The universal evolution of antibiotic resistance poses a serious threat to the health of hospitalized patients, with the use of antibiotics being considered a major determinant in resistance development [Reference Gyssens1]. In addition to increased morbidity, mortality and hospital costs, inappropriate antibiotic use may result in minimizing the ‘lifespan’ of antibiotic drugs and therefore limiting available treatment options [Reference Paterson2, Reference Smith and Gould3]. The need to reverse the resistance patterns has prompted calls for prudent antibiotic prescribing for hospital inpatients, through the development and the implementation of antibiotic stewardship programmes [Reference Paterson2, Reference MacDougall and Polk4]. However, experience has shown that more insight into the implementation of antibiotic guidelines in routine practice is needed in order to determine drivers of antibiotic prescribing [Reference Aldeyab5]. Point prevalence surveys of antibiotic use provide useful data on the magnitude of inappropriate prescribing, thus, forming the basis for defining antibiotic-resistance control strategies and priorities [Reference Aldeyab5–Reference Ansari7]. A recent antibiotic point prevalence survey, which utilized data from 20 European hospitals, was performed in order to standardize a method for antibacterial surveillance in hospitals and to identify targets for quality improvement [Reference Ansari7]. In the latter study, the authors identified the following three targets for quality improvement: duration of preoperative prophylaxis, indication documented in case-notes, and adherence to hospital antibiotic guidelines [Reference Ansari7]. A detailed knowledge of these performance indicators is essential for informing more focused interventions aimed at the containment of antibiotic resistance and improving treatment protocols.
The objective of this research was first to assess current patterns of antibiotic prescribing in secondary healthcare settings in Northern Ireland, and second to determine targets for improving the quality of antibiotic prescribing, utilizing the European Surveillance of Antimicrobial Consumption (ESAC) audit tool [Reference Ansari7]. ESAC is an international data collection network which aims to improve antimicrobial prescribing through collecting data on patterns of antibiotic prescribing utilizing a standard validated method [Reference Vander Stichele8]. A total of four acute teaching hospitals participated in the point prevalence study. These hospitals represent four of the main acute-care hospitals from four of the five Health and Social Care (HSC) Trusts in Northern Ireland (the total five HSC Trusts serve a total population of about 1·7 million), as follows: Antrim Area Hospital (426 beds) in the Northern Health and Social Care Trust (NHSCT), Ulster Hospital (561 beds) in the South Eastern Health and Social Care Trust (SEHSCT), Altnagelvin Hospital (495 beds) in the Western Health and Social Care Trust (WHSCT) and Craigavon Hospital (426 beds) in the Southern Health and Social Care Trust (SHSCT). These hospitals all provide acute, general medical and surgical services, support a range of outpatient facilities and act as centres for the coordination of health service provision throughout their defined geographical areas in Northern Ireland.
The point prevalence survey involved collecting specific information, utilizing the ESAC audit tool (as described elsewhere) [Reference Aldeyab5], regarding patients who were in the hospital at 08:00 hours on the day of the survey. Clinical pharmacists were asked to conduct this survey on a specific day on their respective wards; the required data were gathered through reviewing patients' case-notes and prescribing charts. The survey was completed over a 2-week period in May–June 2009 for all participating hospitals; the average time required for conducting the survey varied between hospitals (2 days to 2 weeks). Of note, the audit was, in general, simple and precise in design and was completed in a straightforward manner. Data collected included details on the number of inpatients in each department, patients' age and gender, antimicrobial agents used, dose per administration, number of doses per day, route of administration, anatomical site of infection, duration of surgical prophylactic antibacterials (coded as 1 dose, 1 day, >1 day), compliance with the local hospital antibiotic guideline, and indication for therapy, i.e. community-acquired infection, hospital-acquired infection or medical/surgical prophylaxis.
