Colorectal cancer (CRC) is the third and second most common cancer in men and women respectively, worldwide. Almost 55 % of cases occur in more developed countries( Reference Ferlay, Soerjomataram and Ervik 1 ) and CRC is the third most common cancer in both genders in the USA( Reference Siegel, DeSantis and Jemal 2 ).
Results from international correlation and migrant studies suggest that environmental factors, including diet, may cause many cases of CRC( Reference Armstrong and Doll 3 ). Observational studies( Reference Austin, Adair and Galanko 4 – Reference Wakai, Date and Fukui 14 ) find that higher intakes of red meat and fat from animal sources (or saturated fat), and lower intakes of vegetables, fruits, Ca and dietary fibre, are associated with increased risk of CRC.
Dairy foods are consumed frequently around the world. The National Health and Nutrition Examination Survey estimated that 92 % of Americans consume dairy products. Dairy products and Ca may affect health, including the risk of cancer. Intriguingly, they may have both protective and harmful effects( 15 ). For CRC, many epidemiological studies have suggested protection, but as yet results are inconclusive( Reference Murphy, Norat and Ferrari 16 – Reference Cho, Smith-Warner and Spiegelman 19 ). The World Cancer Research Fund/American Institute for Cancer Research Continuing Update Report( 20 ) classifies dairy and Ca as ‘probable’ protective agents for CRC.
Preliminary investigations from the Adventist Health Study-2 population found a reduced incidence of cancers of the gastrointestinal tract among those who adopted a lacto-ovo-vegetarian dietary pattern( Reference Tantamango-Bartley, Jaceldo-Siegl and Fan 21 ). The range of dairy product consumption in this cohort is wide, as some are vegan (no dairy), many are lacto-ovo-vegetarian (low dairy on average) and about half are non-vegetarian (typical dairy consumption). Moreover, as much of their Ca intake comes from vegetable sources, the correlation between log(Ca) and log(dairy food) intakes, as used in analyses, was only 0·35. Thus, variance inflation and collinearity are not major problems when evaluating independent associations of these variables. Within this unusually informative population, in the present study we investigated independent associations between dairy foods, total dairy intake, as well as Ca from different sources (including supplements), and the risk of CRC.
Methods
Population
The Adventist Health Study-2 is an ongoing prospective study designed to investigate the association between diet and lifestyle factors and various diseases including cancer. A detailed description of the study design has been published( Reference Butler, Fraser and Beeson 22 ). The study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving human subjects/patients were approved by the Loma Linda University Internal Review Board. Written informed consent was obtained from all subjects/patients.
Enrolment between 2002 and 2007 was indicated by completion of a detailed baseline questionnaire that included the food frequency items. Finally, there were 96 001 enrolees to the present study, all US or Canadian members of the Adventist Health Study-2. Of these approximately 25 % were Black subjects (African Americans and West Indians living in the USA or Canada). Other races were labelled ‘White’ and included about 3 % Hispanics and 3 % Asians. In total, individuals included in the present study represented an estimated 25–30 % of the American Adventist population eligible to participate.
Linkage with cancer registries was possible for the 90 844 Adventist Health Study-2 participants who lived in forty-eight US states or Washington DC. Excluded were those with: (i) age less than 25 years or missing data for age or sex (n 446); (ii) improbable response patterns in questionnaire data (n 369); (iii) greater than sixty-nine missing values in dietary data (n 1720); (iv) estimated energy intake <2093 kJ/d (<500 kcal/d) or >18828 kJ/d (>4500 kcal/d; n 3174); (v) a history of prevalent cancers (n 7402), (vi) no informed consent (n 17); and (vii) missing date of cancer diagnosis (n 4). Therefore, the final analytic population comprised 77 712 subjects.
Data collection and exposures of interest
At baseline, study participants completed a validated self-administered mailed FFQ( Reference Jaceldo-Siegl, Fan and Sabaté 23 ), which assessed the usual diet during the past year. This questionnaire contained over 200 food items including fruits, vegetables, legumes, grains, oils, dairy products, meats, beverages, and commercially prepared products such as dietary supplements, dry cereals, meat substitutes and soya products. Participants reported their average frequency of intake and usual serving size using predefined frequency and serving size categories according to the food under evaluation.
