED/UV index unit×2·54 UV index units)×(25·5 % exposed body surface area in baseline formula ÷ 11·5 % of exposed body surface area at 11·1°C))(Reference Taksler, Cutler and Giovannucci36, 38, 40, Reference Herman and Celarier41, Reference Terushkin, Bender and Psaty43). Similarly, we estimated that Hispanics and dark-skinned blacks would synthesize 15 µg (600 IU) vitamin D in 69 and 153 min, respectively(Reference Taksler, Cutler and Giovannucci36, 38, Reference Holick49, Reference Herman and Celarier41, Reference Terushkin, Bender and Psaty43). Therefore, estimated racial/ethnic differences in the number of minutes to synthesize 15 µg (600 IU) of vitamin D were 41 min for Hispanics and 126 min for dark-skinned blacks, each as compared with fair-skinned whites.
Second, we used multiple regression models to assess the relationship between serum vitamin D and the UV index. The dependent variable was vitamin D (ng/ml) at the time of NHANES examination and the main independent variables of interest were each participant’s race/ethnicity and the average UV index 1 month prior to the exam. We selected 1 month prior to the exam because vitamin D has an estimated half-life of 15 d(Reference Jones27), and the former was more highly correlated with vitamin D than the average UV index in the month of the exam or 2–3 months prior to the exam. Control variables were as described above. Observations were weighted using NHANES-provided weights for its medical examination sample, and robust standard errors were used. In sensitivity analyses, we considered categorizations of the UV index in groups (<3·0, 3·0–3·9, 4·0–4·9, 5·0–5·9, 6·0–6·9, 7·0–7·9, ≥8·0). We also considered an interaction term between the UV index and overweight/obese BMI (25·0–25·9 kg/m2, ≥30·0 kg/m2), because studies suggest that vitamin D may be stored in adipose tissue(Reference Holick47), with levels inversely related to obesity(Reference Holick47, Reference McCullough, Weinstein and Freedman48).
Third, we estimated the population-level distribution of vitamin D levels by race and ethnicity in January, the month with the lowest average UV index(45). To do so, we adjusted vitamin D levels for each NHANES participant to January by performing the regression of vitamin D v. race/ethnicity and eleven monthly dummies (February–December, relative to January). We then subtracted the coefficient for the month of each participant’s NHANES laboratory examination from his/her actual vitamin D level, to estimate January-adjusted vitamin D. For each race/ethnicity, we obtained the 2nd, 5th–95th (in 5 percentage point increments) and 98th percentiles of adjusted vitamin D levels, and bootstrapped to obtain intermediate points in the distribution. (NHANES Restricted Data Center privacy regulations prohibited us from recording the entire distribution.) We performed a similar analysis stratified by race/ethnicity and quartile of the January UV index. We then examined the proportion of each group estimated to be deficient (<15 ng/ml)(1) or insufficient (<20 ng/ml according to the Institute of Medicine-recommended thresholds(1), or <30 ng/ml according to alternative criteria for insufficiency suggested by some researchers(Reference Holick49)) in vitamin D in January.
Results
Table 1 presents summary statistics. Non-Hispanic Whites were older, wealthier and had more sunburns than Hispanics and non-Hispanic Blacks. Non-Hispanic Whites had the highest average vitamin D levels (25·87 ng/ml), followed by Hispanics (20·72 ng/ml) and non-Hispanic Blacks (14·93 ng/ml).
Figure 1 shows racial/ethnic differences in the estimated number of minutes required to synthesize 15 µg (600 IU) of vitamin D from sunshine in January, using the relationship between temperature and sun exposure described in the Methods. Hispanics required 23, 78 and 110 min more than fair-skinned whites in the counties containing Los Angeles, New York City and Chicago, respectively, based on UV index and expected body area exposed to sunlight. Dark-skinned blacks required ≥120 min more than fair-skinned whites in the vast majority of the USA. For comparison, in July, the racial difference was typically <15 min between Hispanics and fair-skinned whites, and <30 min between dark-skinned blacks and fair-skinned whites (not shown).