Community-acquired infections were defined as cases where symptoms or antibiotics started <48 h after a patient was admitted while hospital-acquired infections were defined as cases where symptoms started ⩾48 h after admission to hospital. The latter was classified into five categories as follows: (i) post-operative infection (within 30 days after surgery or 1 year after implant surgery), (ii) other intervention related infections (i.v. catheter, ventilator-associated pneumonia, continuous ambulatory peritoneal dialysis), (iii) Clostridium difficile-associated diarrhoea >48 h after admission or <30 days after previous admission, (iv) other hospital-acquired infection, and (v) infection present on admission from another hospital. Measuring adherence to the hospital antibiotic guideline was achieved by answering ‘yes’, ‘no’, ‘not assessable’, where the diagnosis was unclear or no local guideline existed for the specific indication, or ‘no information’. The empirical antibiotic guidelines recommend first-line antibiotic therapy for the treatment of common infections, e.g. respiratory tract infections, intra-abdominal infections, sepsis syndromes and urinary tract infections. Advice on accurate diagnosis, dosing, timing and duration of the antibiotic regimens was also provided in these guidelines. A quick reference guide was made available on each ward in addition to the full guidelines. Electronic versions of the guidelines were also available on Trust intranets. Compliance assessment was done based only on the information written in the patients' notes. This is based on the principle that recording indication for treatment is essential for compliance with the guideline. The percentage adherence rate to the hospital antibiotic guideline was calculated by dividing the number of adherent observations by the overall number of observations (i.e. adherent, non-adherent, not-assessable observations). Antibacterials were presented as classes belonging to group J01 (antibacterials for systemic use) of the Anatomical Therapeutic Chemical (ATC) classification system from the World Health Organization (WHO) Collaborating Centre for Drug Statistics Methodology. The study results were entered into Microsoft Access® database (Microsoft Corp., USA) and descriptive statistics and frequency analyses were performed using SPSS version 18 for Windows (SPSS Inc., USA).
A total of 1615 patients were surveyed in the four hospitals. Of the 1615 patients, 512 (32%) were treated with antibiotics, of whom 250 (49%) were male and 262 (51%) were female; the median age was 70 years. The treated patients received a total of 713 antibiotics. A relatively higher usage (n=416, 58·5%) of parenteral antibiotics was observed, compared with oral antibiotic use (n=295, 41·5%); variations in parenteral/oral antibiotic use were observed in the studied hospitals (Table 1). The indications for the prescribed 713 antibiotics were community-acquired infection (n=400, 56·1%), hospital-acquired infection (n=198, 27·8%), surgical prophylaxis (n=77, 10·8%), and medical prophylaxis (n=36, 5%); data for two patients were not available. In relation to patients with hospital-acquired infections, the highest antibiotic use was observed in the other hospital-acquired infection subgroup (64·1%), followed by the post-operative infection subgroup (18·7%), C. difficile-associated diarrhoea subgroup (7·1%), other intervention-related infections subgroup (5·6%), and infection present on admission from another hospital subgroup (4·5%). The most common sites of infection were the respiratory tract (29·9%), and the gastrointestinal tract (18·3%, Table 1). The characteristics of patients included in the point prevalence study and the antibiotic used are shown in Table 1.
Table 1. Patients' characteristics and patterns of antibiotic prescribing for patients who received antibiotic treatment during the point prevalence surveys, May 2009
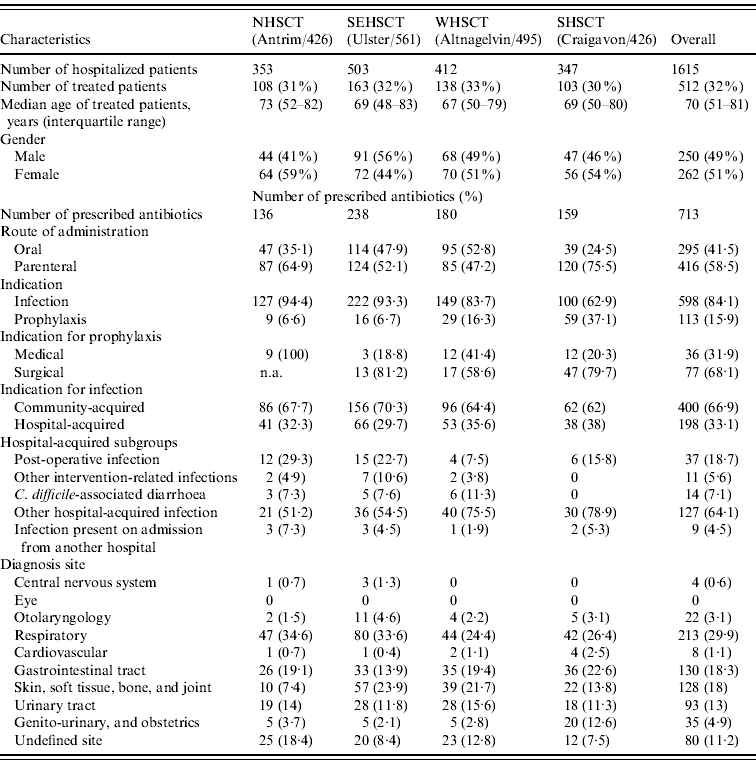
NHSCT, Northern Health and Social Care Trust; SEHSCT, South Eastern Health and Social Care Trust; WHSCT, Western Health and Social Care Trust; SHSCT, Southern Health and Social Care Trust.
n.a., Not applicable; surgical prophylaxis was not evaluated, for Antrim hospital, during this survey.