One variable of interest for the current study was total dairy( 24 ). Total kilocalories (i.e. energy from) and grams of dairy intake, and grams of intake of individual dairy foods (cheese, milk and yoghurt), were computed by multiplying the frequency of consumption of each dairy product by the kilocalories or grams of dairy content of the specific serving size. We prefer units of kcal/d over g/d for total dairy, given that the latter has an over-weighting for milk consumption, given its content of water, that is presumably biologically inert in this context. Nevertheless, an analysis for total dairy using g/d is included (Table 4) for comparison with other studies that used this metric. Fat, protein and carbohydrate from dairy products were also assessed in the present study. Detailed descriptions of the methods for measuring nutrient intake using the FFQ, and its validation by repeated 24 h recalls, have been published elsewhere( Reference Jaceldo-Siegl, Knutsen and Sabate 25 ). Dairy protein had an energy-adjusted de-attenuated validity correlation of 0·77 (95 % CI 0·73, 0·80) for Whites and 0·58 (95 % CI 0·51, 0·64) for Blacks. For dairy fat, the corresponding correlations were 0·66 (95 % CI 0·61, 0·71) and 0·56 (95 % CI 0·49, 0·62), respectively( Reference Jaceldo-Siegl, Knutsen and Sabate 25 ).
Ca was also of special interest, specifically total Ca, Ca from dietary sources, from dairy products and from supplements. Energy-adjusted validity correlations were 0·63 (95 % CI 0·58, 0·68) for White and 0·73 (95 % CI 0·68, 0·77) for Black subjects. For dietary Ca, the corresponding correlations were 0·53 (95 % CI 0·47, 0·59) and 0·54 (95 % CI 0·46, 0·60), respectively( Reference Jaceldo-Siegl, Knutsen and Sabate 25 ).
Cancer and death ascertainment
Cancer cases were identified by computer-matching identifiers from Adventist Health Study-2 cohort members to those from State Cancer Registries. At the time of writing, matches had been made with all US states excepting Maine and Wisconsin. Most registries had complete data through 2011 at the time of matching, and a few through 2012. Supplementing this, a biennial mailed questionnaire contained questions about any new cancer diagnosis. If a self-reported cancer was not found in the registry linkage, further follow-up by telephone clarified whether it was a true cancer case or not. If a new cancer seemed possible, medical records were requested and reviewed by the principal investigator.
Deaths were ascertained by annual matching with the US National Death Index. As vital status for Canadian subjects is not yet available, Canadian subjects (n 3800) are not included in these analyses. New cancers of the colon and/or rectum were those diagnosed after baseline for each subject. Those with previous cancers were excluded. The cancer site was identified using the International Classification of Diseases-10 (ICD-10-CM). Cancers of the sigmoid colon were allocated to the rectum. Carcinomas in situ were not included as cases.
Statistical analysis
All analyses were performed with the statistical software package SAS version 9.3 or the R computer language( 26 ). Multiple imputation was used to fill the small amounts (2–10 % for different variables) of missing dietary data, at the level of the FFQ question. This imputation was usually guided by a random sub-sample of originally missing data that were subsequently filled in by telephone contact( Reference Fraser and Yan 27 ). Multivariate hazard rates and confidence intervals were calculated using five imputed data sets, and the five sets of β coefficients and their estimated variances were used to form composite point estimates and variances( Reference Schafer 28 ).
Multivariate Cox regression models were developed to estimate hazard ratios (HR) and 95 % CI. Attained age (ages during the period of observation) was the time variable and all models were left-censored so excluding person-time before the ages at entry. Person-time was counted through to a CRC outcome, the year end for cancer registry completion in the state of that subject’s residence, or censoring for other reason (e.g. death, or rarely emigration, request to drop from the study).
A basic model including only the independent variables of interest, namely race and gender, was evaluated first. Other candidate covariates were selected based on review of the literature. These included: family history of CRC cancer (yes/no), education (high school or less, some college, at least college graduate), duration of vigorous activity (none, less than 60 min/week, at least 60 min/week), BMI (less than 25 kg/m2, 25 to 30 kg/m2, more than 30 kg/m2), alcohol consumption within 2 years (yes/no), smoking status (ever/never), appropriate screening for CRC cancer (yes/no), personal history of peptic ulcer (yes/no), personal history of polyps (yes/no), personal history of diabetes mellitus (yes/no), aspirin use, statin use (for both medications, at least weekly for at least the last 2 years; yes/no), fibre (grams of intake), unprocessed red meat, processed red meat, fish and poultry (grams of intake). Ca and total dairy were included in the same models as appropriate.