Fig. 1 (colour online) Racial/ethnic differences in estimated number of minutes required to synthesize 15 µg (600 IU) of vitamin D from sunlight, in January. Results shown for noontime, assuming no sunscreen and a relationship between average maximum daily temperature and sun exposure (≥23·9°C (≥75°F): face, neck, hands, arms exposed; 15·6–23·8°C (60–74°F): face, neck, hands, lower arms exposed; –1·1 to 15·5°C (30–59°F): face, neck, hands exposed; –9·4 to –1·2°C (15–29°F): face, neck exposed; ≤–9·5°C (≤14°F): face exposed) for: (a) black (darkest skin) minus white (fairest skin); (b) Hispanic minus white (fairest skin)
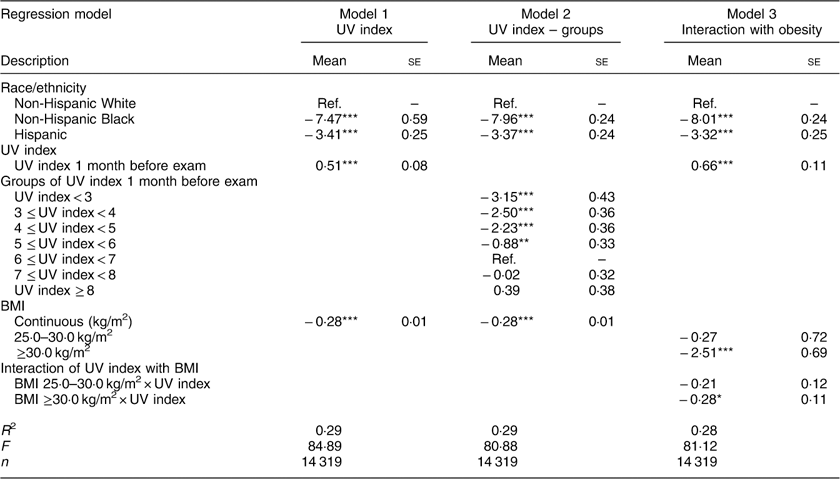
Table 2 shows regression results for key variables of interest. (See Appendix 2 for complete results.) A 1-point increase in the UV index 1 month prior to the examination was associated with a 0·51 ng/ml increase in 25(OH)D levels (95 % CI 0·35, 0·67 ng/ml, P<0·001; model 1). Non-Hispanic Black race and Hispanic ethnicity were associated with 25(OH)D levels lower by 7·47 ng/ml (95 % CI −8·63, −6·31 ng/ml) and 3·41 ng/ml (95 % CI −3·89, −2·93 ng/ml), respectively (both P<0·001; model 1). This association was most evident when the UV index was <5·0 (model 2).
Table 2 Regression results. Dependent variable: serum 25-hydroxyvitamin D (ng/ml)

Ref., reference category.
Robust standard errors were used.
Coefficients for demographics, sun behaviours, other personal characteristics and month/year of exam are shown in Appendix 2.
*P<0·05, **P<0·01, ***P<0·001.
Obesity, defined as a BMI ≥30·0 kg/m2, was associated with a 2·51 ng/ml decrease in vitamin D (95 % CI −3·86, −1·17 ng/ml, P<0·001; model 3). Additionally, obese individuals had a 0·28 ng/ml attenuated association of vitamin D with the UV index (95 % CI −0·50, −0·05 ng/ml, P=0·015). For example, controlling for the interaction of BMI group with vitamin D (model 3), a 1-point increase in the UV index was associated with an average 0·66 ng/ml increase in vitamin D. For an obese individual, this association was attenuated to a 0·38 ng/ml increase in vitamin D (0·66 ng/ml average minus 0·28 ng/ml interaction term).
