Parkinson’s disease (PD) and dementia with Lewy bodies (DLB), together referred to as Lewy body disease, are neurodegenerative synucleinopathies that lead to parkinsonism and dementia. Neuropsychiatric manifestations—including hallucinations, delusions and behavioural symptoms—may respond to treatment with antipsychotic medication. However, this class of medication may worsen motor functioning and may be associated with increased mortality in patients with Lewy body disease and in elderly patients with dementia without parkinsonism.Reference Weintraub, Chiang and Kim 1 , Reference Grimes, Gordon and Snelgrove 2
The STOPP (Screening Tool of Older Persons’ Prescriptions) criteria were developed to identify potentially inappropriate prescribing practices. One criterion (version 2, section D6) identifies antipsychotics (i.e., other than quetiapine or clozapine) in those with parkinsonism or Lewy body disease as a potentially inappropriate prescribing practice because of the risk of severe extrapyramidal side effects.Reference O’Mahony, O’Sullivan, Byrne, O’Connor, Ryan and Gallagher 3
Several evidence-informed treatment guidelines recommend against the use of typical antipsychotics in patients with Lewy body disease and recommend the use of atypical antipsychotics, quetiapine and clozapine.Reference Grimes, Gordon and Snelgrove 2 , Reference Ferreira, Katzenschlager and Bloem 4 , Reference Miyasaki, Shannon and Voon 5 Clozapine requires intensive blood monitoring and is therefore not commonly selected.Reference Marras, Kopp and Qiu 6
The Nova Scotia Seniors Pharmacare Program (NSSPP) is a provincial drug insurance program for residents aged 65 years and above who are without other prescription drug coverage. In 2013, the NSSPP provided benefits for approximately 107,800 of 166,900 (64.6%) persons aged 65 or older in the province.Footnote a The purpose of the present study is to determine the frequency with which potentially inappropriate antipsychotics are dispensed to patients on levodopa in the NSSPP population.
NSSPP prescription claims data from 1 April 2009 to 31 March 2014 were employed. Beneficiaries with a claim for an antipsychotic medication (>30 day supply) were identified. Concomitant prescribing (>30 days) of an antipsychotic and a levodopa-containing medication were identified.Footnote b An 80% medication adherence was calculated to ensure regular use of a medication.Footnote c For those dispensed more than one antipsychotic agent during the study period, only the first was included in the analysis.
Antipsychotics were categorized as preferred atypical (quetiapine or clozapine), potentially inappropriate (second-line atypical), or potentially harmful (typical antipsychotics) in line with evidence-informed treatment guidelines in North America and Europe.Reference Grimes, Gordon and Snelgrove 2 , Reference Ferreira, Katzenschlager and Bloem 4 , Reference Miyasaki, Shannon and Voon 5
Descriptive statistics were utilized to describe antipsychotic dispensations in those with concomitant claims for levodopa-containing medications. Antipsychotic prescriptions were categorized by treatment classification and beneficiaries, with each classification summarized by age and sex category. Logistic regression was used to model the probability of being prescribed a preferred atypical antipsychotic compared to an inappropriate or harmful antipsychotic medication. The covariates included in the model were age, sex and the interaction of age and sex. Prescribed daily dose mean ± standard deviation and interquartile range for antipsychotic medications were calculated.Footnote d , Footnote e For calculation of prescribed daily dose, it was assumed that all doses were oral; however, a small number of oral liquids and non-oral formulations may be included in the data.Footnote f
All analyses were performed using SAS/STAT software (v. 9.2, SAS Institute Inc., Cary, North Carolina).
Our cohort included 294 beneficiaries (58% male) who made concomitant claims for an antipsychotic (Table 1) and levodopa-containing medications. Among this cohort, 59.9% (n=176) were dispensed a preferred atypical antipsychotic, 27.6% (n=81) a potentially inappropriate antipsychotic and 12.6% (n=37) a potentially harmful antipsychotic (Table 2). The prescribing practices did not appear to differ significantly according to patient age, gender or year the medication was dispensed, except in those 80 years of age or older, and females were less likely to receive a preferred atypical antipsychotic (unadjusted OR=0.240, CI 95%=0.105-0.547, p=0.0007) (Tables 2 and 3). With an adjustment for multiple comparisons, none of the groups remained statistically significant. Dosages are presented in Table 4.
Table 1 Antipsychotic medications dispensed to Nova Scotia Seniors Pharmacare Program beneficiaries concomitantly with levodopa-containing medications (N=294)

Table 2 Nova Scotia Seniors Pharmacare Program beneficiaries dispensed concomitant antipsychotic medication and levodopa-containing medications, by age and sex, categorized by treatment appropriateness
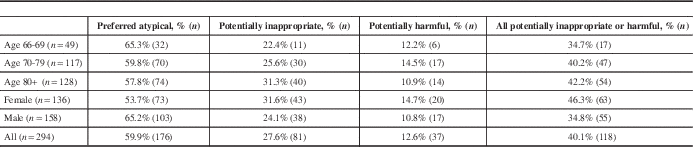
Table 3 Nova Scotia Seniors Pharmacare Program beneficiaries dispensed antipsychotic medications concomitant with levodopa-containing medications, by year, categorized by treatment appropriateness

Table 4 Antipsychotic dosageFootnote * in NSSPP patients with concomitant levodopa-containing medication claims
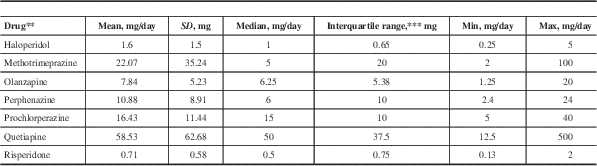
* For calculation of prescribed daily dose, it was assumed that all doses are oral; however, a small number of oral liquids and non-oral formulations may be included in the data.