Nutrient composition was estimated using the Nutrition Data System for Research (NDS-R) 2008, a database updated annually while maintaining relevant information true to the version used for data collection( Reference Schakel, Sievert and Buzzard 29 ). Where commercially prepared food products were not found here, we created recipes to generate nutrient composition at the ingredient level.
For the CRC screening variable, participants over 50 years old and at low or average risk of developing CRC (based on absence of history of adenomatous polyps, inflammatory bowel disease, family history of CRC, polyps, a hereditary CRC syndrome), and who reported that they had been screened by a colonoscopy within the last 5 years, and/or faecal occult blood test within the last year, were considered compliant (American Cancer Society criteria). Those at higher risk of developing CRC were considered compliant if they reported screening before the age of 50 years old with colonoscopy every 1 to 2 years( 30 ). A nested analysis among those who should have screened used an indicator variable to identify those at least 50 years of age or less than 50 but high risk, and then evaluated the product between this variable and screening status.
Dietary variables were log-transformed prior to model building. All non-zero dietary variables were energy-adjusted (residual method), except for Ca from supplements, and modelled as continuous variables. Then, for each dairy nutrient, dairy foods and Ca, the HR comparing mid-points of extreme quintiles (Qupper and Qlower) on risk of CRC was calculated by (Qupper/Qlower) β , where β is the regression coefficient for the variable of interest. The energy adjustments were race specific as Black subjects consumed much less dairy on average and validity coefficients also varied somewhat by race.
Possible interactions between covariates and the relative risks of CRC at different levels of the exposures of interest were tested using product terms between the exposures of interest and these covariates. As no interaction terms were statistically significant, none were included in the final model. The dairy nutrients (kilocalories, protein, fat) were highly correlated with each other (r≥0·8). Therefore, individual models were built for each nutrient. All dairy foods, however, could be included in a single model since intercorrelations between them were relatively low.
Finally, using a concurrent second FFQ and repeated 24 h dietary recalls from our calibration study subjects (n 1011), who represent the cohort( Reference Jaceldo-Siegl, Knutsen and Sabate 25 ), we formed new variables, E(Recall|FFQ data, covariates), for all dietary variables in selected models in a race-specific fashion. These data were then used for regression calibration( Reference Thomas, Stram and Dwyer 31 , Reference Fraser 32 ) of selected key analyses to at least partially correct for the effects of measurement errors. Confidence intervals for calibrated coefficients were estimated by the bootstrap BCa method( Reference Efron and Tibshirani 33 ) (n samples 2000).
Results
During a mean follow-up of 7·8 years, 380 new cases of colon cancer and 111 cases of rectal cancer were identified.
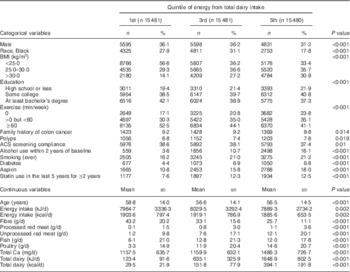
Table 1 shows the baseline characteristics of the study population, comparing subjects at first, third and fifth quintiles of intake of dairy foods. Higher dairy intake was positively associated with BMI, alcohol use within 2 years, a smoking history, prevalent diabetes, use of aspirin and statins, consumption of each of the meats, and also Ca. Dairy intake was negatively associated with male sex, percentage of Black subjects, exercise, age, and fibre consumption.
Table 1 Demographic characteristics by quintile of energy from total dairy intake in participants of the Adventist Health Study-2, USA, 2002–2011
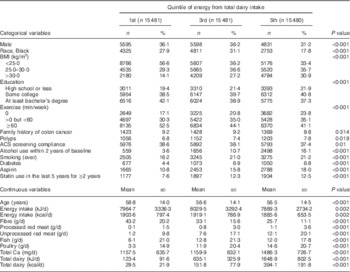
ACS, American Cancer Society.
The χ 2 test for homogeneity was used to compare quintiles of energy from total dairy intake for categorical variables, while ANOVA was used for comparisons of continuous variables.