Figure 2 shows the estimated population-level distribution of serum vitamin D in January, by race/ethnicity (across all geographic locations). Mean January-adjusted vitamin D levels were 23·75 (sd 8·30) ng/ml, 19·29 (sd 6·94) ng/ml and 13·21 (sd 6·49) ng/ml for non-Hispanic Whites, Hispanics and non-Hispanic Blacks, respectively. Vertical lines in Fig. 2 indicate thresholds for vitamin D deficiency (<15 ng/ml)(1) and insufficiency (<20 ng/ml or <30 ng/ml, depending on the definition used)(1, Reference Holick49). In January, an estimated 65·4 % of non-Hispanic Blacks were deficient in vitamin D, compared with 28·9 % of Hispanics and 14·0 % of non-Hispanic Whites. An estimated 84·2 % of non-Hispanic Blacks were insufficient (<20 ng/ml) in vitamin D v. 56·3 % of Hispanics and 34·8 % of non-Hispanic Whites. Using alternative criteria for insufficiency (<30 ng/ml), all non-Hispanic Blacks were insufficient in vitamin D, compared with 91·9 % of Hispanics and 78·2 % of non-Hispanic Whites.

Fig. 2 (colour online) Estimates of serum 25-hydroxyvitamin D, adjusted to January, by race/ethnicity; adults aged ≥18 years (n 14 319), National Health and Nutrition Examination Survey (NHANES), 2000–2006. Vertical lines represent thresholds for vitamin D deficiency (<15 ng/ml) and insufficiency (<20 ng/ml or <30 ng/ml, depending on the source)
Figure 3 repeats the analysis by race/ethnicity and geographic location (as categorized by approximate quartile of the January UV index in each participant’s census tract of residence). Mean January-adjusted vitamin D levels ranged from 19·79 to 24·38 ng/ml for non-Hispanic Whites, from 16·74 to 19·90 ng/ml for Hispanics and from 10·62 to 14·27 ng/ml for non-Hispanic Blacks. For each race/ethnicity, mean January-adjusted vitamin D increased as the UV index rose. In the lowest quartile (January UV index<3·44), an estimated 75·1 % of non-Hispanic Blacks and 38·1 % of Hispanics were deficient in vitamin D, compared with 25·7 % of non-Hispanic Whites. In the highest quartile (January UV index > 6·00), an estimated 60·2 % of non-Hispanic Blacks and 25·6 % of Hispanics were deficient in vitamin D, compared with 12·8 % of non-Hispanic Whites.

Fig. 3 (colour online) Estimates of serum 25-hydroxyvitamin D, adjusted to January, by January UV index in census tract of residence; adults aged ≥18 years (n 14 319), National Health and Nutrition Examination Survey (NHANES), 2000–2006. Results shown for: (a) January UV index <3·44 (quartile 1); (b) January UV index 3·44–4·16 (quartile 2); (c) January UV index 4·17–6·00 (quartile 3); and (d) January UV index >6·00 (quartile 4). Vertical lines represent thresholds for vitamin D deficiency (<15 ng/ml) and insufficiency (<20 ng/ml or <30 ng/ml, depending on the source)
Discussion
Previous literature suggests that it is difficult for adults residing in Boston and north to produce enough vitamin D in winter(Reference Terushkin, Bender and Psaty43, Reference Holick47, Reference Webb, Kline and Holick50), resulting in widespread wintertime vitamin D deficiency at northern latitudes(Reference Merewood, Mehta and Grossman51). Our results suggest that wintertime vitamin D insufficiency in minority populations is pervasive across the USA. Using nationally representative NHANES data from 2000–2006, we estimated that 84·2–100 % of non-Hispanic Blacks and 56·3 –91·9 % of Hispanics were insufficient in vitamin D in January (depending on a 20 v. 30 ng/ml threshold). Even in non-Hispanic Whites, vitamin D insufficiency during winter was not uncommon, with an estimated 34·8–78·2 % of individuals affected.
Moreover, the association of race with wintertime vitamin D appeared at least as strong as that of geography. The estimated disparity in January vitamin D levels between non-Hispanic Whites and non-Hispanic Blacks across the USA (23·75 – 13·21=10·54 ng/ml, Fig. 2) exceeded that between non-Hispanic Whites residing in the lowest v. highest quartile of the January UV index (24·38 – 19·79=4·59 ng/ml, Fig. 3). Similarly, more non-Hispanic Blacks were estimated to be deficient in January vitamin D in the highest quartile of the UV index than were non-Hispanic Whites in the lowest quartile of the UV index (60·2 % v. 25·7 %, Fig. 3). The association of Hispanic ethnicity with vitamin D appeared similarly strong to geography; 25·7 % of non-Hispanic Whites residing in counties in the lowest quartile of the January UV index and 25·6 % of Hispanics residing in counties in the highest quartile of the January UV index were deficient in vitamin D (Fig. 3).