Out of the 198 antibiotics prescribed for hospital-acquired infections, 60·6% were in the medical department, 20·7% were in the surgical department, 10·1% were in the intensive care unit (ICU), and 8·6% were classified as ‘others’. Different trends in antibiotic use were observed across the studied hospitals (Table 2). The most commonly used antibiotics were combinations of penicillins including β-lactamase inhibitors (33·6%); this involved the use of amoxicillin-clavulanic acid (21·6%) and piperacillin- tazobactam (11·9%). The other most widely prescribed antibiotics were as follows: metronidazole (9·1%), macrolides (8·1%), glycopeptides (6·3%), gentamicin (6%), and penicillins with an extended spectrum (5·3%). The use of second-generation cephalosporins, third-generation cephalosporins, and fluoroquinolones were low (0·8%, 1%, 3·9%, respectively). Of the total prescribed antibiotics, 53·5% were prescribed in medical departments, followed by 30·3%, 10·7%, 5·5% in surgical, other, and ICU departments, respectively (Table 3). Different trends in antibiotic use were observed across the studied specialities (Table 3).
Table 2. Trends in antibiotic use, for each Trust, in patients who received antibiotic treatment during the point prevalence survey May 2009
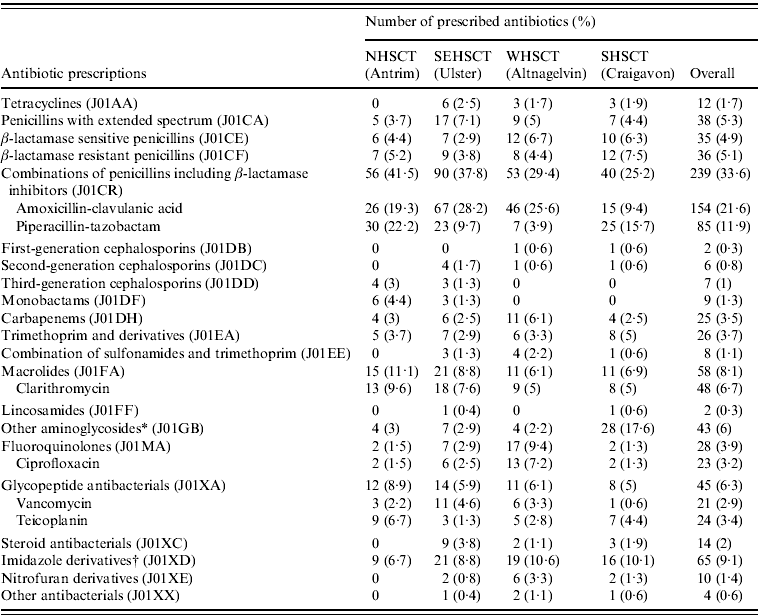
NHSCT, Northern Health and Social Care Trust; SEHSCT, South Eastern Health and Social Care Trust; WHSCT, Western Health and Social Care Trust; SHSCT, Southern Health and Social Care Trust.
* Other aminoglycosides=gentamicin.
† Imidazole derivatives=metronidazole.
Table 3. Trends in antibiotic use in patients who received antibiotic treatment, according to specialities, during the point prevalence survey, May 2009
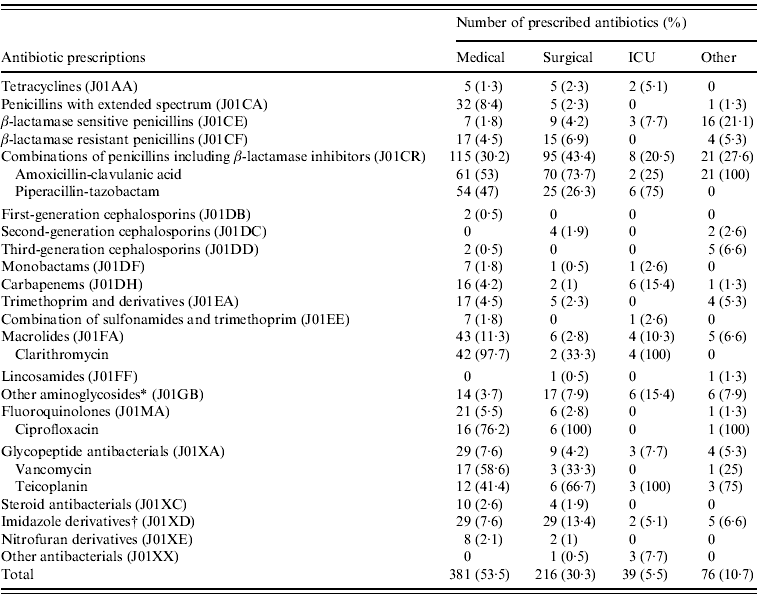
ICU, Intensive care unit.