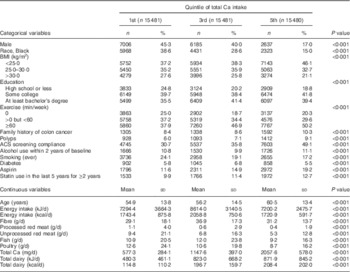
Table 2 compares the same variables across the first, third and fifth quintiles of Ca consumption. Higher Ca consumption was positively associated with education, exercise, family history of colon cancer, previous colon polyps, American Cancer Society screening compliance, use of aspirin and statins, age and dairy intake. Ca intake was negatively associated with male sex, percentage of Black subjects, BMI, a smoking history, and consumption of red meat and poultry.
Table 2 Demographic characteristics by quintile of total calcium intake in participants of the Adventist Health Study-2, USA, 2002–2011
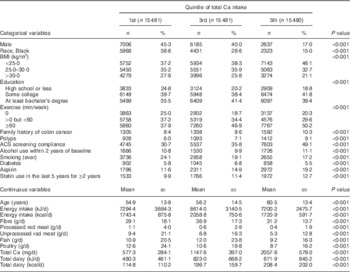
ACS, American Cancer Society.
The χ 2 test for homogeneity was used to compare quintiles of Ca intake for categorical variables, while ANOVA was used for comparisons of continuous variables.
Similar intakes of fibre, meat (including red meat), dairy nutrients (total, protein, fat, carbohydrate) and Ca (total, from diet, from diary and from supplements) were observed among cancer cases as compared with the cohort in univariate analyses (Table 3).
Table 3 Mean intakes of dairy and calcium by cancer of the colon and rectum among participants of the Adventist Health Study-2, USA, 2002–2011
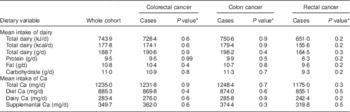
* Testing null hypothesis of no difference between cases and the whole cohort.
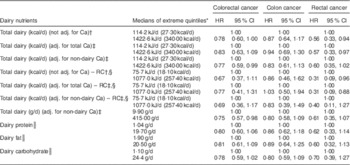
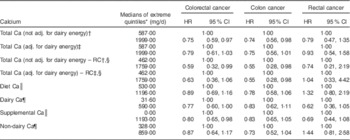
Proportional hazard regression analyses, associating intake of each selected dairy nutrient with risk of all CRC, cancers of the colon and rectum separately, are presented in Table 4. Results from regression calibration (measurement error-corrected) analyses gave support to a statistically significant inverse association between dairy intake (either adjusted for and hence independent of Ca, or unadjusted) and the risk of rectal cancer (HR=0·31, 95 % CI 0·09, 0·88 when adjusted for Ca), comparing extreme quintiles of intake as measured by the repeated recalls. In uncorrected analyses, comparing extreme quintiles of FFQ intake, rectal cancer again showed statistically significant inverse relationships with total energy from dairy intake whether this was adjusted for Ca or not (HR=0·57, 95 % CI 0·33, 0·97; P<0·041 when adjusted for Ca). Similar but non-significant trends were seen with dairy fat, protein and less clearly carbohydrate.
Table 4 Multivariate-adjusted (adj.) uncorrected and regression calibration (RC) hazard ratios (HR) describing associations between dairy intake and incident cancers of the colon and rectum among participants of the Adventist Health Study-2, USA, 2002–2011
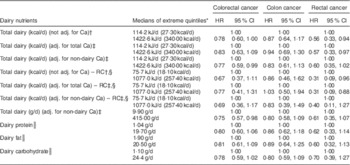
* Except for RC analyses, quintiles are from FFQ data.
† Adjusted for fibre, unprocessed red meat, processed red meat, fish, poultry, gender, race, BMI, education, alcohol consumption, cigarette smoking history, diabetes, use of aspirin, use of statin, physical activity, family history of colorectal cancer, history of polyps, screening for colorectal cancer.
‡ Adjusted in addition for either total Ca or only supplemental and non-dairy Ca (as indicated).
§ RC analyses. Units are kcal/d and quintiles are those from repeated recalls, as is appropriate for the RC.
║ Dairy nutrient variables are assessed individually in the regression models.