Alternatively viewed, 95 % confidence intervals for our regression models suggested that a 1-point increase in the UV index was associated with a 0·35–0·67 ng/ml increase in 25(OH)D (coefficient=0·51; Table 2, model 1). Since the January UV index ranges from 0 to 5 in the USA(40), geography contributed up to 3·35 ng/ml of wintertime vitamin D (0·67×5). This magnitude was about the same as the decrease in January vitamin D associated with Hispanic ethnicity (3·41 ng/ml) but well below that for non-Hispanic Black race (7·47 ng/ml; Table 2, model 1).
These results suggest a potentially more pervasive vitamin D insufficiency among non-Hispanic Blacks in winter than previously known. For example, a 2011 Institute of Medicine report using 2003–2006 NHANES data suggested that 53·6 %, 27·2 % and 10·6 % of non-Hispanic Blacks, Mexican Americans and non-Hispanic Whites had vitamin D levels <16 ng/ml (40 nmol/l)(1, Reference Looker, Dawson-Hughes and Calvo2), similar to the percentages we found for vitamin D deficiency in Fig. 2. Differences may be in part due to methodology; the Institute of Medicine adjusted vitamin D levels to northern latitudes (across seasons)(1), while we adjusted to January (across latitudes). Our analysis further adds the distribution of vitamin D levels by race/ethnicity and geography. Using the Institute of Medicine’s threshold of ≤20 ng/ml for vitamin D insufficiency, we found that 84·2 % of non-Hispanic Blacks, 56·3 % of Hispanics and 34·8 % of non-Hispanic Whites were insufficient in January; results were more extreme (including 78·2 % of non-Hispanic Whites) at thresholds of ≤30 ng/ml.
Previous research also suggests that it may be infeasible to synthesize adequate quantities of vitamin D from sunshine in the northern USA(Reference Godar, Pope and Grant52–Reference Webb and Engelsen54). Our results extend this finding to wide portions of the USA for Hispanics and non-Hispanic Blacks. In particular, it may be much more time-consuming, and therefore impractical, for minorities to synthesize the RDA of vitamin D from sunshine in winter, compared with non-Hispanic Whites (Fig. 1).
Our results support the need for further evidence to evaluate the benefits of prioritized vitamin D testing and supplementation of minority populations in winter. The US Preventive Services Task Force found incomplete evidence to recommend for or against vitamin D supplementation in postmenopausal women to prevent fractures(Reference Moyer55), but did not consider the general population or subgroups based on race/ethnicity. Similarly, the Canadian province of British Columbia, which borders Washington state, suggested that most adults would benefit from a supplement of 20–25 µg (800–1000 IU) daily, but did not consider potential differences based on race/ethnicity(56). Some evidence from community-based studies suggests that adults could tolerate far higher doses without toxicity(Reference Garland, French and Baggerly57), although the exact threshold is unknown. These potential benefits would need to be weighed against lower fracture risk in blacks(Reference Jacobsen, Goldberg and Miles58) and recent research suggesting that black individuals tend to have lower levels of vitamin D-binding protein(Reference Powe, Evans and Wenger59), potentially signifying fewer health risks from lower total vitamin D than the general population(Reference Powe, Evans and Wenger59). Yet, given the possibility that vitamin D may be associated with disease(1–Reference Lappe and Heaney23), it may be reasonable to investigate a potential association between racial disparities in vitamin D and racial disparities in mortality.
Limitations
NHANES conducts examinations at northern latitudes during summer months and southern latitudes during winter months(Reference Terushkin, Bender and Psaty43). However, because the decrease in vitamin D associated with a UV index <3 was larger than the increase associated with a UV index ≥8 (Table 2), we expect that deficiencies would be even more prevalent were wintertime data available at all latitudes. Second, we did not have information on dietary intake of vitamin D (outside of supplementation), physical activity and outdoor occupation, which may be positively associated with vitamin D(Reference McCullough, Weinstein and Freedman48, Reference Freedman, Dosemeci and McGlynn60). Third, although our findings suggest that individuals with more sun exposure had higher vitamin D, we did not consider the risk of skin cancer.