* Other aminoglycosides=gentamicin.
† Imidazole derivatives=metronidazole.
In the total sample, the indication for treatment was recorded in 84·3% of the prescribing episodes. A small fraction (3·9%) of the surgical prophylactic antibiotic prescriptions was for >24 h. The results of the survey showed that overall 52·4% (compliant, n=331; not compliant, n=101; not assessable, n=200) of the prescribed antibiotics were in compliance with the hospital antibiotic guidelines.
Although resistance is a worldwide concern, it is a local problem in the first instance as selection for, and spread of, resistant microorganisms occurs in individual hospitals and communities [Reference MacDougall and Polk4]. Thus, assessing local patterns of antibiotic prescribing forms an important basis for informing robust antibiotic stewardship. The objective of this research was first to assess current patterns of antibiotic prescribing in secondary healthcare settings in Northern Ireland, and second to determine targets for improving the quality of antibiotic prescribing. The antibiotic use prevalence in the hospitalized patients surveyed (32%) was similar to other studies [Reference Seaton6, Reference Ansari7]. Of note, a high prevalence of antibiotic use for treating infections (84·1%) was related to treating community-acquired infections (66·9%), highlighting the burden of community-acquired infections on healthcare resources and the importance of addressing infections more robustly within primary care.
Since local prescribing guidelines for the management of infection were not uniform across the studied sites, relative differences in prescribing were observed between hospital sites (Table 2). The variability between the participating hospitals in prescribing practices (Tables 2, 3) may require further work using risk-adjusted models, which may explain a significant proportion of the variation and allow for inter-hospital comparisons for benchmarking purposes [Reference MacDougall and Polk9]. Interestingly, all study sites demonstrated similar usage patterns in relation to specific antibiotic classes. For example, the use of second-generation cephalosporins, third-generation cephalosporins and fluoroquinolones in the four study sites were low. This represents good clinical practice since the use of these antibiotics has been shown to be implicated in an increased occurrence of C. difficile infection (CDI) and methicillin-resistant Staphylococcus aureus (MRSA) [Reference Aldeyab10, Reference Aldeyab11]. It is clear that the study sites favoured the use of combinations of penicillins including β-lactamase inhibitors and macrolides; compliance rates with the Trust's antibiotic guidelines regarding the use of amoxicillin-clavulanic acid were the lowest (47·2%). In two recent investigations conducted in the Antrim Area Hospital [Reference Aldeyab10, Reference Aldeyab11], the use of amoxicillin-clavulanic acid and macrolides was, however, also shown to be a risk factor for the development of hospital-acquired MRSA and CDI. Thus special attention should be devoted to optimize the use of these latter antibiotics.
The importance of the current point prevalence study lies in identifying three key targets for quality improvement in Northern Ireland. Of note, our findings were consistent with other published ESAC reports, confirming the value and the generalizability of the data from these types of surveys [Reference Zarb and Goossens12]. First, although there was a high rate of reporting on indications for treatments in patient notes (84·3%), which represents good clinical practice, it is recommended that >95% should be expected [Reference Ansari7]. Second, duration of surgical prophylaxis has been identified as a target for quality improvement, where the target rate for duration of prophylaxis should not be >24 h [Reference Ansari7]. The overall rate in the study site hospitals (3·9%) was almost in line with these recommendations; however, the target rate for duration of prophylaxis >24 h should be 0%. Third, the value of antibiotic guidelines in decreasing antibiotic use and associated cost has been documented [Reference Davey13]. The overall adherence rate to hospital guidelines in this study (52·4%) was low, and full compliance with the guidelines should be targeted.
The survey involved the main hospitals in four of the five HSC Trusts in Northern Ireland and as such provided a broad perspective of antibiotic use in the province. Nonetheless, observed prescribing may not consistently reflect antibiotic prescribing practices within the studied hospitals due to the nature of a point prevalence design. The study should be repeated on a regular basis to facilitate the documentation of changes in antibiotic prescribing, thus, guiding local antibiotic stewardship.
In conclusion, the study highlights a number of interesting issues related to hospital antibiotic guidelines. First, the study demonstrated the benefits of the application of a simplified audit tool (i.e. the ESAC audit tool) in providing comprehensive details regarding antibiotic prescribing patterns in hospitals. Second, the results of the study strongly suggest that antibiotic use could be improved by taking steps to address targets for quality improvement which have been alluded to earlier. Finally, point prevalence surveys may be considered beneficial in optimizing antibiotic therapy in secondary healthcare settings.
ACKNOWLEDGEMENTS
We acknowledge the contribution of Professor Peter Davey as Chair of the UK ESAC Network and the lead for the Hospital Care Subproject.
DECLARATION OF INTEREST
None.