Associations between risk of colon cancer and total dairy (corrected and uncorrected analyses), dairy protein, dairy fat and colon cancer in uncorrected analyses were mostly close to the null when comparing extreme quintiles of intake. When adjusting only for non-dairy Ca (hence the total dairy variable includes any Ca effect), the HR for colon cancer was 0·83 (but not statistically significant) in both corrected and uncorrected analyses. Significant protective associations between total dairy intake and risk of colorectal cancer, whether dairy was measured in kcal/d (HR=0·77, 95 % CI 0·59, 0·99; P=0·048) or g/d (HR=0·75, 95 % CI 0·57, 0·98; P=0·037), were seen when adjusted for only non-dairy Ca, thus leaving a total dairy variable that includes any effects of dairy Ca. Significant protective associations between dairy variables and rectal cancer were not clearly seen when Black subjects were analysed separately (data not shown). However, with only twenty-five rectal cancer cases, no conclusions can be drawn. Supplemental Table 1 (see online supplementary material) shows corresponding results for each quintile of intake.
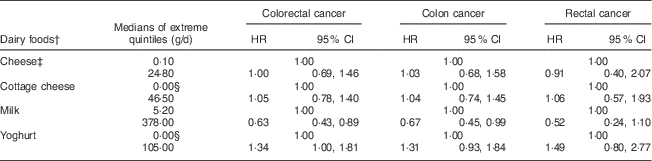
When analysing the nominated extreme intakes (usually quintiles) of specific dairy foods, evaluated independently and adjusting for supplemental and non-dairy Ca (Table 5), no significant effects were seen for cheese, cottage cheese or yoghurt. However, for milk, the results for risks of CRC (HR=0·63, 95 % CI 0·43, 0·89; P=0·013) and colon cancer (HR=0·67, 95 % CI 0·45, 0·99; P=0·047) were statistically significant, and that for rectal cancer trended in the same direction (HR=0·52, 95 % CI 0·24, 1·10; P=0·091). Supplemental Table 2 (see online supplementary material) shows corresponding results for each quintile of intake.
Table 5 MultivariateFootnote *-adjusted hazard ratios (HR) of the associations between dairy foods and incident cancers of the colon and rectum among participants of the Adventist Health Study-2, USA, 2002–2011
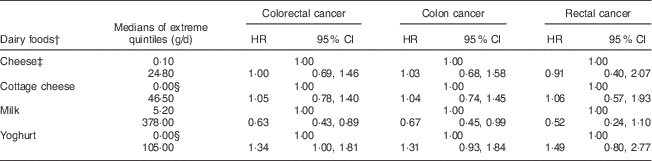
* Adjusted for supplemental Ca, non-dairy Ca, fibre, unprocessed red meat, processed red meat, fish, poultry, gender, race, BMI, education, alcohol consumption, cigarette smoking history, diabetes, use of aspirin, use of statin, physical activity, family history of colorectal cancer, history of polyps, screening for colorectal cancer.
† Exposures adjusted for each other.
‡ Excluding cottage cheese.
§ Compares zero intake with the median of the upper quartile of non-zero intakes.
Table 6 presents results of proportional hazard regression models where the exposures are total Ca intake, also particular sources of Ca, comparing the nominated extreme intakes (usually quintiles). Measurement error-corrected analyses gave significant support for the existence of a significant inverse association between total Ca intake and risk of colon cancer, whether adjusted for dairy energy or not (HR=0·55, 95 % CI 0·28, 0·98 when adjusted for dairy energy), comparing extreme quintiles of repeated dietary recall data. In uncorrected analyses, a statistically significant association was observed when comparing extreme quintiles of intake of total Ca, for risk of colorectal and colon cancers, and associations with total, dietary and non-dairy Ca were borderline significant when dairy energy was adjusted for. Associations between total and dietary sources of Ca and risk of rectal cancer were non-significant, either closer to the null or trended in the positive direction, with the exception of dairy Ca that was not adjusted for dairy energy (due to severe collinearity) and hence included non-Ca components of dairy. Supplemental Ca was significantly negatively associated with risk of CRC (HR=0·80, 95 % CI 0·65, 0·98; P=0·034); this also being nearly significant for both rectal and colon cancer end points while adjusting for dairy energy and dietary Ca. Supplemental Table 3 (see online supplementary material) shows corresponding results for each quintile of intake.
Table 6 Multivariate-adjusted (adj.) uncorrected and regression calibration (RC) hazard ratios (HR) describing associations between calcium intake and incident cancers of the colon and rectum among participants of the Adventist Health Study-2, USA, 2002–2011
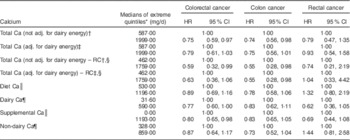
* Except for RC analyses, quintiles are from FFQ data. However, for supplemental Ca compares zero intake with the median of the upper quartile of non-zero supplemental Ca.