** Clozapine, chlorpromazine, fluphenazine, loxapine, and trifluoperazine dosages have not been reported due to small sample size.
*** Interquartile range is calculated as the 75th percentile minus the 25th percentile.
Only 59.9% of this cohort were dispensed a preferred atypical antipsychotic, and a potentially harmful agent was dispensed to 12.6%. Several population-based studies suggest that, since the mid-90s, antipsychotic prescribing practices in patients with PD/DLB are changing. Between 1992 and 1996, only 1% of antipsychotics prescribed in a cohort of nursing home residents with PD in the United States were atypical agents.Reference Lapane, Fernandez and Friedman 7 A population-based study from the NetherlandsReference van de Vijver, Roos, Jansen, Porsius and de Boer 8 found that only 16% of antipsychotics dispensed to patients with PD between 1991 and 1998 were atypical agents. A Canadian studyReference Marras, Kopp and Qiu 6 pertaining to antipsychotic prescribing practices in PD from Ontario in 2002 noted that an atypical antipsychotic was selected as the initial prescription for 91% of older individuals with PD (which represents an increase from 44% in 1998). However, there appear to be differences in prescribing practices between countries. In the year prior (2001), a study from TaiwanReference Wang, Lian, Yeh, Yen, Ma and Chan 9 indicated that only 11% of a cohort of PD patients who were initially prescribed an antipsychotic were given an atypical agent.
Though most patients in our study were dispensed an atypical antipsychotic (87.5%), the STOPP criteria hold clinicians to a stricter standard. STOPP identifies any antipsychotic other than quetiapine or clozapine as potentially inappropriate. By those terms, only ~60% of our cohort received an appropriate agent. In the 2002 Canadian cohort reported by Marras et al.,Reference Marras, Kopp and Qiu 6 the proportion prescribed quetiapine was only 38% (clozapine was not analyzed but was felt to be rarely used). Clearly, there is an opportunity for education and improvement in prescribing practices.
The application of tools such as the STOPP criteria to health administrative databases allows valuable feedback about prescribing practices in populations, some of which may be modifiable. Inappropriate prescribing in older patients can lead to increased morbidity and mortality, hospitalizations and healthcare costs. Many strategies have been proposed to help healthcare professionals improve prescribing. These include educational opportunities as well as computerized screening and clinical decision support tools.Reference Cahir, Fahey, Teeling, Teljeur, Feely and Bennett 10
Our study has some limitations. Detailed clinical information about beneficiaries was not available, therefore, claims for levodopa-containing medications were used as a proxy to identify patients with parkinsonism. Other parkinsonian medications—for example, dopamine agonists, monoamine oxidase B inhibitors, anticholinergics and amantadine—may have other indications.
Neither the severity of motor impairment nor the nature of the psychiatric illness was known. This lack of clinical data limits the ability to assess the appropriateness of prescribing practices in individual clinical circumstances. For example, a typical antipsychotic may be considered appropriate even though not in line with the guidelines when psychosis poses a risk to self or others. Furthermore, some of the beneficiaries may have had drug-induced parkinsonism as a result of antipsychotic use for psychiatric illness rather than as a primary neurodegenerative disorder. However, even in cases of drug-induced parkinsonism, an atypical antipsychotic would generally be preferred over a typical agent.
This study suggests that potentially inappropriate prescribing practices are common in the neuropsychiatric management of patients with parkinsonism, even allowing for circumstances in which there is an appropriate deviation from the STOPP criteria (i.e., full compliance would not be expected on a population-wide scale). There is a need to better understand the patient, clinician and system factors that influence prescribing behaviours in this context so that targeted interventions with maximal impact can be implemented.
Disclaimer
The data (or portions of the data) used in this report were made available by Health Data Nova Scotia of Dalhousie University. Although this research is based on data obtained from the Nova Scotia Department of Health and Wellness, the observations and opinions expressed are those of the authors and do not represent those of either Health Data Nova Scotia or the Nova Scotia Department of Health and Wellness.
Disclosures
Heather Rigby and Kara Matheson have nothing to disclose. Ingrid Sketris has received salary support in part from the Canadian Institutes of Health Research (CIHR) through the Canadian Network for Observational Drug Effect Studies (CNODES). Sara Rehan and Barbara Hill-Taylor have received salary support in part from the Drug Evaluation Alliance of Nova Scotia (DEANS).