Conclusions
Wintertime vitamin D insufficiency is pervasive among minority populations, potentially contributing to racial disparities in disease incidence and mortality. Additional research is needed to assess the benefits of preferential routine vitamin D supplementation in minorities.
Acknowledgements
Acknowledgements: This study was completed while G.B.T. was with the Departments of Population Health and Medicine, New York University School of Medicine, New York, NY, USA. The work was performed at the National Center for Health Statistics, Hyattsville, MD, USA and the National Bureau of Economic Research, Cambridge, MA, USA. Financial support: This study was funded by the A. David Mazzone Awards Program of the Dana-Farber Cancer Institute (grant number 2011_DR_002). The A. David Mazzone Awards Program had no role in the design, analysis or writing of this article. Conflict of interest: None. Authorship: conception and design: G.B.T., D.M.C. and N.L.K.; analysis and interpretation of the data: G.B.T., D.M.C., E.G. and N.L.K.; drafting of the article: G.B.T.; critical revision of the article for important intellectual content: G.B.T., D.M.C., E.G. and N.L.K.; final approval of the article: G.B.T., D.M.C., E.G. and N.L.K.; provision of study materials or patients: not applicable; statistical expertise: G.B.T.; administrative, technical or logistic support: G.B.T.; collection and assembly of data: G.B.T. Ethics of human subject participation: This study was considered exempt by the Institutional Review Board of Harvard Medical School and did not meet criteria for human subjects review for the Institutional Review Board of New York University School of Medicine.
Appendix 1
Further details on the outdoor exposure required to synthesize vitamin D
As described in the main text, we estimated the number of minutes outdoors at noon in each calendar month for fair-skinned white, Hispanic and dark-skinned black individuals to synthesize 15 µg (600 IU) of vitamin D, the (RDA) for adults ≤70 years(1). This calculation was defined as the minimum erythemal dose (MED) of UV radiation required for individuals of each race to synthesize 15 µg (600 IU) of vitamin D, divided by the MED supplied per minute of noontime outdoor exposure. In this Appendix, we describe our calculations in more detail.
For the numerator, we state in the main text that we followed dermatology literature to expect that fair-skinned white, Hispanic and dark-skinned black individuals would require 0·25, 0·56 and 1·25 MED of UV radiation, respectively, to synthesize 15 µg (600 IU) of vitamin D, with 25·5 % of body surface area – the head, neck, arms and hands – uncovered. These numbers derive from an estimated 0·42, 0·94 and 2·08 MED (87·6, 197·0 and 437·8 J/m2) of UV radiation, respectively, to synthesize 25 µg (1000 IU) of vitamin D(Reference Terushkin, Bender and Psaty43). Since the dermatology literature suggests linearity of this relationship(Reference Terushkin, Bender and Psaty43), we multiplied by 60 % (15 µg/25 µg, 600 IU/1000 IU).
For the denominator, we used the definition of the UV index (1 unit=0·0072 MED/min)(Reference Kiedron, Stierle and Lantz44). Each unit of the UV index is defined as 25 mW/m2, or 0·43 MED/h(Reference Kiedron, Stierle and Lantz44).
We also assumed a relationship between average maximum daily temperature and body parts exposed to sunlight (≥23·9°C (≥75°F): face, neck, hands, arms exposed; 15·6–23·8°C (60–74°F): face, neck, hands, lower arms exposed; −1·1 to 15·5°C (30–59°F): face, neck, hands exposed; −9·4 to −1·2°C (15–29°F): face, neck exposed; ≤−9·5°C (≤14°F): face exposed). Following previous literature, we assumed that the face, neck, hands, upper arms and lower arms encompassed 3·5 %, 2·0 %, 6·0 %, 8·0 % and 6·0 % of body surface area, respectively(Reference Lund and Broder46).
Appendix 2
Complete regression results for Table 2. Dependent variable: serum 25-hydroxyvitamin D (ng/ml)