† Model 1: total Ca, fibre, unprocessed red meat, processed red meat, fish, poultry, gender, race, BMI, education, alcohol consumption, cigarette smoking history, diabetes, use of aspirin, use of statin, physical activity, family history of colorectal cancer, history of polyps, screening for colorectal cancer.
‡ Model 2: Model 1 with addition of dairy energy.
§ Quintiles are from the repeated dietary recall data as is appropriate for the RC.
|| Model 3: Ca from diet and Ca from supplements replace total Ca as separate variables. Other covariates are identical to Model 2.
¶ Model 4: Ca from dairy, non-dairy Ca and Ca from supplements are the three calcium variables, and dairy energy is omitted. Other covariates are identical to Model 2.
Discussion
To summarize these findings, it is clear that dairy and/or Ca intake and milk consumption are significantly negatively associated with risk of CRC in these data. Risk of colon cancer appears to be most strongly associated with Ca intake and not with dairy intake when adjusted for total Ca intake. Findings for rectal cancer have wider confidence intervals and are therefore less secure, but this cancer appears to be negatively associated with dairy intake, and more clearly with the non-Ca dairy components. The rectal cancer association with dairy is most significant after adjusting for total Ca, and neither total Ca nor non-dairy Ca showed a protective association after adjustment for dairy intake. Milk intake was significantly associated with risk of colon cancer and also strongly associated with rectal cancer (but here only borderline significant). This presumably reflects its Ca and non-Ca components. Supplemental Ca was also significantly associated with risk of CRC, there not being a clear difference between colon and rectal cancers in this case. However, this somewhat surprising result for rectal cancer may be due to chance (P=0·105). Regression calibration applied to the major results revealed stronger associations as expected and generally preserved statistical significance. In our data, the magnitudes of the significant regression calibration HR estimates were substantial, in the range of 0·31 to 0·55, when comparing extreme quintiles.
The association between dairy intake and the risk of CRC has been explored in other epidemiological studies( Reference Murphy, Norat and Ferrari 16 – Reference Cho, Smith-Warner and Spiegelman 19 , Reference McCullough, Robertson and Rodriguez 34 – Reference Aune, Lau and Chan 36 ). The evidence generally favours a protective effect of dairy. However, most studies appear to have used models including either Ca or dairy alone, rather than evaluating both independently. Hence from those results, a dairy effect, a Ca effect, or both, is possible. Of the dairy foods, associations with milk have been most consistent as we found, although some studies disagree.
Higher consumption of total dairy products has been associated with reduced risk of CRC (both colon and rectal cancers) in the EPIC (European Prospective Investigation into Cancer and Nutrition) cohort study( Reference Murphy, Norat and Ferrari 16 ) and a cohort of Swedish men( Reference Larsson, Bergkvist and Rutegard 35 ). A large Finnish cohort study also indicated that milk protein and the consumption of milk products were inversely associated with CRC risk( Reference Pietinen, Malila and Virtanen 37 ). In another Swedish cohort, greater intake of high-fat dairy foods was associated with a reduced risk of CRC, a protective effect that was stronger for distal colon cancer, but was not seen for rectal cancer. Considering individual dairy foods, the strongest inverse association was observed for high consumption of cheese( Reference Larsson, Bergkvist and Wolk 38 ). Findings from other studies on the relationship between cheese consumption and CRC risk have mostly been null( Reference Ralston, Truby and Palermo 39 ). Furthermore, a systematic review and meta-analysis of nineteen cohort studies( Reference Aune, Lau and Chan 36 ) found that high intakes of total dairy products were associated with a statistically significant reduction in CRC risk and there was evidence for an inverse association with milk but not cheese or other dairy products. When stratified to colon and rectal sites the inverse associations were only present for colon cancer. These inconsistent results may be due to variable confounding between dairy and Ca (which cannot be determined from the published data) or perhaps to chance.
Biologically, a protective effect of dairy may be explained by its relatively high content of Ca, vitamin D, lactoferrin and conjugated linoleic acid (CLA). Each of these has been postulated to protect against the development of CRC( Reference Norat and Riboli 40 ). Lactoferrin is an Fe-binding antimicrobial protein present in exocrine secretions such as milk( Reference Norat and Riboli 40 ). As found in animal studies, this glycoprotein may enhance CRC mucosal immunity and decrease inflammation-related malignant transformation( Reference Ye, Zheng and Fan 41 ). Lactoferrin also increases apoptosis and the expression of Fas in the colon mucosa during both early and late stages of carcinogenesis in rats. Fas is a member of the TNF receptor family( Reference Fujita, Matsuda and Sekine 42 ). It thus may act as a chemo-preventive factor for carcinogenesis. Lipid components of dairy products, including butyric acid and CLA, may protect against tumorigenesis( Reference Norat and Riboli 40 ). Butyric acid is a major metabolite in colonic lumen that possesses anti-inflammatory and anti-tumour properties( Reference Celasco, Moro and Aiello 43 ). Milk lipid is the main dietary source of CLA. Studies of cultured cell lines have shown that CLA may inhibit the proliferation of CRC tumour cells by modifying the fluidity of cell membranes, reducing the synthesis of prostaglandins and stimulating the immune response( Reference Norat and Riboli 40 ). Amounts of these substances may differ greatly in different dairy foods depending on fat content (butyric acid, CLA)( Reference Meraz-Torres and Hernandez-Sanchez 44 ), protein content (lactoferrin)( Reference Kruger 45 ) and bacterial activity. Differences in the colonic microbiome between vegans and lacto-ovo vegetarians( Reference Zimmer, Lange and Frick 46 ) may also result from differences in dairy consumption.
Several epidemiological studies have found a 14 to 45 % lower risk of CRC in subjects with at least moderate intakes of Ca (≥700 mg/d)( Reference McCullough, Robertson and Rodriguez 34 , Reference Pietinen, Malila and Virtanen 37 , Reference Wu, Willett and Fuchs 47 , Reference Terry, Baron and Bergkvist 48 ). However, in one US study this benefit appeared to plateau, or level off, at an intake of approximately 1200 mg/d( Reference McCullough, Robertson and Rodriguez 34 ). It has been reported that a protective association with Ca intake seemed more evident for distal than proximal colon cancer( Reference Wu, Willett and Fuchs 47 , Reference Terry, Baron and Bergkvist 48 ). Inconsistencies between findings are particularly evident when assessing sources of this mineral. One study found that total Ca intake of greater than 1445 mg/d, compared with less than 956 mg/d, was associated with a statistically significant 32 % decrease in risk of CRC and a 39 % decrease in rectal cancer, and that dietary Ca was also associated with decreased risk of CRC( Reference Larsson, Bergkvist and Rutegard 35 ). Ca from supplements has also been associated with reduced risk of CRC among people who consumed ≥500 mg/d( Reference McCullough, Robertson and Rodriguez 34 ). In the Iowa Women’s Health Study of postmenopausal women, Ca intake of ≥1280 mg/d from both diet and supplements combined was associated with a 41 % reduction in risk of rectal cancer( Reference Zheng, Anderson and Kushi 49 ).
Ca has been shown to reduce proliferation, stimulate cellular differentiation, and induce apoptosis in cells of the gastrointestinal tract. Additionally, the binding of Ca to bile and fatty acids in the gastrointestinal tract has been hypothesized to reduce damage to the large bowel mucosa( Reference Lipkin and Newmark 50 ).
Is it possible that these associations, as is suggested by our results, could be rather different in the rectum as compared with the colon? The rectal mucosa is exposed to the final faecal products (including bacterial products) for longer periods than the colonic mucosa as the rectum is a storage organ. Conceivably, colonic bacterial products resulting from interactions with residual dairy components may not be fully expressed until the rectum.
Strengths of the present study are its prospective design, the validation of new colon and rectum cancers by matching to cancer registries or by review of medical records, and the unique lifestyle of the Adventist Health Study-2 population with its wide variety of dietary habits. Many vegetarians obtain much Ca from non-dairy sources. Thus, collinearity between dairy and Ca is reduced. Few other Western cohorts have large numbers consuming no dairy or very low dairy. This produces extreme quintiles of dairy that are far apart, which also increases statistical power and reduces the likelihood of confounding by non-dietary factors. Another strength is the representative calibration study that allows application of regression calibration as a measurement error correction strategy. Finally, the Adventist Health Study-2 cohort is a sociodemographically diverse population, aside from membership in the Seventh-day Adventist Church, potentially producing results that have general application.
Limitations of our study include unavoidable inaccuracies in the assessment of food consumption, although published results comparing questionnaire with six 24 h dietary recalls show relatively good validity correlation for both the dairy foods( Reference Jaceldo-Siegl, Fan and Sabaté 23 ) and nutrients( Reference Jaceldo-Siegl, Knutsen and Sabate 25 ) that are the focus of the current paper. Any such misclassification should be non-differential, usually biasing the results towards the null. The regression calibration analyses partially allay this concern. These results come from a population where about half are vegetarian, which must be considered when generalizing results. However, we did adjust for obvious differences from the general population in the analyses.
Conclusion
Dairy intake, independent of Ca intake, was associated with a decreased risk of rectal cancer, and Ca intake was associated with decreased risk of colon cancer independent of dairy intake. Milk intake and supplemental Ca also gave evidence of protection although not clearly differing between the two cancer sites. Inconsistencies in other work may result from confounding between Ca and dairy. Further studies are needed to confirm these independent associations of Ca and dairy products, and to further assess possible differences between associated risks of colon and rectal cancers.
Acknowledgements
Acknowledgements: The authors thank the following cancer registries for their collaboration: ‘Alaska Cancer Registry’, ‘Alabama Statewide Cancer Registry’, ‘Arizona Cancer Registry’, ‘Arkansas Central Cancer Registry’, ‘California Cancer Registry’, ‘Colorado Central Cancer Registry’, ‘Connecticut Tumor Registry’, ‘District of Columbia Cancer Registry’, ‘Delaware Cancer Registry’, ‘Florida Cancer Data System’, ‘Georgia Comprehensive Cancer Registry’, ‘Hawaii Tumor Registry’, ‘Cancer Data Registry of Idaho’, ‘Iowa Cancer Registry’, ‘Illinois State Cancer Registry’, ‘Indiana State Cancer Registry’, ‘Kansas Cancer Registry’, ‘Kentucky Cancer Registry’, ‘Louisiana Tumor Registry’, ‘Maryland Cancer Registry’, ‘Massachusetts Cancer Registry’, ‘Michigan Cancer Surveillance System’, ‘Minnesota Cancer Surveillance System’, ‘Mississippi Cancer Registry’, ‘Missouri Cancer Registry and Research Center’, ‘Montana Central Tumor Registry’, ‘Nebraska Cancer Registry’, ‘Nevada Central Cancer Registry’, ‘New Hampshire State Cancer Registry’, ‘New Jersey State Cancer Registry’, ‘New Mexico Tumor Registry’, ‘New York State Cancer Registry’, ‘North Carolina Central Cancer Registry’, ‘North Dakota Statewide Cancer Registry’, ‘Ohio Cancer Incidence Surveillance System’, ‘Oklahoma Central Cancer Registry’, ‘Oregon State Cancer Registry’, ‘Pennsylvania Cancer Registry’, ‘Rhode Island Cancer Registry’, ‘South Carolina Central Cancer Registry’, ‘South Dakota Cancer Registry’, ‘Tennessee Cancer Registry’, ‘Texas Cancer Registry’, ‘Utah Cancer Registry, NCI Contract HHSN261201300071’, ‘Vermont Cancer Registry’, ‘Virginia Cancer Registry’, ‘Washington State Cancer Registry’, ‘West Virginia Cancer Registry’ and ‘Wyoming Cancer Surveillance Program’. Financial support: This study was supported by the National Institutes of Health (grant number U01CA152939); the National Institute of Food and Agriculture (grant number 2010-38938-20924); and the World Cancer Research Fund (UK) (grant number 2009/93). The funders had no role in the design, analysis or writing of this article. Conflict of interest statement: None. Authorship: Y.T.-B. wrote the initial drafts of this manuscript and conducted many statistical analyses. K.J.-S. is the Adventist Health Study-2 study research nutritionist. Over many years she was integrally involved in preparation of the nutritional data for analyses, including the dairy/Ca data and other dietary covariates used in this study. She closely reviewed the final manuscript. A.M. is a statistician/data manager who prepared the data files, with imputation of missing data. He also reviewed the final manuscript. G.E.F. performed the final analyses for this manuscript, and re-wrote segments of the manuscript. He also received funding for this work and conceived of the study. Ethics of human subject participation: This study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving human subjects were approved by the Internal Review Board of Loma Linda University. Written informed consent was obtained from all subjects.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/S1368980017001422